Why are they Called Addition Reactions?
Let’s first understand why the reactions of alkenes are called addition reactions. The general formula of alkenes is CnH2n which is two hydrogens less than alkanes (CnH2n+2). This means alkanes are saturated as there is no possible way of adding more hydrogens to alkanes:
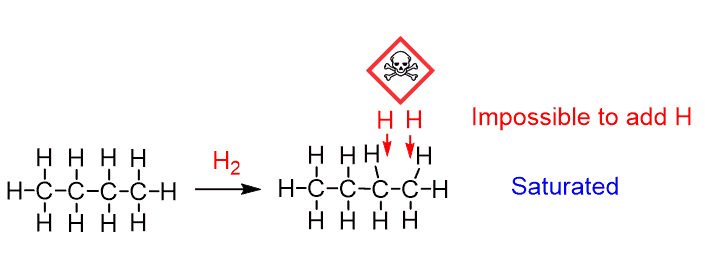
Alkenes, on the other hand, are not saturated because we can add two hydrogen atoms to the double bond and still not exceed the 4-bond limit of the carbon:
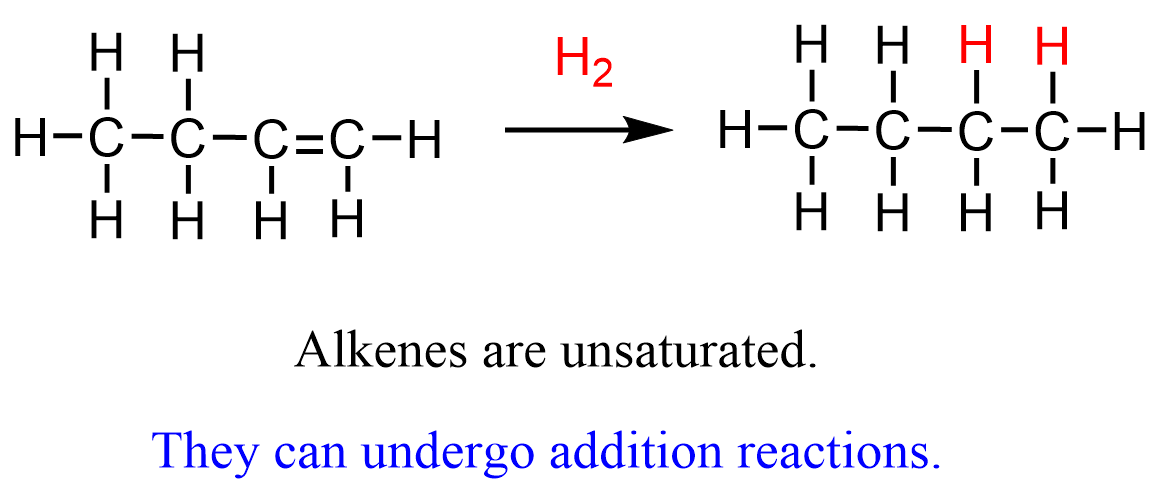
So, the hydrogens are literally added to the double bond converting it to two single bonds. Now, hydrogen is not the only molecule that adds across the double bond. The addition of hydrogen halides, water, halogens, and some other important species make the important class of electrophilic addition reactions to alkenes:

From the general name, the next question we need to address is why these are called electrophilic additions.
Why are they Called Electrophilic Additions?
An electrophile is a species that likes electrons, and this is not what alkenes do. Alkenes are electron-rich because of the extra two electrons in the double bond compared to a single bond.
