We have seen that the product of the aldol reaction is a β-hydroxy carbonyl compound:
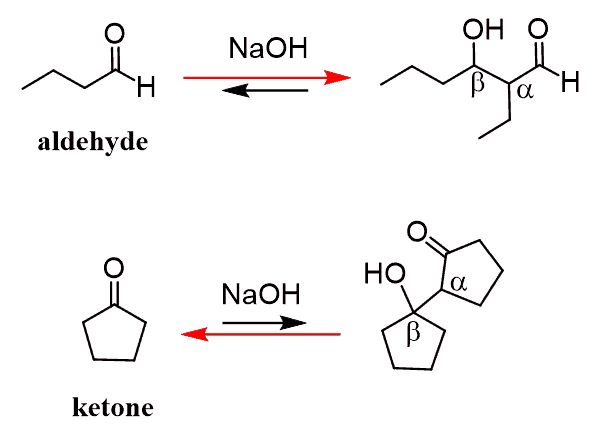
One can look at it as an alcohol, and what is interesting about this alcohol is that it undergoes an elimination when heated in presence of a base or an acid:
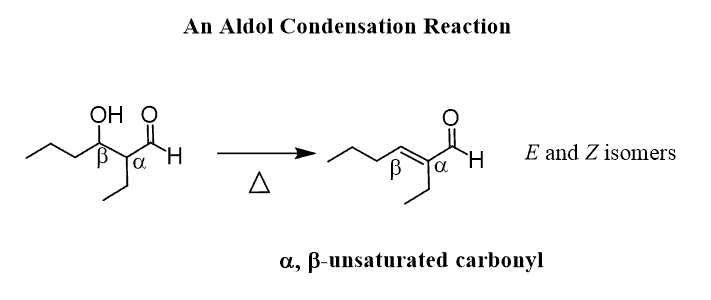
This two-step process is the aldol condensation and the final product of it is an ɑ, β-unsaturated carbonyl compound.
The E1CB Mechanism in Aldol Condensation
Let’s sum this one more time – the aldol addition reaction is the reaction between the enolate and the carbonyl producing an β-hydroxy carbonyl compound. This compound is then converted into an ɑ, β-unsaturated carbonyl compound via an elimination reaction. The two steps together are referred to as the aldol condensation reaction.
Now, what is the mechanism of this elimination? We don’t normally expect an alcohol to undergo an elimination reaction since the OH is a poor leaving group. It only happens through and E1 by using a strong acid or by converting the OH into a good leaving group such as a mesylate, tosylate or a simple halide.
So, the fact that elimination works with a moderate base such as NaOH, indicates two things; 1) that the OH is, for some reason, kicked out and 2) the hydrogen between the OH and C=O group is removed as judged from the position of the double bond in the product.
Let’s address these by starting from the hydrogen. The only difference in β-hydroxy carbonyl compounds compared to regular alcohols is the presence of the carbonyl group and makes the hydrogen more acidic:
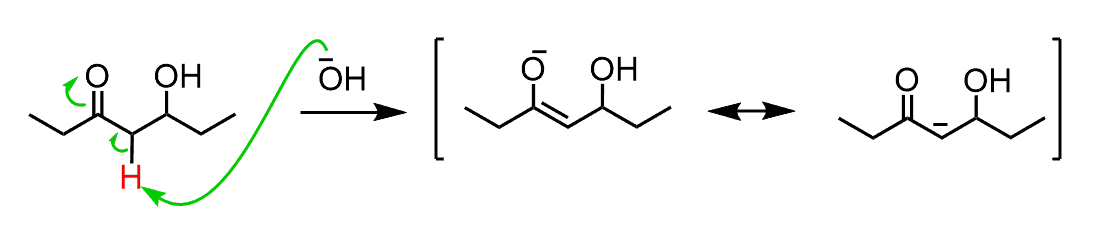
However, carbonyl compounds are not deprotonated by hydroxides to a significant degree either while these eliminations have quite high yields. And this has to do with the OH group, which after being expelled by the π electrons forms am energetically favored conjugated system:

Once the ɑ, β-unsaturated carbonyl compound is formed, it shifts the equilibrium of the whole process to the right as it brings in an irreversible step.
Unlike the aldol addition reaction, the aldol condensation does not generally occur at room temperature. Just like other elimination reactions, it is entropically favored at higher temperature (~100 oC)
Some exceptions are when the final product is a conjugates system of multiple π bonds since this gives additional stability and serves as a driving force for the reaction.
