The mechanism of electrophilic addition to dienes is similar to what we learn in the addition reactions of alkenes. The difference here is that there are two double bonds, and the question you may start wondering about is:
“Which of the dienes am I supposed to protonate, and would it follow Markovnikov’s rule?”
The short answers to these are:
You need to protonate both double bonds.
Yes, it does follow Markovnikov’s rule (kind of).
However, there are some details that must be considered for different types of dienes and the conditions the reaction is carried out.
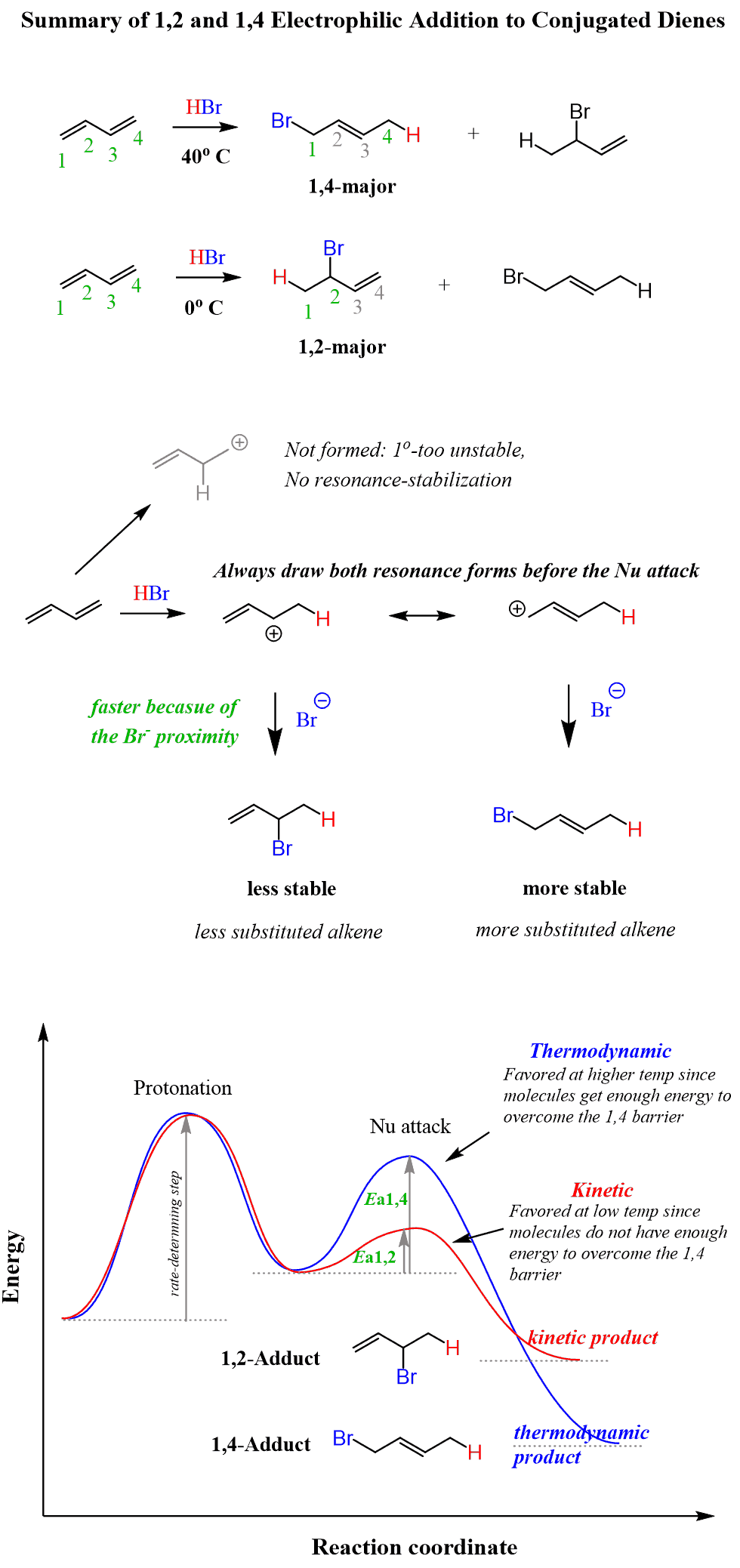
Let’s go into some details, starting from the regiochemistry of the reaction. You can pick any of the double bonds and protonate it on both carbons to compare which of the resulting carbocations is more favorable:
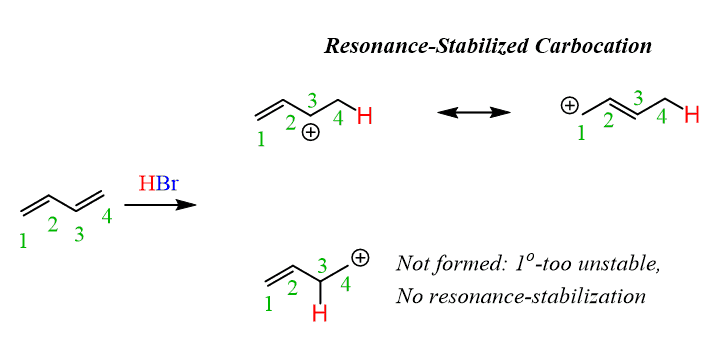
The first one is preferred since it is a secondary carbocation and, in addition, it is resonance-stabilized.