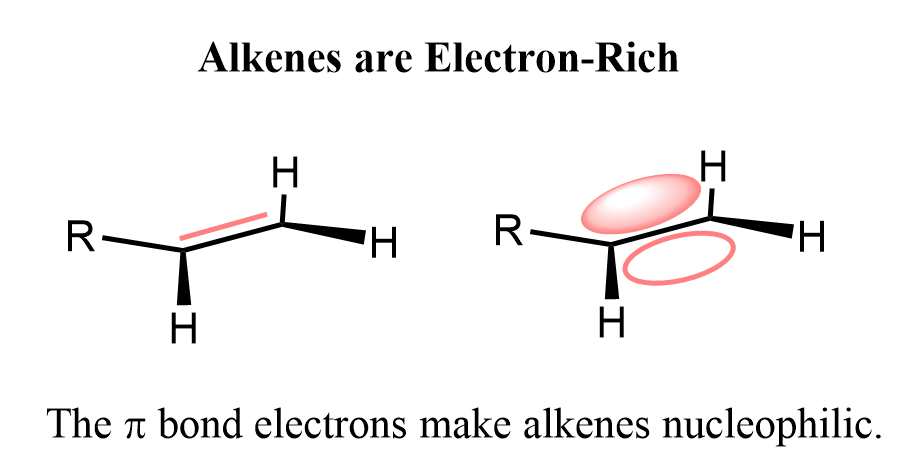
These are the electrons of the π bond, and we need to remember that π bonds are weaker than σ bonds. In other words, they are more reactive, so what we have is an electron pair that is quite reactive, and when encountered with electron-deficient species (electrophile), they are going to act as a nucleophiles. So, in a typical electrophilic addition reaction to an alkene, it is the nucleophile and the other species is the electrophile.
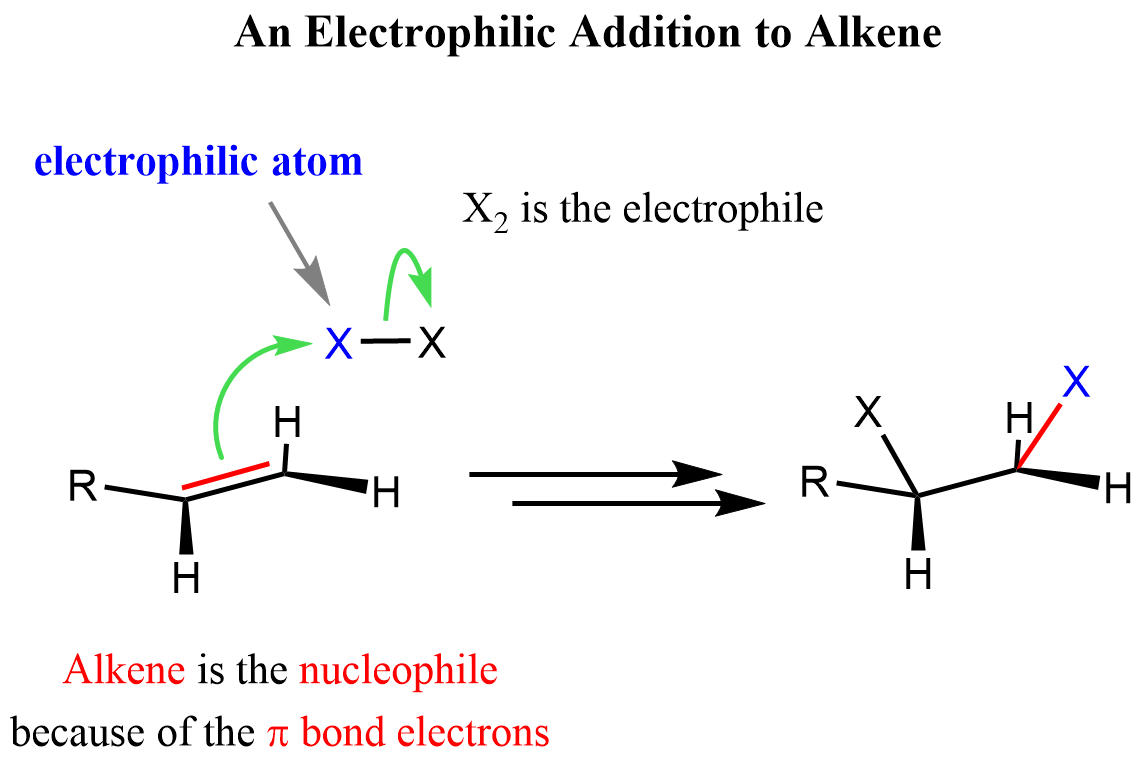
Double arrows indicate that it is not a one-step conversion, and in this particular case, the step is the addition of the other X atom.