For example, acetophenone undergoes aldol condensation so readily that the intermediate β-hydroxy carbonyl compound is not isolated as it immediately transforms into a highly conjugated system:
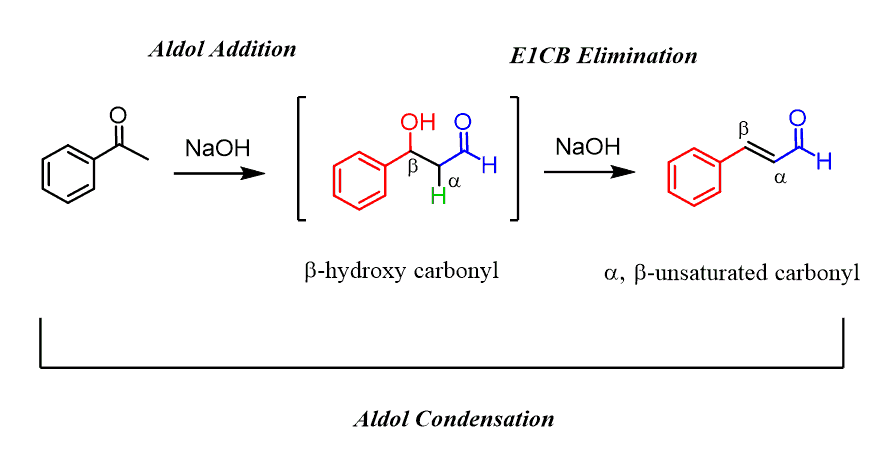
The aldol condensation is also efficient for ketones which at lower temperature establish an unfavorable equilibrium of an aldol addition reaction:

This, again, is explained by the formation of a conjugated system of π electrons which are not present in the aldol addition product.
The Shortcut of Aldol Condensation Reactions
Remember, we talked about the strategy for quickly determining the product of aldol addition reactions. In short, you needed to draw the “V” shape of the β-hydroxy carbonyl unit and add the remaining alkyl groups based on the aldehyde or the ketone being reacted:
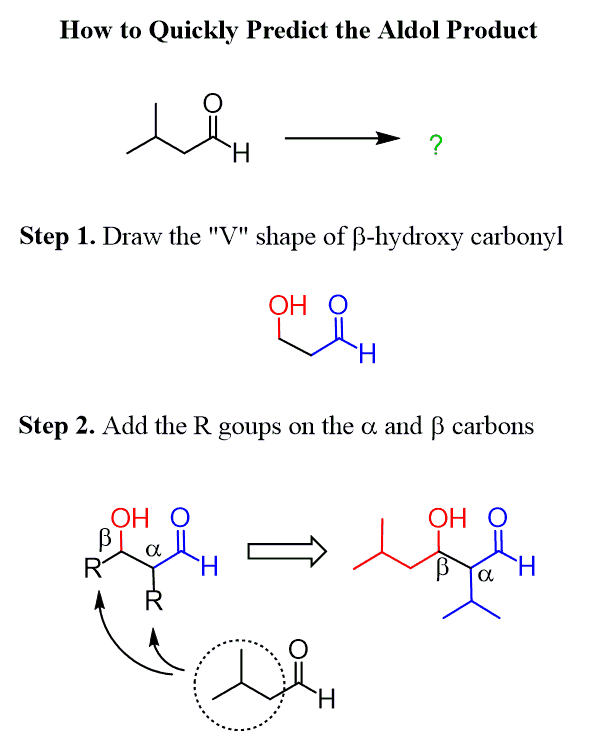
Now, to determine the product of an aldol condensation, you can follow the steps shown above, and in the end, remove the OH by adding a C=C double bond conjugated to the carbonyl.
