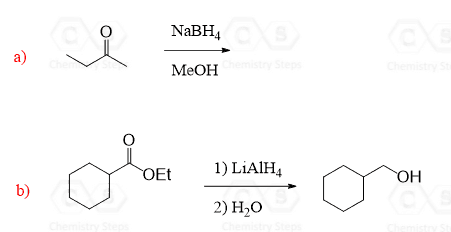
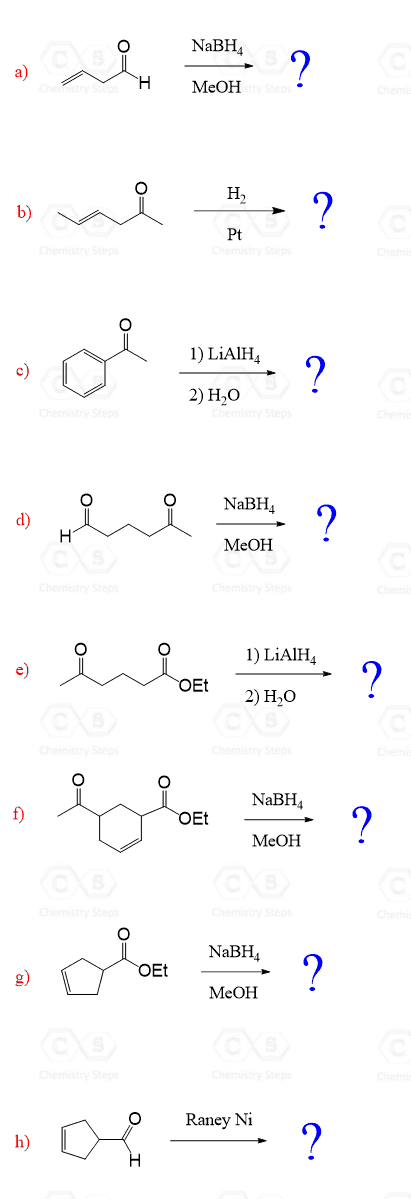
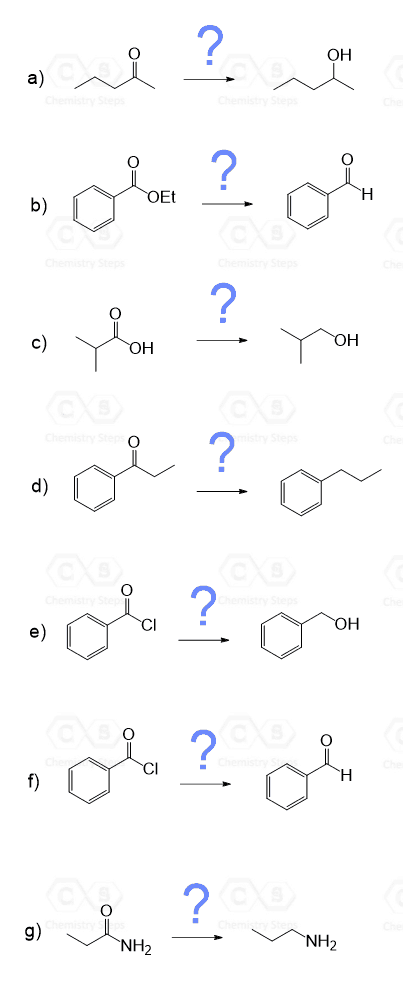
4.
Oximes (R₂C=NOH) are versatile intermediates in organic synthesis. When the following oxime is reacted with lithium aluminum hydride (LiAlH₄) in THF, the expected primary amine is formed together with the secondary amine shown below. Interestingly, when the reaction is carried out using hexamethylphosphoramide (HMPA) as the solvent, the corresponding ketone is formed. When sodium cyanoborohydride (NaBH₃CN) is used as the reducing agent, the hydroxylamine shown in the diagram is obtained.
