Let’s see how the ether and ester derivatives of carbohydrates are prepared and used in organic synthesis. We have seen that carbohydrates undergo different epimerization and isomerization reactions in basic aqueous solutions and are therefore not stored under basic conditions. Aside from this, if we look at a carbohydrate molecule even in its cyclic form, where there is no risk of deprotonation at the ɑ position, it still has lots OH groups which are alcohols and quite reactive towards many functional groups and reagents. Because of this, in certain cases, it is desired to protect them just like we use protecting groups for other organic molecules. Typical protecting groups for the OH groups in carbohydrates are ethers, esters, and acetals.
Methyl and primary ethers of carbohydrates can be prepared by the Williamson ether synthesis. Remember, the Williamson ether synthesis involves conversion of alcohols into alkoxide ions using a strong base such as a hydride because alkoxides are better nucleophiles favoring the SN2 mechanism which gives a higher yield:

However, this exact strategy is often not suitable for carbohydrates because of the undesired reactions of isomerization that carbohydrates undergo in the presence of a strong base.
To overcome this issue, a mild base such as silver oxide is used. Ag2O deprotonates all the alcohol groups forming alkoxides which readily react with CH3I in an SN2 reaction:

Although all the ether groups look identical, one of them (on the traditional anomeric carbon) is part of an acetal and can be hydrolyzed back to the alcohol as we have seen in the discussion about glycosides.
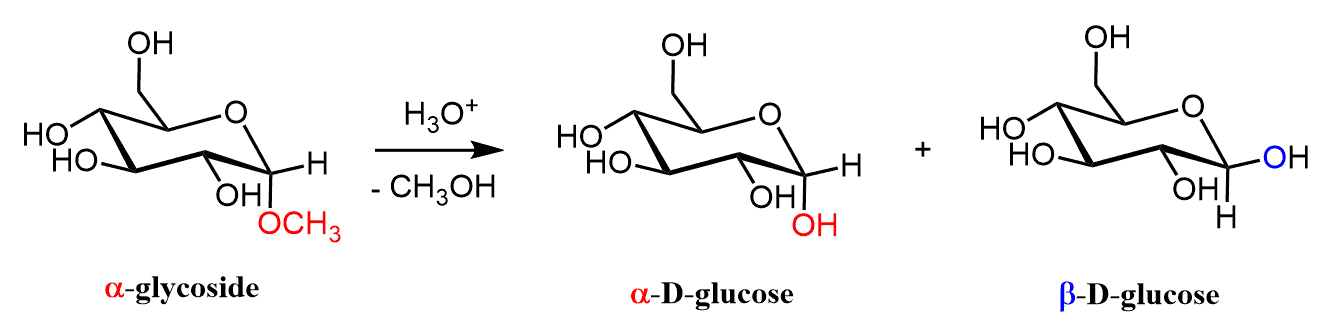