As an example, let’s consider the addition of hydrohalic acids (HCl, HBr, and HI) to alkenes. In the first step, the alkene uses the electrons of the π bond to act as a nucleophile, and the acid is the electrophile. The halogen, like in many substitution reactions we saw earlier, is the leaving group:
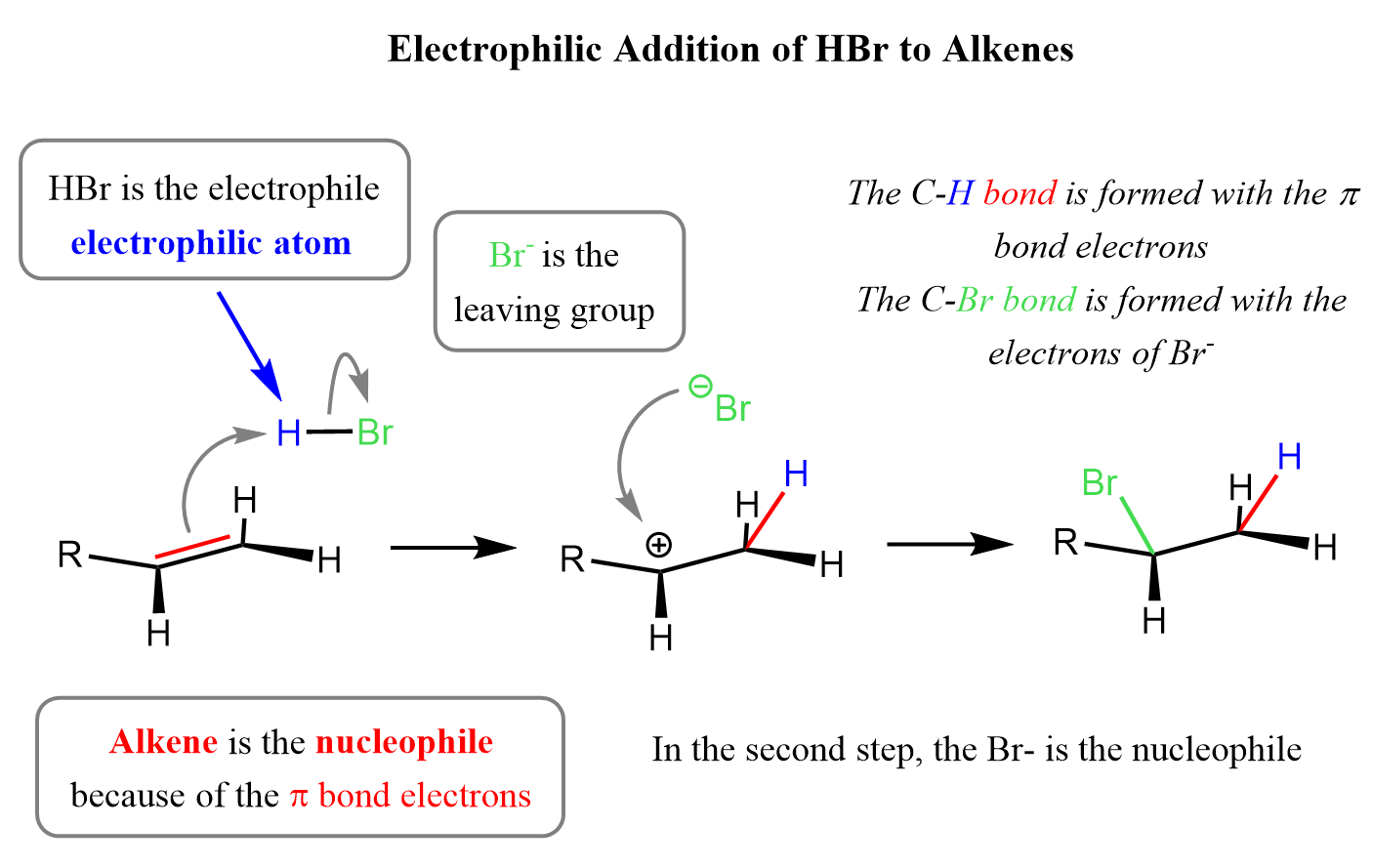
In the last step, the resulting carbocation intermediate is then attacked by the bromide ion, so in this last, step, it is the bromide ion that acts as a nucleophile. The rate-determining step of this electrophilic addition reaction is the first where the π bond is broken. The carbocation is very electron-deficient and is quickly attacked by the bromide ion.
