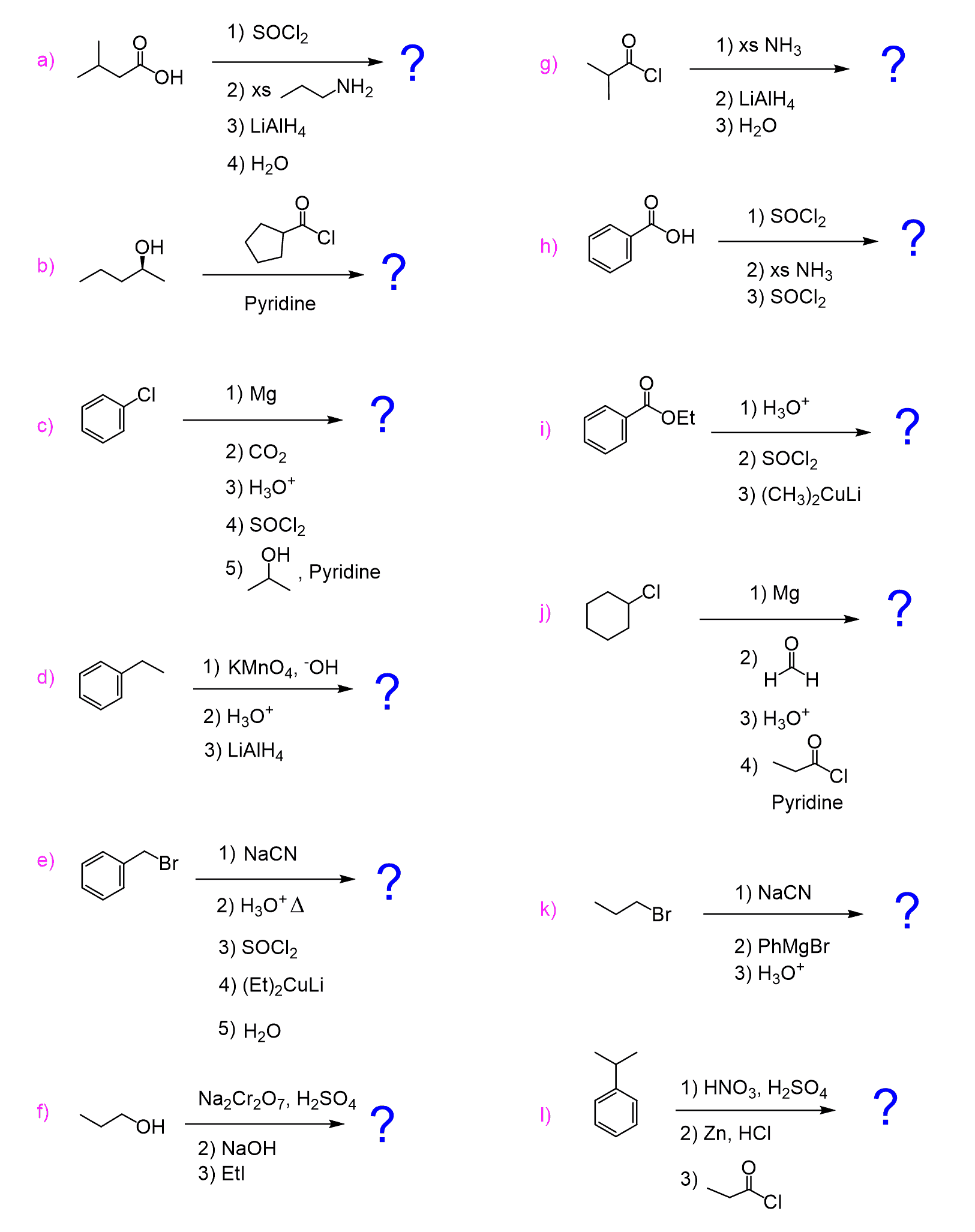
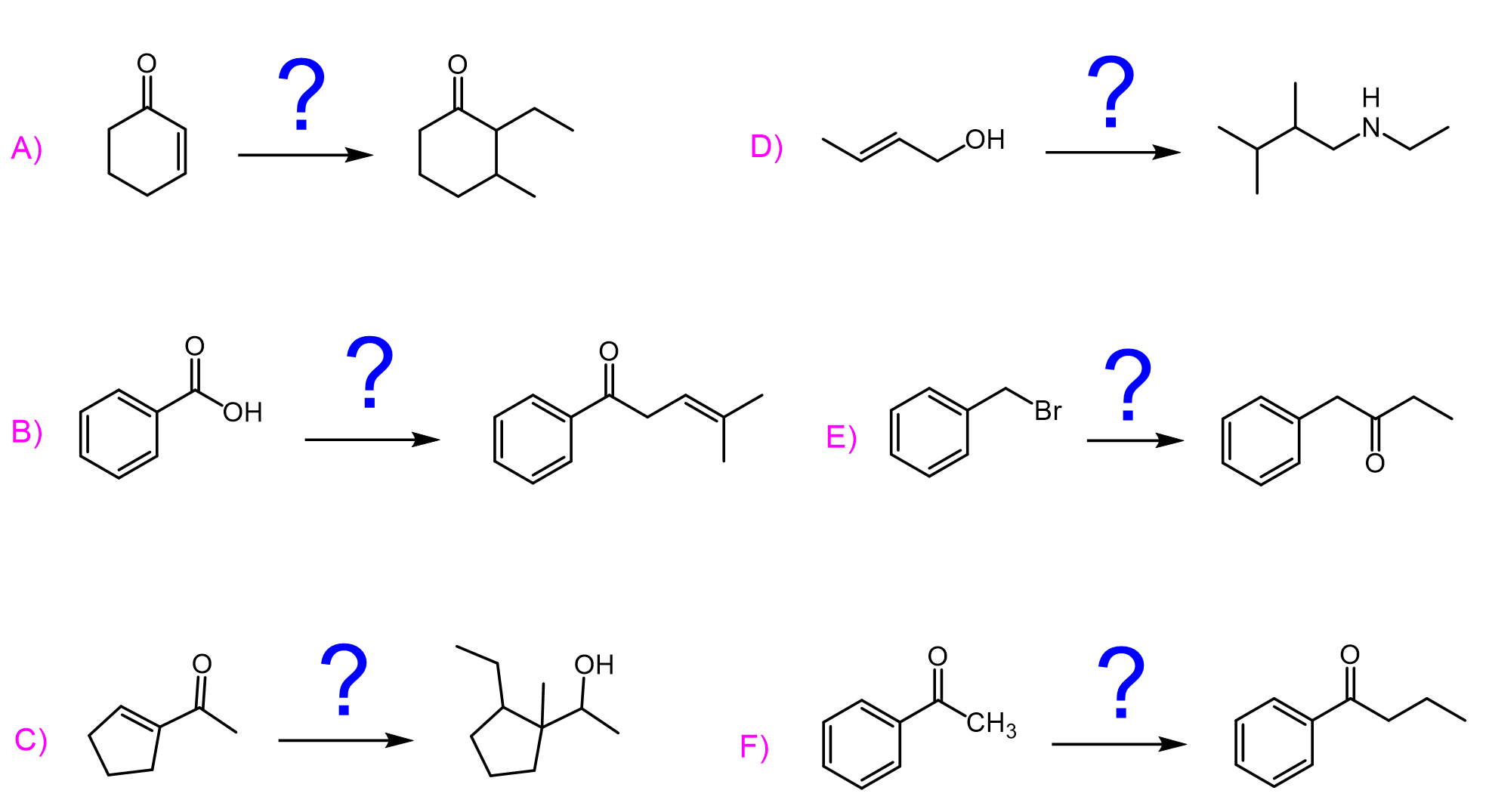
Acyl chlorides, being the most reactive among carboxylic acid derivatives, undergo a wide variety of nucleophilic acyl substitution reactions.
