The Kiliani–Fischer synthesis lengthens the carbon chain of carbohydrates by adding one carbon to the aldehyde group of an aldose.
For example, using this method, D-arabinose can be converted into a mixture of D-glucose and D-mannose:
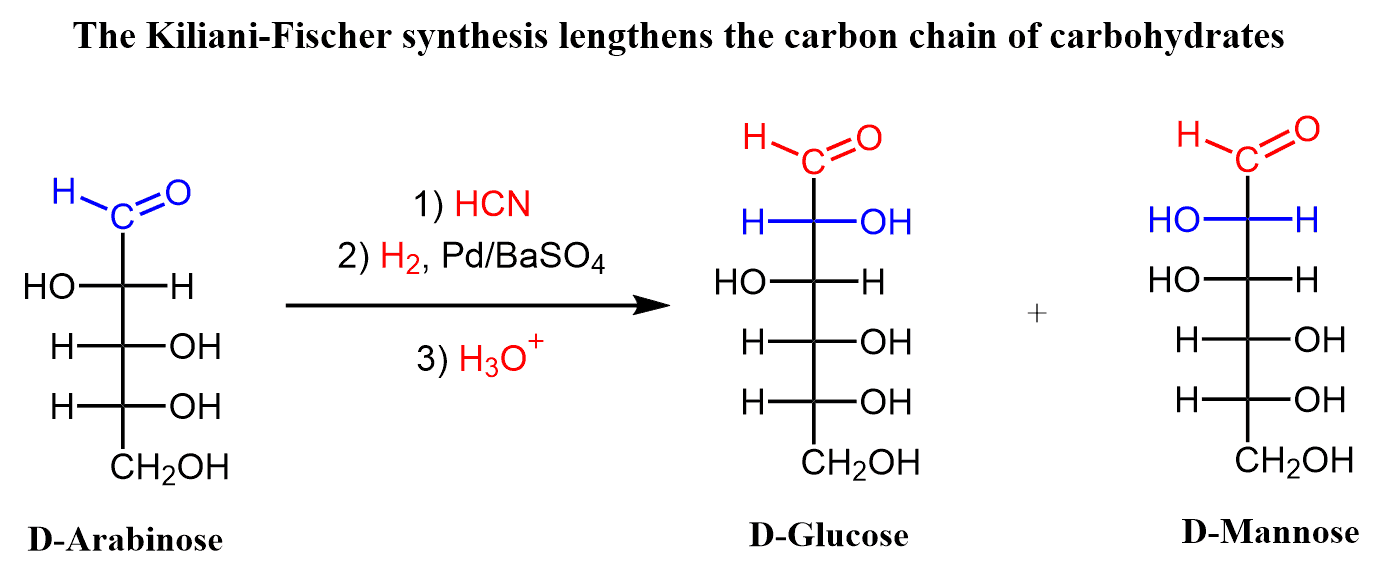
Notice that two epimers are formed as a product since they only differ at the new stereogenic center at C2.
So, let’s now understand the mechanism behind the formation of these epimers. In the first part of the Kiliani–Fischer synthesis, we have an addition reaction of the cyanide to the aldehyde forming a cyanohydrin. This is when a new carbon is added to the carbohydrate chain:

Remember, we have seen this quite often in the synthesis of carboxylic acids and their derivatives. The nitrile group is a good electrophile that can further be reacted with a variety of nucleophiles such as Grignard or Organocuprate nucleophiles.
Now, in this particular synthesis, the nitrile group is reduced to an imine using H2 and Pd-BaSO4 catalyst, and in the last step, we only need to convert the imine to an aldehyde by an acidic hydrolysis:
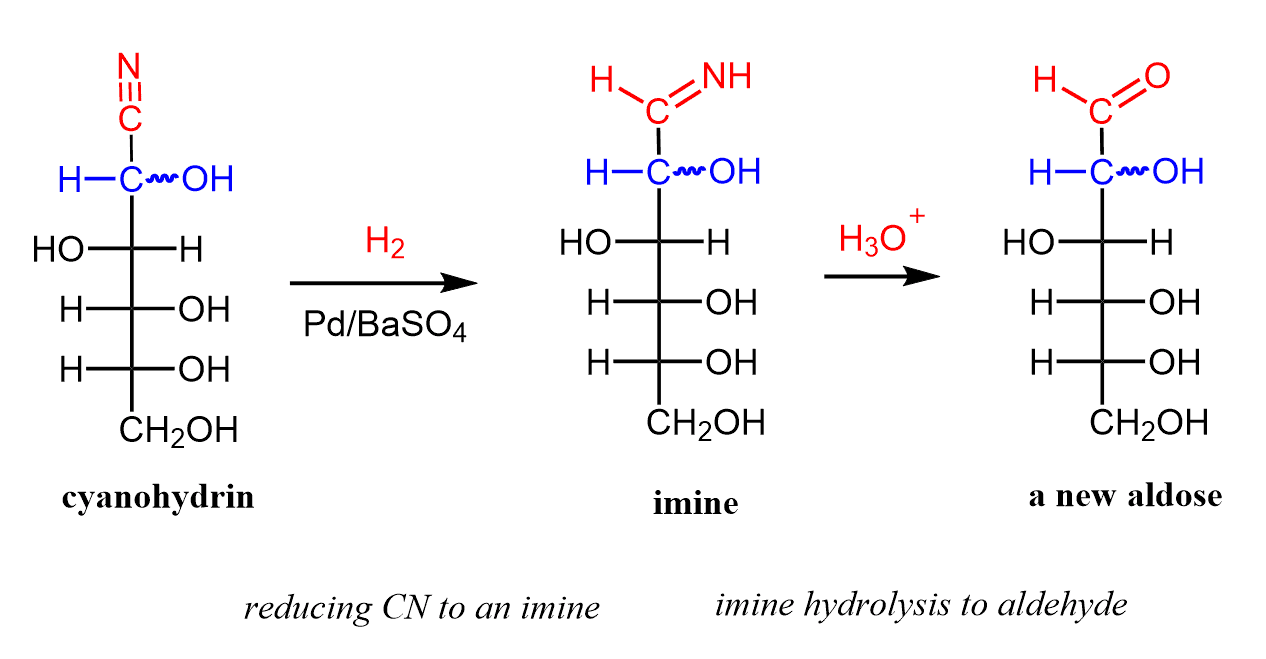
Let’s put these steps into a little summary of the Kiliani–Fischer synthesis which increase the carbohydrate chain of an aldose by on carbon atom:

Need some practice on carbohydrates?
Check this Multiple-Choice, summary quiz on the structure and reactions of carbohydrates with a 40-min video solution!
Carbohydrates Practice Problem Quiz
Check also in Carbohydrates
- Carbohydrates – Structure and Classification
- Erythro and Threo
- D and L Sugars
- Aldoses and Ketoses: Classification and Stereochemistry
- Epimers and Anomers
- Converting Fischer, Haworth, and Chair forms of Carbohydrates
- Mutarotation
- Glycosides
- Isomerization of Carbohydrates
- Ether and Ester Derivatives of Carbohydrates
- Oxidation of Monosaccharides
- Reduction of Monosaccharides
- Kiliani–Fischer Synthesis
- Wohl Degradation


This is so helpful.
Thanks indeed
Thanks, Anthony.