Next, draw the resonance structure of this carbocation to attack both of them by the bromide ion.
Two alkenes are formed, and which one do you think is more favorable?
Think about the stability of alkenes.
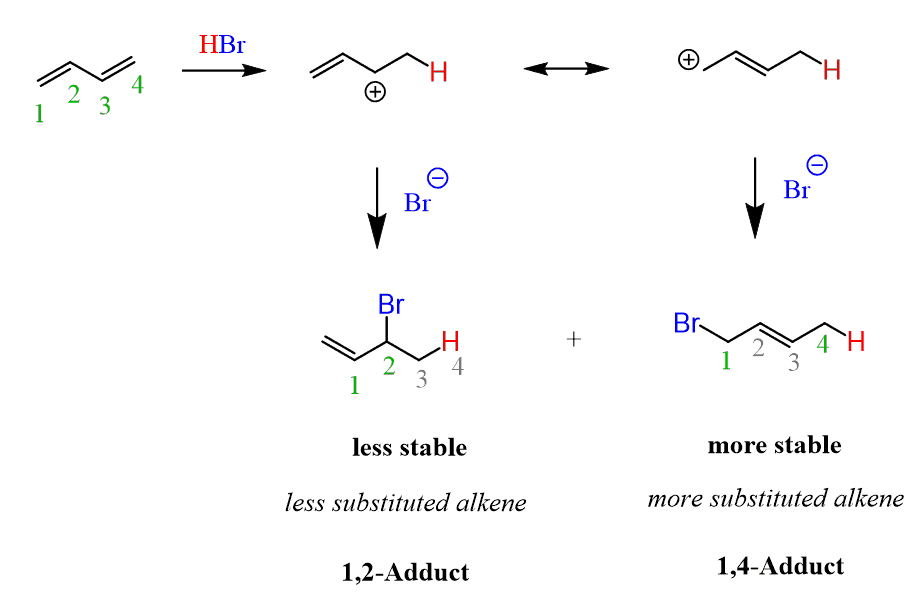
The first alkene is monosubstituted, while the second one is disubstituted. Therefore, it is more stable and is expected to be the major product.
Let’s number the carbons to distinguish these products more easily. The more stable product is called a 1,4-adduct, and the less stable product is the 1,2-adduct. They are formed by a 1,4- and 1,2-addition, respectively.
The 1,4 adduct is the thermodynamic product of the reaction since it is the more stable product.