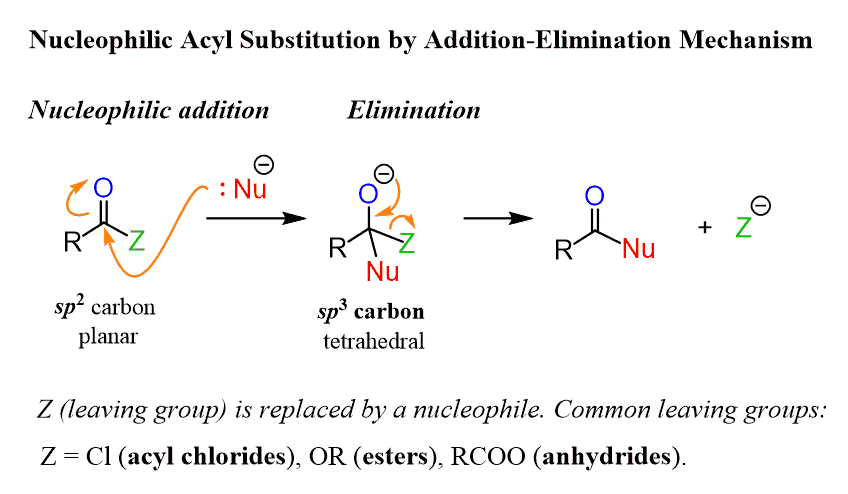
A quick reminder, in a nucleophilic acyl substitution, the leaving group is replaced by a nucleophile by the addition-elimination mechanism:
Let’s first mention that acid chlorides need to be kept in dry conditions since they react readily with water, forming carboxylic acids. The reaction is even faster with hydroxides since those are stronger nucleophiles:
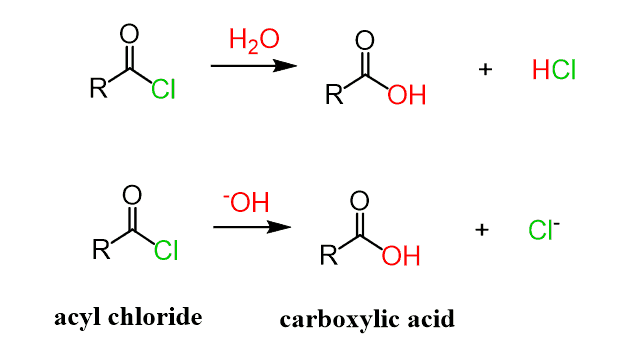
The reaction involves a nucleophilic addition of water, followed by a proton transfer, forming a geminal diol. Geminal diols are not stable, and one of the oxygens kicks out the Cl to eventually produce a carboxylic acid:
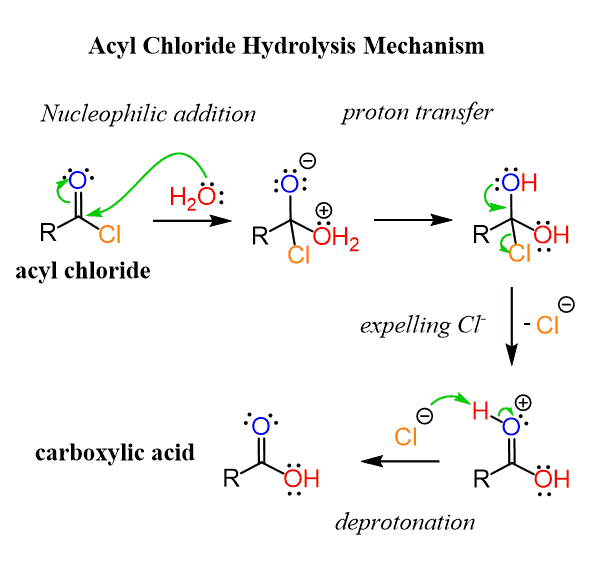
