Introduction to the Hybridization
Let’s start by answering this question: Why do we need the hybridization theory?
Here is one answer to this. It is confirmed experimentally that the carbon atom in methane (CH4) and other alkanes has a tetrahedral geometry. A reminder that in tetrahedral geometry, all the angles are 109.5o and the bonds have identical lengths. Remember also that covalent bonds form as a result of orbital overlapping and the sharing of two electrons between the atoms. So, in order to predict the valency and geometry of the carbon atom, we are going to look at its electron configuration and the orbitals.
C – 1s22s22p2
The valence electrons are the ones in the 2s and 2p orbitals, and these are the ones that participate in bonding and chemical reactions.

You can see from the electron configuration that it is impossible to make four identical in bond length, energy, and everything else (degenerate) bonds because one of the orbitals is s, which is spherical, and the other three are p orbitals. And this is where we get into the need for a theory that can help us explain the known geometry and valency of the carbon atom in many organic molecules. So,
Hybridization is a theory that is used to explain certain molecular geometries that would not have been possible otherwise.
The sp3 hybridization
Now, let’s see how that happens by looking at methane as an example. In the first step, one electron jumps from the 2s to the 2p orbital. This leads to the excited state of the carbon:
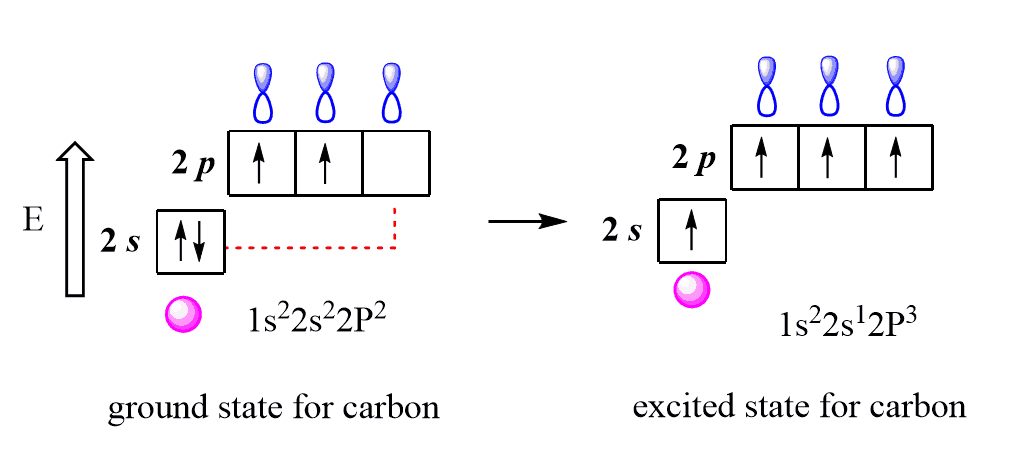
Pay attention that the electron goes uphill as the p subshell is higher in energy than the s subshell, and this is not energetically favorable, but we will see how it is compensated in the next step when orbitals are mixed (hybridized).
So, in the next step, the s and p orbitals of the excited state carbon are hybridized to form four identical in size, shape, and energy orbitals.
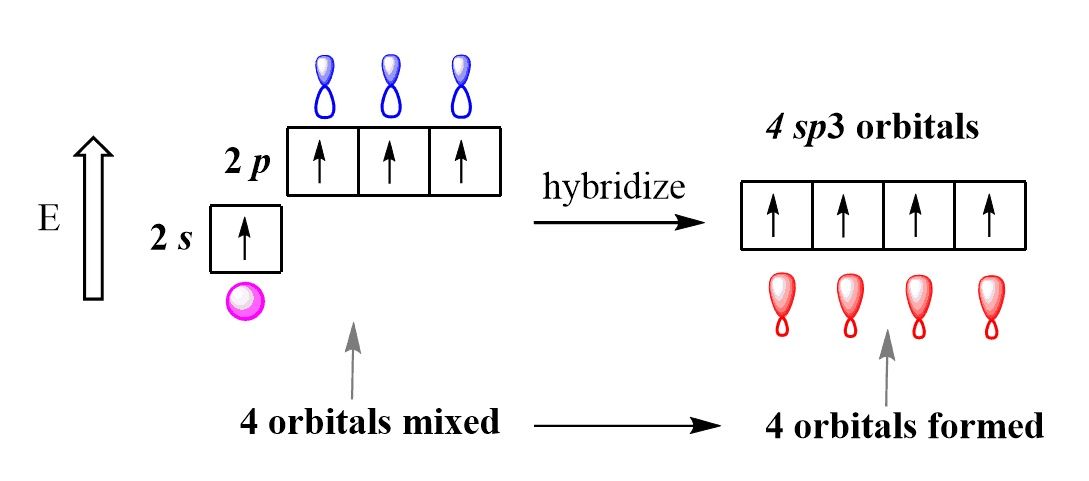