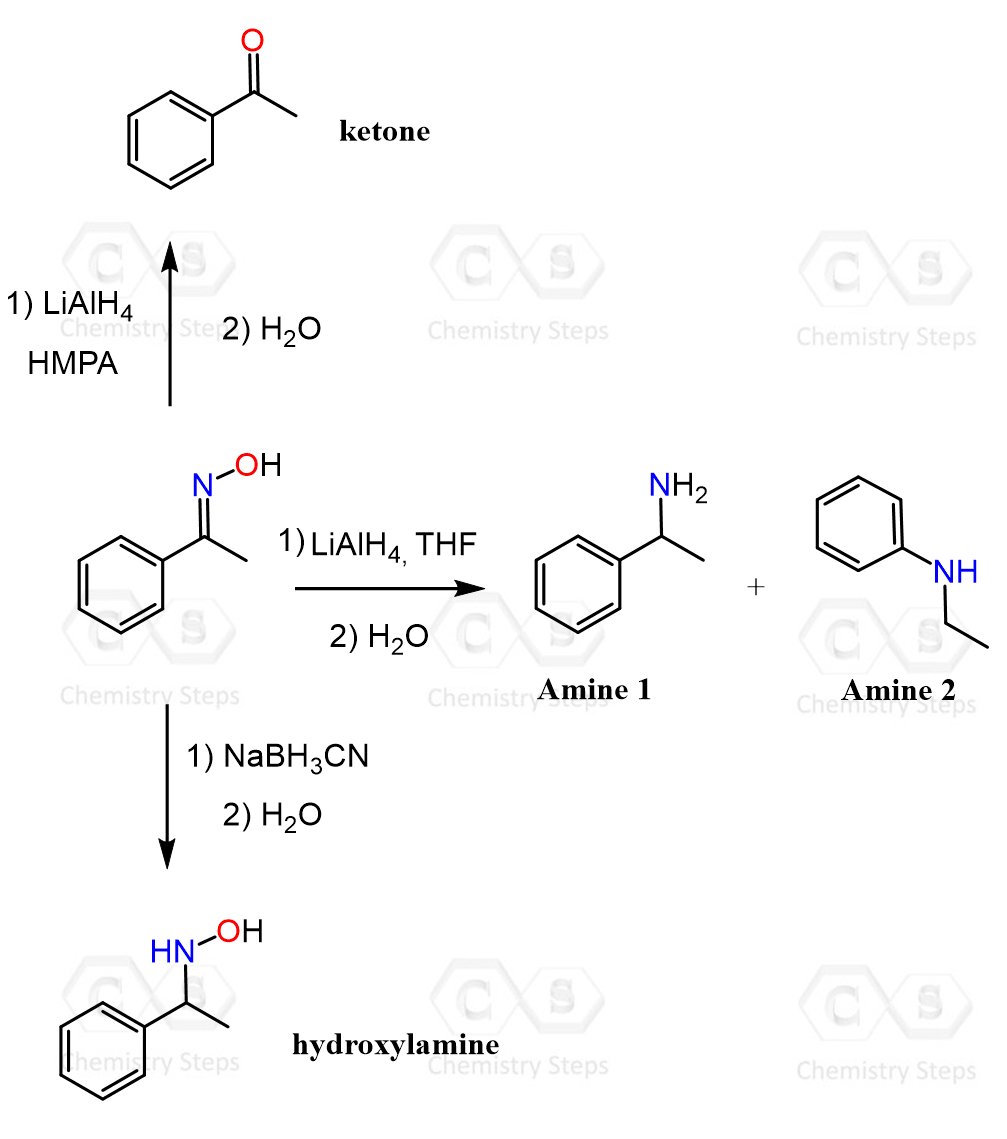
A) Draw plausible curved-arrow mechanisms for the formation of amines 1 and 2.
B) Explain the outcomes when a different solvent is used and draw a mechanism for the formation of the ketone.
C) Explain the formation of the hydroxylamine when NaBH₃CN is used.
This content is for registered users only.
Click here to Register!
By joining Chemistry Steps, you will gain instant access to the answers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps, and the powerful set of Organic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
Click here to Register!
By joining Chemistry Steps, you will gain instant access to the answers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps, and the powerful set of Organic Chemistry 1 and 2 Summary Study Guides.
This content is for registered users only.
Click here to Register!
By joining Chemistry Steps, you will gain instant access to the answers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps, and the powerful set of Organic Chemistry 1 and 2 Summary Study Guides.