The reaction with hydroxide ion has fewer steps and goes faster as it is a better nucleophile:

Reaction of Acid Chlorides with Alcohols
Acid chlorides can easily be converted into esters by reacting with alcohols. A base such as pyridine or triethylamine (TEA) is usually added to speed up the process by deprotonating the positively charged intermediate:
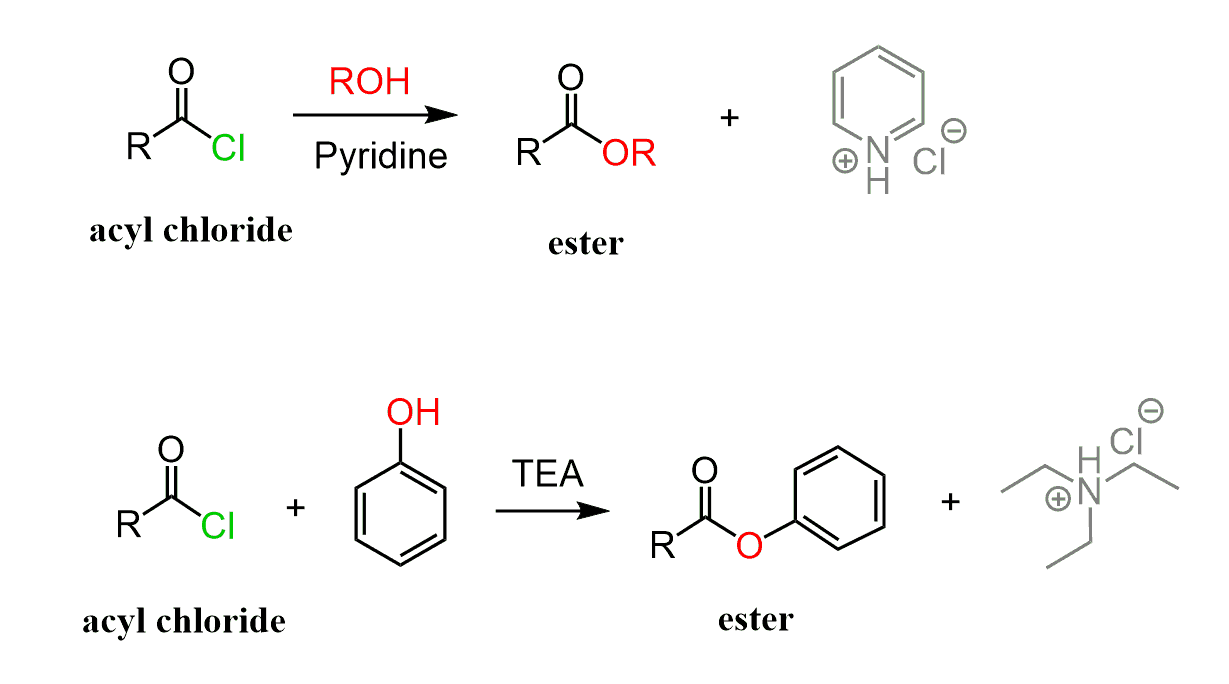
The mechanism is similar to the hydrolysis of acid chlorides, except the nucleophile here is the alcohol:

Reaction of Acid Chlorides with Thiols
Just like the alcohols, thiols also react with acid chlorides, producing thioesters:

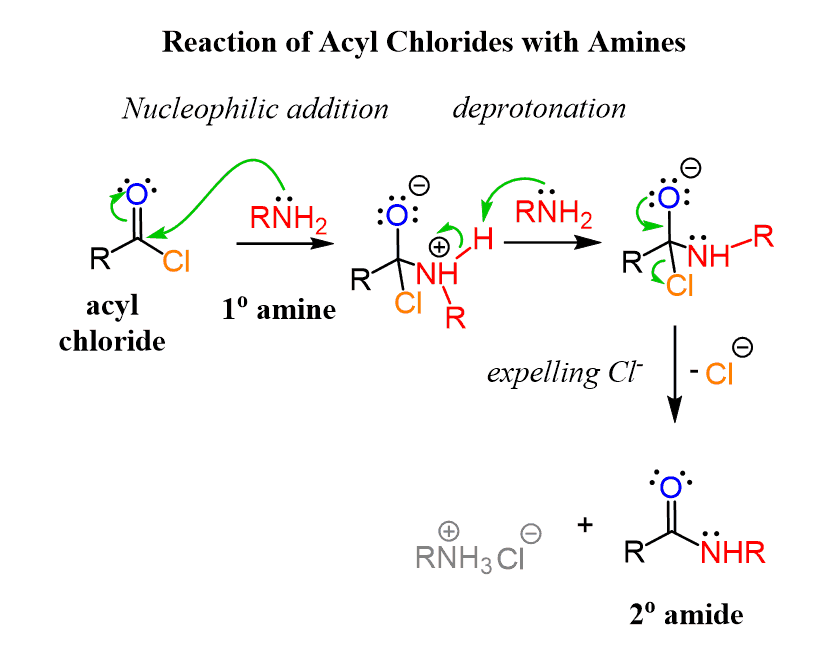
Reactions of Acid Chlorides with Ammonia and Amines
