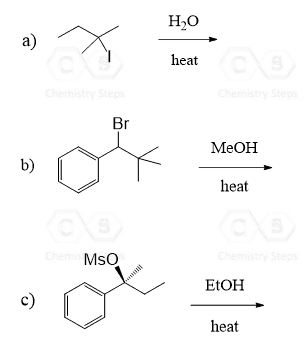
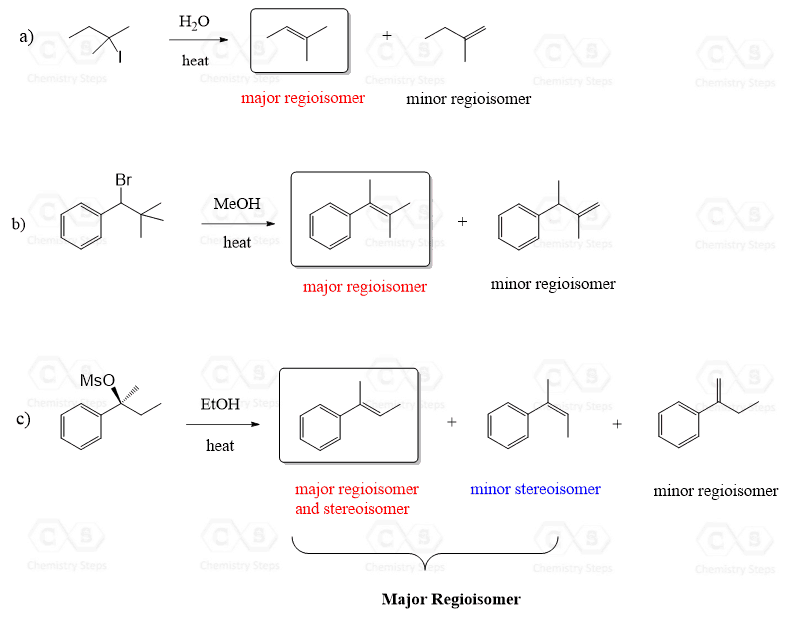
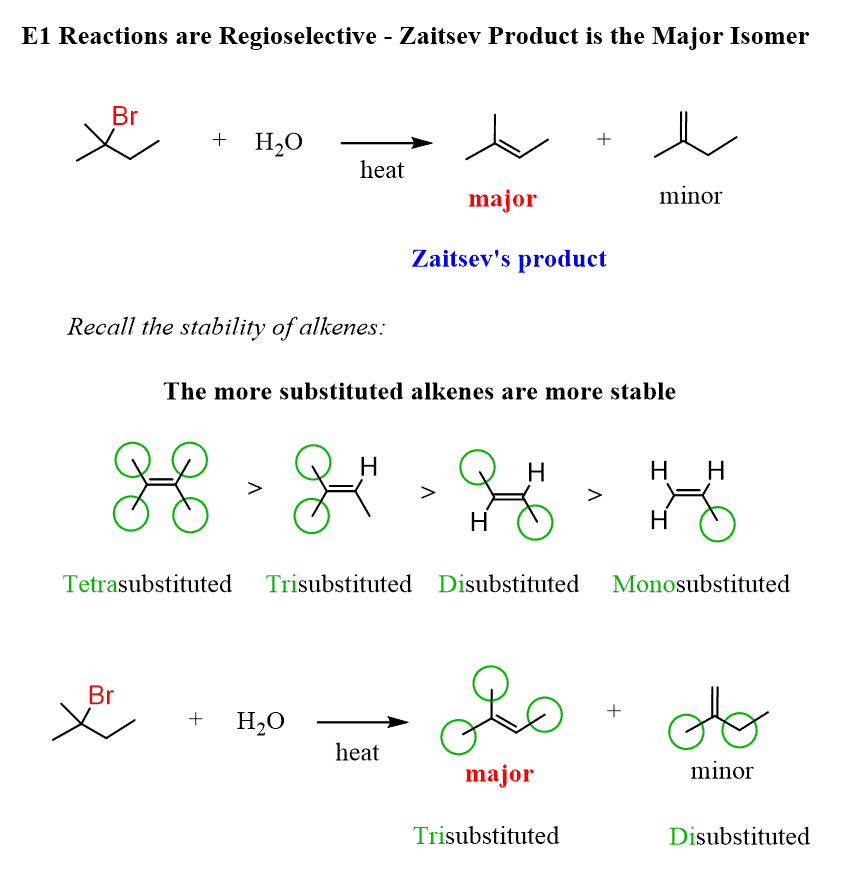
E1 reactions are regioselective and, just like in the E2 mechanism, the Zaitsev product predominates due to the higher stability of the more substituted alkene:
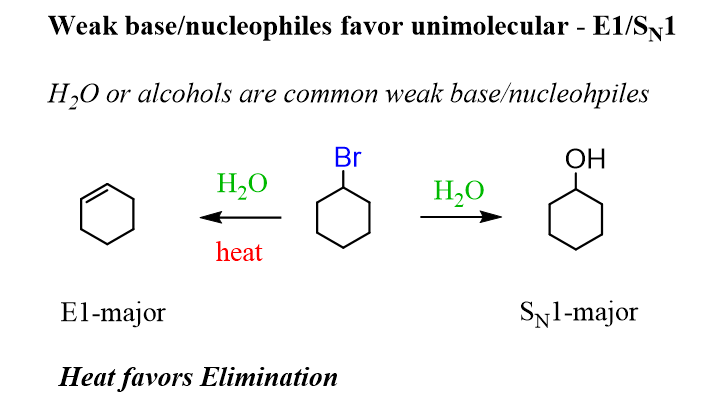
Notice that in the reaction above, water was used as a weak base, and the E1 elimination was favored by heating the reaction. If heat is not applied, a weak base/nucleophile favors the SN1 reaction:
On the other hand, using a strong base favors the E2 mechanism. One advantage of the E2 mechanism is that the regiochemical outcome of the reaction can be controlled by choosing a strong, unhindered, or a sterically hindered base:

Contrary to this, the E1 mechanism favors Zaitsev’s product, and we cannot change this regioselectivity even if a sterically hindered alcohol is used as a weak base.