Alternatively, align the two reacting carbonyls such that the ɑ carbon of the nucleophile is pointing to the C=O group of the electrophile and connected them with a double bond by removing the C=O oxygen:

If this made sense so far, try solving a good set of aldol condensation problems here:
https://staging.chemistrysteps.com/aldol-reaction-practice-problems/
Check Also
- Alpha Halogenation of Enols and Enolates
- The Haloform and Iodoform Reactions
- Alpha Halogenation of Carboxylic Acids
- Alpha Halogenation of Enols and Enolates Practice Problems
- Aldol Reaction – Principles and Mechanism
- Aldol Condensation – Dehydration of Aldol Addition Product
- Intramolecular Aldol Reactions
- Aldol Addition and Condensation Reactions – Practice Problems
- Crossed Aldol And Directed Aldol Reactions
- Crossed Aldol Condensation Practice Problems
- Alkylation of Enolates Alpha Position
- Enolate Alkylation Practice Problems
- Acetoacetic Ester Synthesis
- Acetoacetic Ester Enolates Practice Problems
- Malonic Ester Synthesis
- Michael Reaction: The Conjugate Addition of Enolates
- Robinson Annulation, Shortcut, and Retrosynthesis
- Claisen Condensation
- Dieckmann condensation – An Intramolecular Claisen Reaction
- Crossed Claisen and Claisen Variation Reactions
- Claisen Condensation Practice Problems
- Stork Enamine Synthesis
- Enolates in Organic Synthesis – a Comprehensive Practice Problem

Dear Dr Sargsyan,
I have a question related to the dehydration of an aldol produced through the crossed condensation of ethanal and propanal. Let s assume that ethanal is the ”carbonyl component”, while propanal is the methylene component”. The product of the aldol condensation is 3-hydroxy-2-methylbutanal (taking into account the previous assumption). The dehydration the removal of the acidic H from C2 placing a C=C bond between C2 and C3. Nevertheless, I was wondering what are the chance for an elimination from C4 to take place (so that the C=C bond would appear between C3 and C4).
With very best regards,
Bogdan
Hi Bogdan, that’s a very good question.
The key point is that the dehydration step of an aldol condensation usually proceeds through an E1cb mechanism. In this mechanism, the base first removes the acidic α-hydrogen next to the carbonyl group, forming an enolate. This intermediate is stabilized by resonance with the carbonyl, which makes its formation favorable. After that, the -OH group leaves from the β-carbon, producing the α,β-unsaturated aldehyde.
In your example (3-hydroxy-2-methylbutanal), the base removes the hydrogen at C2, forming the enolate, and elimination then gives the C2–C3 double bond, which is conjugated with the carbonyl group.
Even if the alkene were to form by another mechanism, the C3-C4 alkene would still be less stable, because it would not be conjugated with the carbonyl and it would also be less substituted than the C2-C3 alkene.
If a hydrogen were removed from C4, the resulting carbanion would not be an enolate and would not be resonance-stabilized by the carbonyl. Because the E1cb mechanism relies on forming this stabilized enolate intermediate, that pathway is much higher in energy and essentially does not compete.
Thank you so much for your reply! Therefore, when dealing with water elimination from an aldol the only possible way is dehydration involving a H atom from C alpha of the ”methylene component”. I completely forgot the aspect of alkenes stability (more substituted, more stabile…).
I have graduated Biochemistry and I have worked for 20 years in the Romanian higher education sector teaching Biochemistry and related topics. Nevertheless, I have had the opportunity to deliver for seminars in Organic chemistry (carboxylic acids and their derivatives and some mixed functions classes) as well as practical demonstrations in Organic chemistry (about 10 years). In Romania Organic chemistry is delivered through the presentation of classes and not based more on mechanisms. Since 1,5 years ago I have been delivering Chemistry private classes (inorganic, organic and general chemistry) for high school students unable to keep the pace in class and for those who want to enter medical schools. Basically, I am a freelancer.
Delivering the classes in Organic chemistry has not been very difficult. I mean, I am familiar with many aspects from many topics (reactions, reactivity, problem solving strategies). Nevertheless, I feel the need to enhance my knowledge on reaction mechanisms. I graduated last year an online course in Organic chemistry provided by UC San Diego. Very nice, it helped a lot but the pace was too rapid and I feel that I would have needed some extra time.
I was wondering if you provide online private classes. I would need some highly dense classes on the major mechanisms, including stereochemistry. With crystal clear and straightforward explanations and lots of exercises.
With very best regards,
Bogdan
Hello Bogdan,
I do provide online tutoring for Organic 1 and 2 classes; however, you clearly do not fit in the category of a typical student taking an organic chemistry class, as you already have so much experience. Feel free to send me an email from the contact page with more details of what topics and reactions you need to go over, so we can see how beneficial the lessons may be for you.
Dear Dr Gevorg,
I wrote you an email at “gevorg@chemistrysteps.com”. I hope you got it.
With very best regards,
Bogdan