Ammonia reacts rapidly with acyl chlorides, producing primary amide,s while primary and secondary amines can be used for converting acyl chlorides into secondary and tertiary amides, respectively:
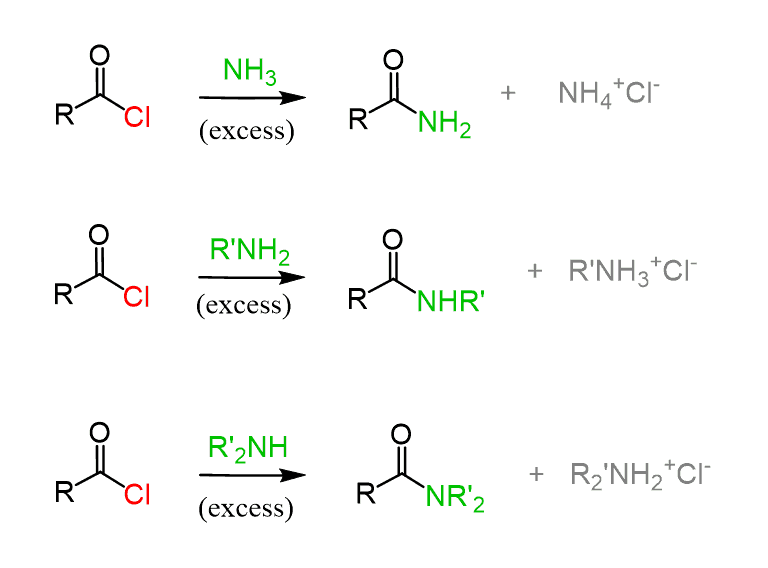
These reactions follow a similar pattern to what we saw above. The amine is used in excess for deprotonating positively charged intermediates:
Reaction of Acid Chlorides with Carboxylate Salts
Carboxylate ion is a good nucleophile and reacts readily with acid chloride, forming carboxylic acid anhydrides:
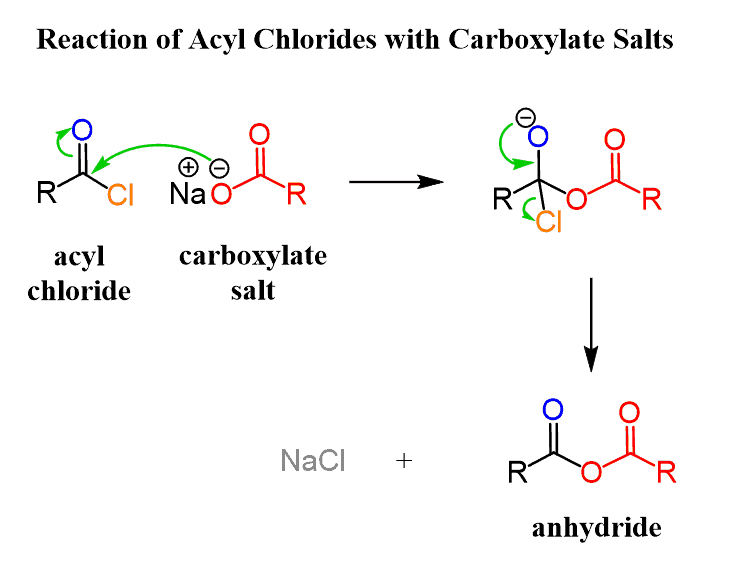
One advantage of preparing anhydrides by this method is that it allows for the synthesis of symmetrical and mixed anhydrides.