The Regiochemistry of Electrophilic Addition Reactions
Notice that the halogen ends up on the more substituted carbon of the double bond. This is the regioselectivity of the hydrohalogenation reaction and it demonstrates the Markovnikov’s rule. It is an important pattern that you will see throughout the addition reactions of alkenes and alkynes. Markovnikov’s rule is followed when the X component of the HX reactant (for example HCl, HBr, HI, HOH) adds to the more substituted carbon atom. For example, acid-catalyzed hydration of alkenes follows the Markovnikov’s rule and adds the OH group to the more substituted carbon atom, whereas the hydroboration-oxidation does an anti-Markovnikov addition of water to alkenes:
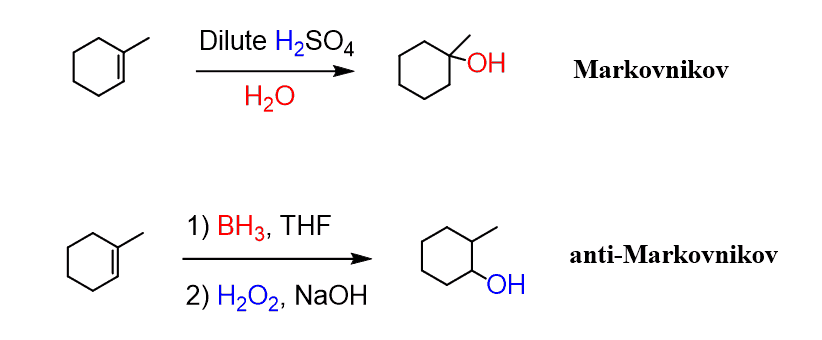
Although reactions such as hydroboration-oxidation follow a different mechanism, but the general theme is the same: The alkene is the nucleophile supplying the electrons for the electrophile addition of the other species.
There are other exceptions to the Markovnikov’s rule mainly due to rearrangements of the carbocation and also in some reactions, a catalyst such as a peroxide is used to alter the mechanism and regiochemical outcome of the reaction.

We will cover these in the next articles dedicated to the addition reactions of alkenes.
Check Also
- Markovnikov’s Rule
- Markovnikov’s Rule with Practice Problems
- Addition of Water to Alkenes
- Acid-Catalyzed Hydration of Alkenes with Practice Problems
- Rearrangements in Alkene Addition Reactions
- Oxymercuration-Demercuration
- Addition of Alcohols to Alkenes
- Free-Radical Addition of HBr: Anti-Markovnikov Addition
- Hydroboration-Oxidation: The Mechanism
- Hydroboration-Oxidation of Alkenes: Regiochemistry and Stereochemistry with Practice Problems
- Halogenation of Alkenes and Halohydrin Formation
- The Regiochemistry of Alkene Addition Reactions
- The Stereochemistry of Alkene Addition Reactions
- Cis product in an anti Addition Reaction of Alkenes
- Ozonolysis of Alkenes with Practice Problems
- Syn Dihydroxylation of Alkenes with KMnO4 and OsO4
- Anti Dihydroxylation of Alkenes with MCPBA and Other Peroxides with Practice Problems
- Oxidative Cleavage of Alkenes with KMno4 and O3
- Alkene Reactions Practice Problems
- Changing the Position of a Double Bond
- Changing the Position of a Leaving Group
- Alkenes Multi-Step Synthesis Practice Problems
- Alkene Addition Reactions Practice Quiz

Thanks so for your wise knowledge and now I Am competent with this chapter