Ketones and aldehydes react with halogens at the alpha position when an acid or a base catalyst is used. The halogenation works with Cl2, Br2, and I2:

Let’s start discussing the mechanism of the acid-catalyzed halogenation. The reaction is carried out either in acetic acid or in another solvent with an added acid catalyst. The role of the acid is to promote a tautomerization of the carbonyl to the corresponding enol which is nucleophilic enough to react with the halogen. Overall, the reaction is a substitution of H by a halogen on the ɑ carbon.
Let’s summarize these two parts of the acid-catalyzed halogenation mechanism below:

This halogenated carbon next to the carbonyl (ɑ-position ) is a new reaction site since the halogen is a good leaving group, therefore, substitution and elimination reactions become possible:

For example, butanal can be converted to 2-bromobutanal and further reacted with a good nucleophile such as a sulfide:
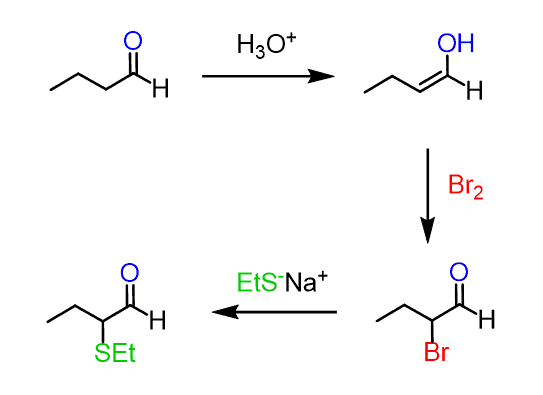
For elimination reactions, pyridine or a carbonate is most often used:

Alpha Halogenation in Base
Under basic conditions, the halogenation occurs via formation of enolates and, despite being similar, the halogenation under basic conditions is not as useful since it is difficult to stop the process at monohalogenation:

What happens is the product, formed after monohalogenation, is more reactive than the starting carbonyl compound and it undergoes another halogenation before an unreacted molecule can start:

The reason for this higher reactivity is the increase of acidity of the alpha proton connected to the carbon with an electronegative halogen atom. As a result, all the protons are substituted by a halogen leading to dihalogenation and a trihalo carbonyl compounds for methyl ketones.
Let’s also look at the mechanism of base-catalyzed halogenation to make this more visual. The reaction starts with the removal of an ɑ proton forming an enolate which can then attack the halogen:

What is interesting is that the trisubstituted ketones are now very reactive towards a nucleophilic acyl substitution and react with the hydroxide converting into a carboxylate salt:
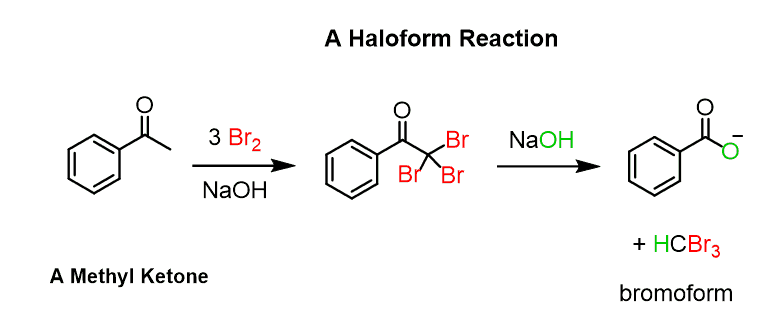
The reaction is possible because the tri halogens stabilize the negative charge on the carbon thus making it a good leaving group.
The reaction of methyl ketones with an excess of the halogen is called the haloform (CHX3) reaction and we will discuss it in a separate post.
Can Monohalogenation be Achieved With acid Catalysis?
The polyhalogenation under basic conditions brings an interesting question – is the monohalogenation possible under acidic conditions, and if yes, why is it so?
And the answer to this question is yes – monohalogenation is observed for the acid-catalyzed halogenation of aldehydes and ketones.
This selectivity indicates that the second halogenation does not occur, or it happens very slowly to a non-significant extent. A possible explanation for this can be the decreased stability of the carbocation intermediate that is formed when the halogenated enol reacts with another molecule of the halogen:
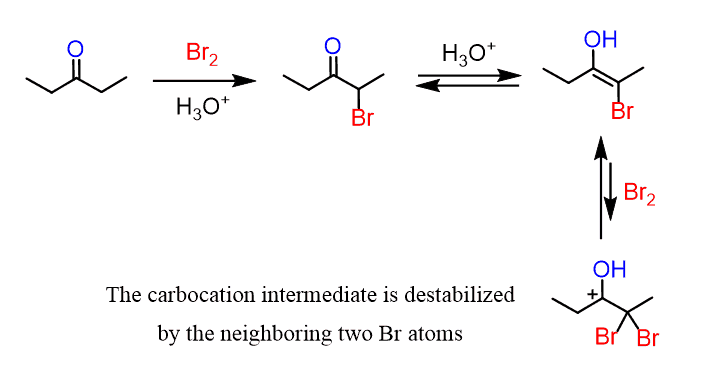
Although the positive charge is resonance-stabilized with the lone pairs of the oxygen, the two electron-withdrawing halogens are destabilizing the carbocation thus slowing down the reaction. Notice that this is different when a base catalyst is used, and the intermediate is a negatively charged enolate which is stabilized by the halogen atoms.
The Regiochemistry of the Alpha Halogenation
Aldehydes have only one ɑ carbon and therefore, there is no regiochemistry to be considered. Ketones, on the other hand, have two ɑ carbons and if it is an unsymmetrical ketone, the halogenation occurs primarily at the more substituted ɑ carbon:
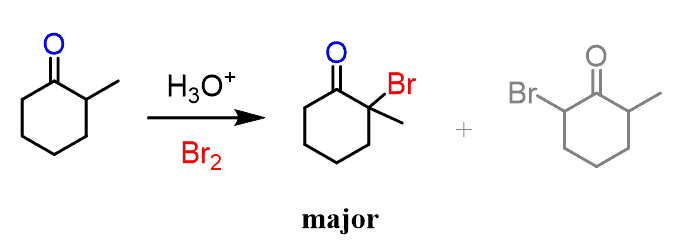
This regioselectivity can be explained by the higher stability of the more substituted enol just like we knew for the alkenes.
You can find additional practice problems on alpha halogenation here:
Alpha Halogenation of Enols and Enolates Practice Problems
Check Also
- Alpha Halogenation of Enols and Enolates
- The Haloform and Iodoform Reactions
- Alpha Halogenation of Carboxylic Acids
- Alpha Halogenation of Enols and Enolates Practice Problems
- Aldol Reaction – Principles and Mechanism
- Aldol Condensation – Dehydration of Aldol Addition Product
- Intramolecular Aldol Reactions
- Aldol Addition and Condensation Reactions – Practice Problems
- Crossed Aldol And Directed Aldol Reactions
- Crossed Aldol Condensation Practice Problems
- Alkylation of Enolates Alpha Position
- Enolate Alkylation Practice Problems
- Acetoacetic Ester Synthesis
- Acetoacetic Ester Enolates Practice Problems
- Malonic Ester Synthesis
- Michael Reaction: The Conjugate Addition of Enolates
- Robinson Annulation, Shortcut, and Retrosynthesis
- Claisen Condensation
- Dieckmann condensation – An Intramolecular Claisen Reaction
- Crossed Claisen and Claisen Variation Reactions
- Claisen Condensation Practice Problems
- Stork Enamine Synthesis
- Enolates in Organic Synthesis – a Comprehensive Practice Problem