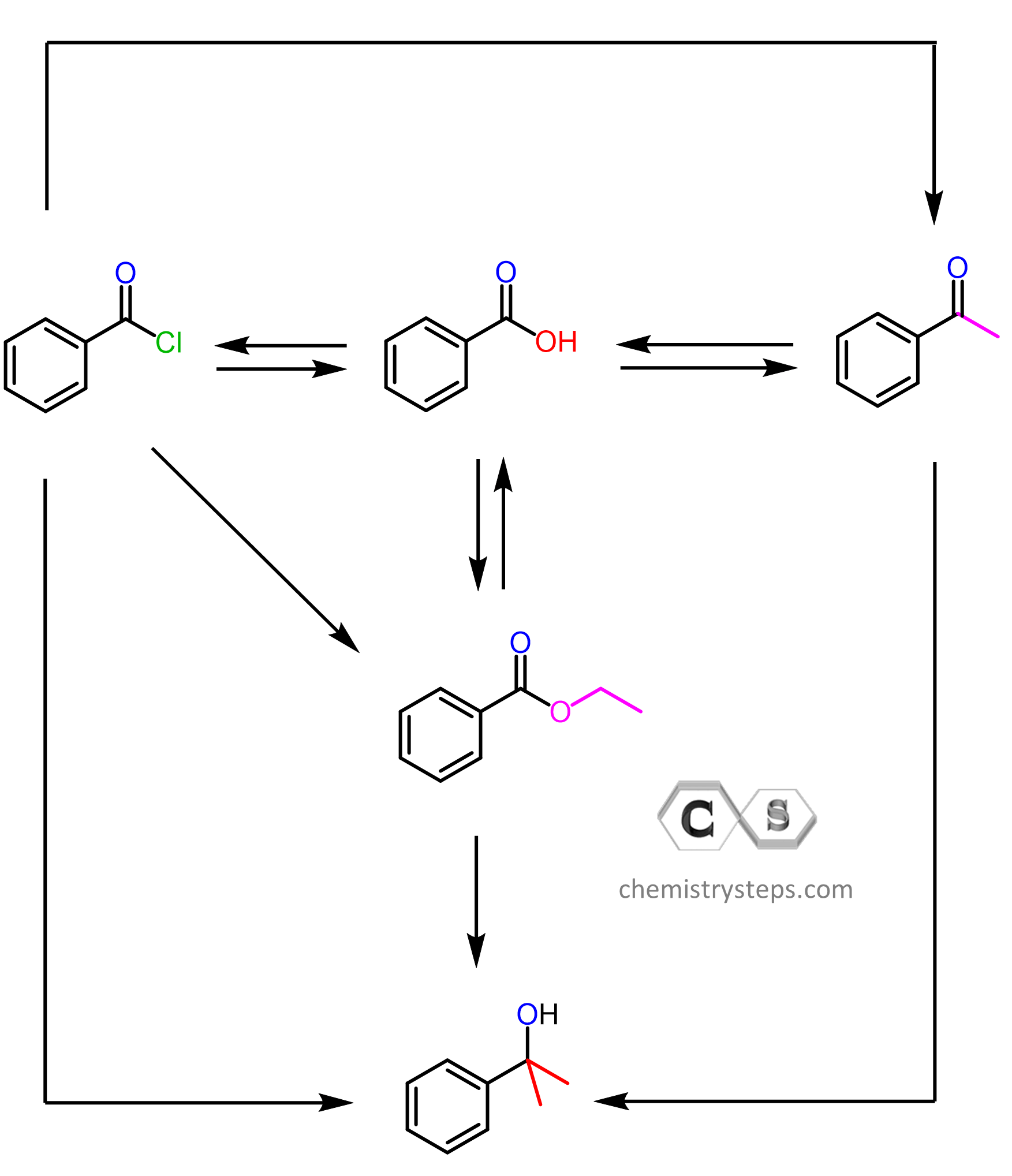
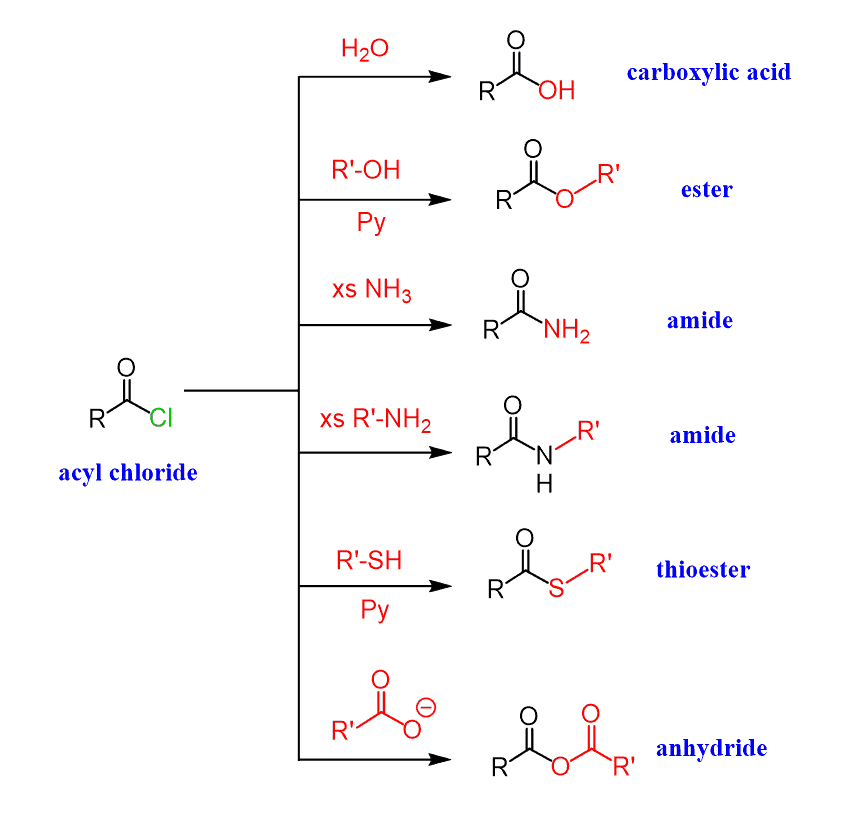
Let’s summarize what we have learned about acyl chlorides so far. Keep in mind that they are the most reactive carboxylic acid derivatives and therefore can be used to ease the path of converting a carboxylic acid into an ester, amide, or anhydride. All of these can be achieved in mild conditions, while the direct conversion from carboxylic acids requires more forcing conditions:
Reduction and Organometallic Reactions of Acid Chlorides
As expected, acyl chlorides also react with stronger nucleophiles such as the hydride (H–) and alkyl ions (R–). They can be reduced to alcohols with LiAlH4, NaBH4, or DIBAL and converted to ketones and alcohols by the Gilman and Grignard reagents, respectively.
These reactions and their mechanisms are covered in the following two posts:
Reduction of Acyl Chlorides with LiAlH4, NaBH4, or DIBAL
Reaction of Acyl Chlorides with Grignard and Gilman’s Reagents