The other four, on the other hand, are very stable and just like regular ethers can only be cleaved with strong acids such as HBr and HI.
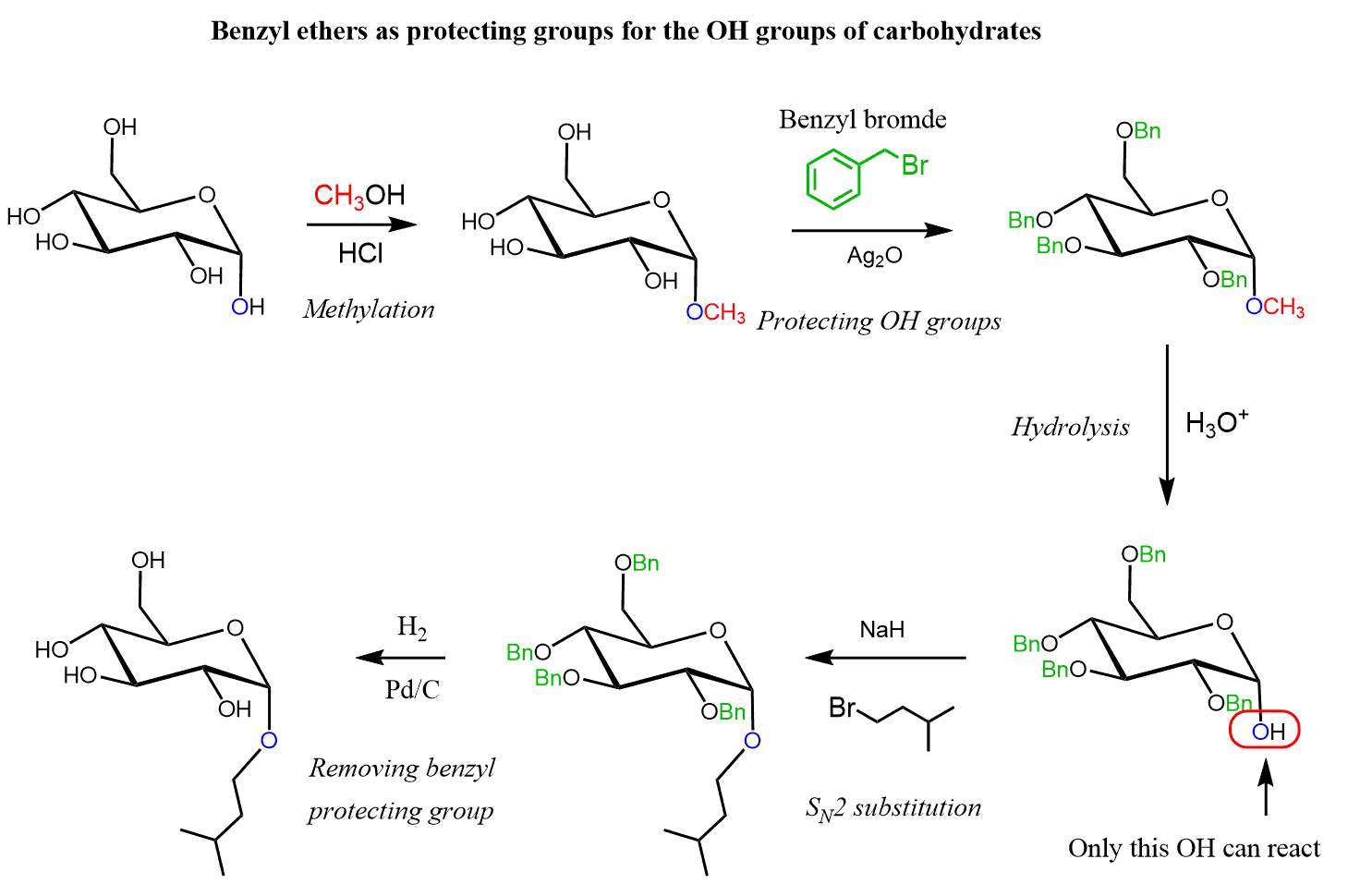
In search of better protecting groups for alcohols, it was found that benzyl ethers are suitable since they are stable under acidic and basic conditions and can be cleaved by catalytic hydrogenation with H2 over Pd/C. Benzyl ethers of carbohydrates are often used in the synthesis of carbohydrates:
Esterification of Carbohydrates
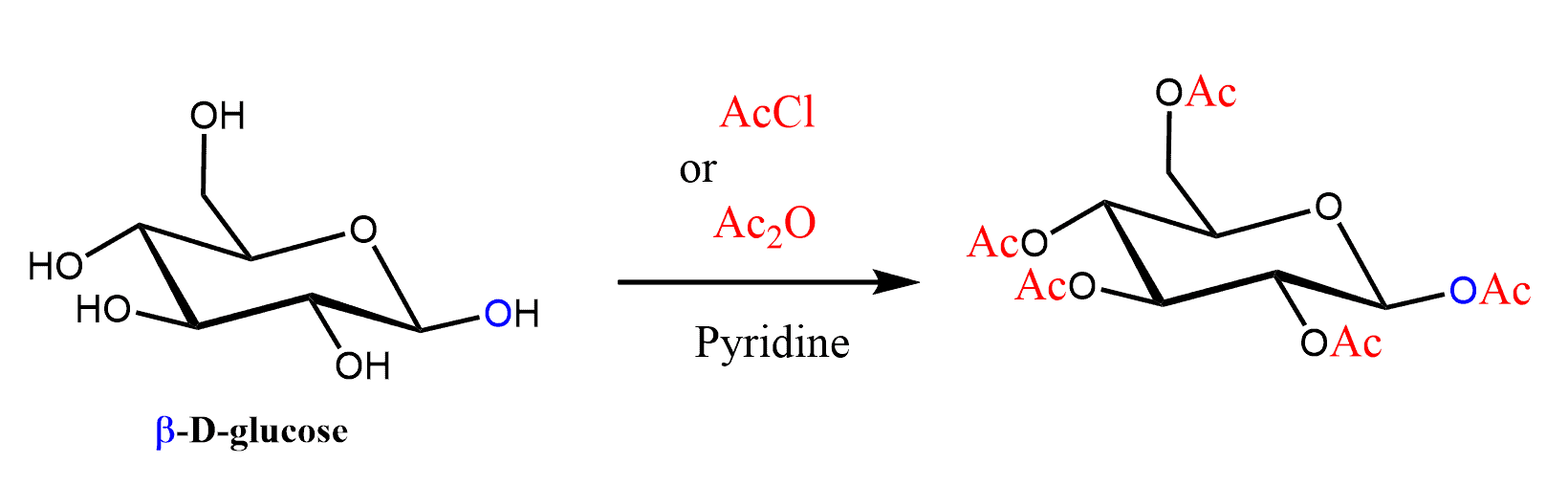
The OH groups of monosaccharides, like those of other alcohols, can be converted to esters by reacting with either anhydrides or acid chloride in the presence of a base. Most often, acetic anhydride and pyridine (a base) are used for the esterification of carbohydrates.

The acetyl group is often abbreviated as Ac to save space and time especially when drawing large molecules like carbohydrates:
Ester derivatives of carbohydrates can be converted back to alcohols by saponification in base or transesterification using alkoxide ion of a different alcohol:

Need some practice on carbohydrates?
Check this Multiple-Choice, summary quiz on the structure and reactions of carbohydrates with a 40-min video solution!
Carbohydrates Practice Problem Quiz
Check also in Carbohydrates
- Carbohydrates – Structure and Classification
- Erythro and Threo
- D and L Sugars
- Aldoses and Ketoses: Classification and Stereochemistry
- Epimers and Anomers
- Converting Fischer, Haworth, and Chair forms of Carbohydrates
- Mutarotation
- Glycosides
- Isomerization of Carbohydrates
- Ether and Ester Derivatives of Carbohydrates
- Oxidation of Monosaccharides
- Reduction of Monosaccharides
- Kiliani–Fischer Synthesis
- Wohl Degradation