The Effect of Temperature
The regiochemistry changes when the reaction is carried out at lower temperatures. The 1,2-addition starts predominating because of the proximity effect. We are referring to the proximity of the Br– ion to the carbocation right after the protonation of the double bond occurs:
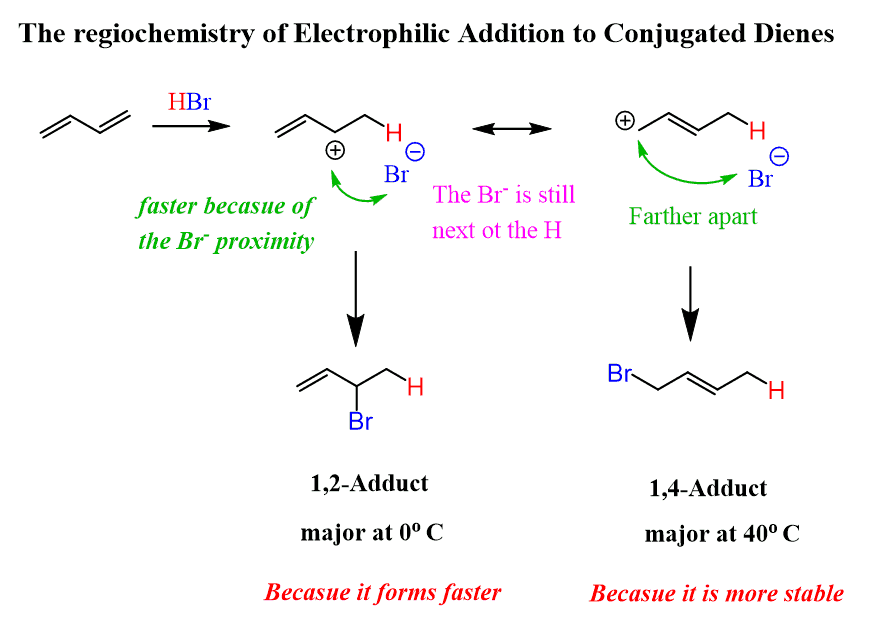
You can think of it this way: the nucleophilic attack of the Br– occurs faster as it does not have to wait for the formation of the second resonance structure (remember, however, that resonance structures are formalities), and it can simply reach the carbon next to it. Therefore, the 1,2 adduct is the kinetic product as it forms faster but is less stable (check the energy diagram above).
Remember, the kinetic product is generally favored at lower temperatures since the molecules do not have sufficient energy to overcome the energy barrier (activation energy) and form the more stable (thermodynamic product).
Here is the summary of the electrophilic addition to conjugated alkenes, considering the regiochemistry at low and higher temperatures, explained by the corresponding energy diagrams:
How to Predict All the Products of Electrophilic Additions to Diene
In general, to predict the products of an electrophilic addition to a diene, you need to identify whether the diene is symmetrical or unsymmetrical.
If it is symmetrical, you only need to protonate one of the double bonds since both double bonds result in the same products.
If it is unsymmetrical, then you need to consider the protonation of both double bonds.
In each case, you also need to draw both resonance forms of the resulting carbocation for the nucleophilic attack.
All of these, plus the stereochemistry of the addition reactions to dienes, are summarized in the following section.
The Stereochemistry of Electrophilic Additions to Dienes
Since the addition reaction to the diene goes through the formation of a carbocation, the nucleophilic attack occurs from both faces of the sp2-hybridized carbon, and a new chirality center is formed as a racemic mixture.
Here is a summary for predicting the products illustrating the Stereochemistry of Electrophilic Additions to Diene:

Electrophilic Additions to Unsymmetrical Dienes
It becomes a bit more complicated when the diene is not symmetrical, since you now need to consider the protonation of both double bonds. For example, in penta-1,3-diene, there are four carbons with a double bond, and protonating all of them will lead to a very large number of molecules.
However, the good news is that you only need to protonate the ones that form resonance-stabilized carbocations!

Summarizing Electrophilic Additions to Dienes
Electrophilic addition to conjugated dienes follows similar principles to alkene additions, but with some important twists:
- Both double bonds can be protonated, so you must consider multiple possibilities.
- Markovnikov’s rule still applies, which aligns with the formation of resonance-stabilized carbocations.
- Two major products are typically formed:
- 1,2-adduct (kinetic product): Forms faster, favored at low temperatures
- 1,4-adduct (thermodynamic product): More stable, favored at higher temperatures
To predict the outcome:
- Check for symmetry to simplify your work, as, in this case, you don’t need to worry about which double bond protonated.
- Draw resonance forms of the carbocation to identify all possible points of nucleophilic attack.
- Analyze alkene stability to determine the thermodynamic product.
- Account for stereochemistry, since addition to carbocations gives racemic mixtures at new chiral centers.
When working with unsymmetrical dienes, protonate the double bonds such that resonance-stabilized carbocations are generated. The addition of the halide to these carbocations leads to the major products.





I didn’t know there was so much to this… thanks
Yes, these can get overwhelming especially if stereochemistry needs to be considered too.