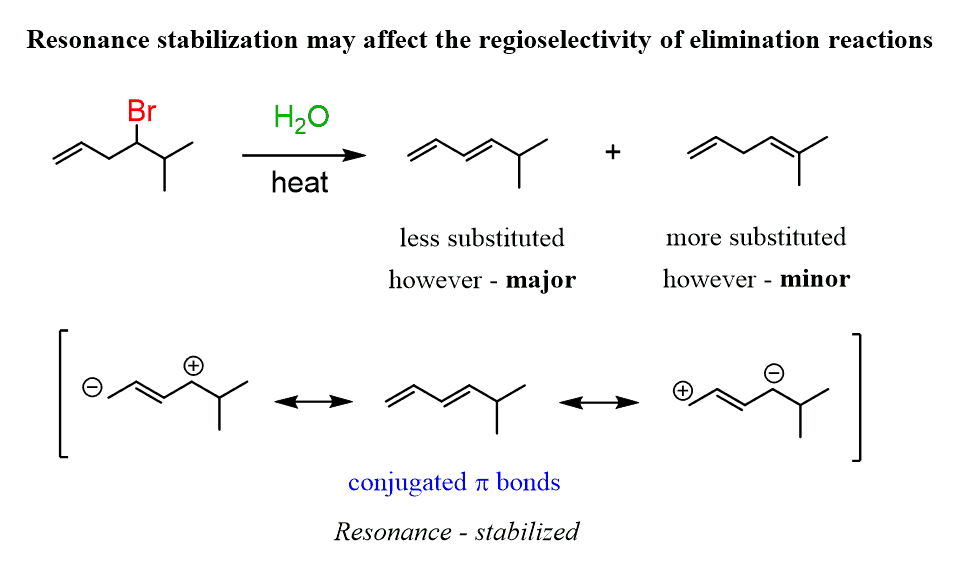
One possibility of forming the Hoffman product in an E1 and E2 reaction is the resonance-stabilization of the less-substituted alkene:
Rearrangements in E1 Reactions
Even though Zaitsev’s product is the major regioisomer in the E1 reaction, you need to always keep in mind the possibility of rearrangements for any unimolecular mechanism going through a carbocation intermediate:
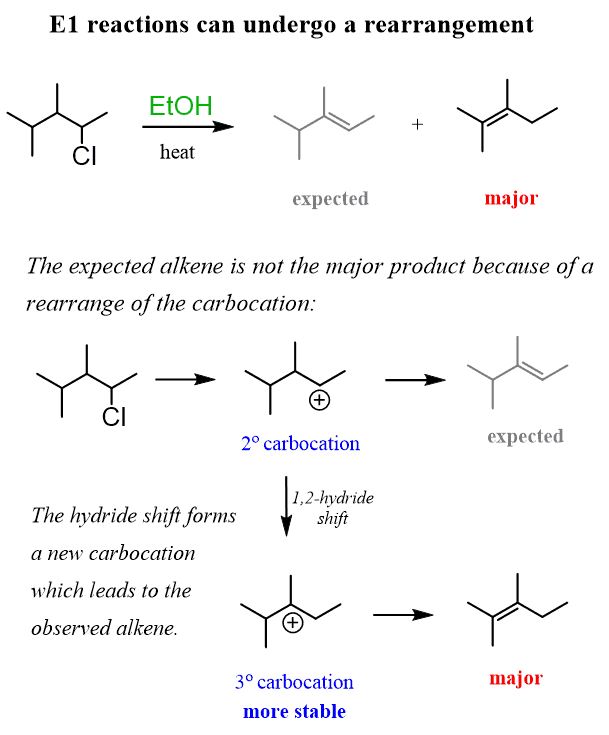
The carbocation formed in the first step undergoes a 1,2-hydride shift to form a more stable tertiary carbocation, which is then attacked by the base.
In general, remember, E2 and SN2 reactions are more useful because of better control on the regio- and stereochemistry of the reaction since they do not form carbocations, which may undergo rearrangements.