The number of the hybrid orbitals is always the same as the number of orbitals that are mixed. So, four orbitals (one 2s + three 2p) are mixed, and the result is four sp3 orbitals. These are hybrid orbitals and look somewhat like the s and p orbitals. And again, we call them sp3 because they are formed from one s orbital and three p orbitals.
The formation of these degenerate hybrid orbitals compensates for the energy uphill of the s-p transition, as they have lower energy than the p orbitals.
The four sp3-hybridized orbitals arrange in a tetrahedral geometry and make bonds by overlapping with the s orbitals of four hydrogens: This explains the symmetrical geometry of methane (CH4), where all the bonds have the same length and bond angle.
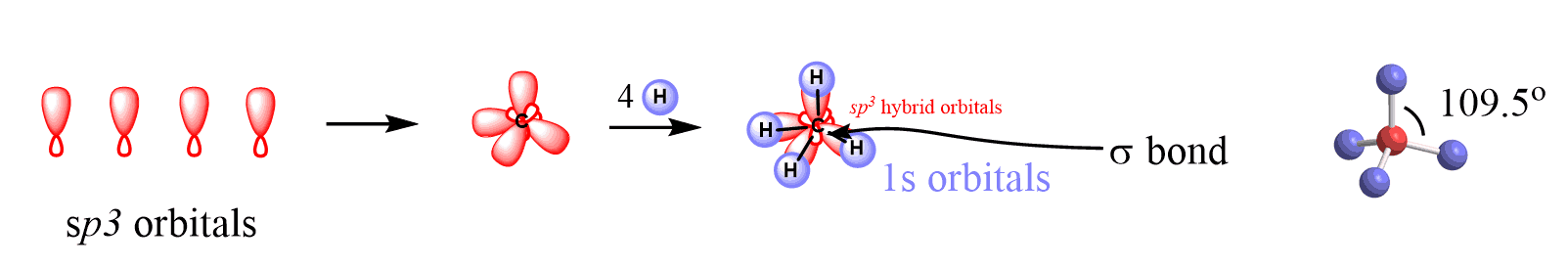
All four C – H bonds in methane are single bonds that are formed by head-on (or end-on) overlapping of the sp3 orbitals of the carbon and s orbital of each hydrogen.
The bonds that form by the head-on overlap of orbitals are called σ (sigma) bonds because the electron density is concentrated on the axis connecting the C and H atoms.
Ethane – CH3-CH3 and other alkanes
If instead of one hydrogen, we connect another sp3-hybridized carbon, we will get ethane:

And consequently, in all the alkanes, there is a sigma bond between the carbon atoms and the carbon-hydrogen atoms, and the carbons are sp3 hybridized with tetrahedral geometry:

To generalize this, any atom with four groups (either an atom or a lone pair) is sp3 hybridized. And the way to look at this is, in order for the four groups to be as far away from each other as possible, like we learned in the VSEPR theory, the groups need to be in identical four orbitals, which is only possible in the sp3 hybridization.
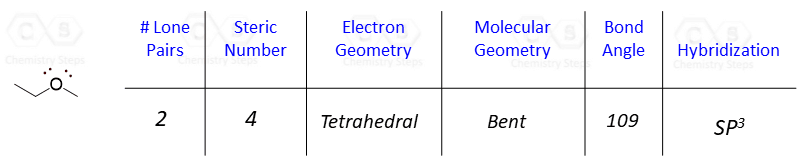
For example, what is the hybridization of the oxygen in the following molecule?
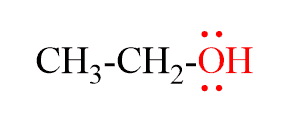
The oxygen is connected to two atoms and has two lone pairs. In total, four groups, and that is why it is sp3 hybridized.
The sp2 hybridization
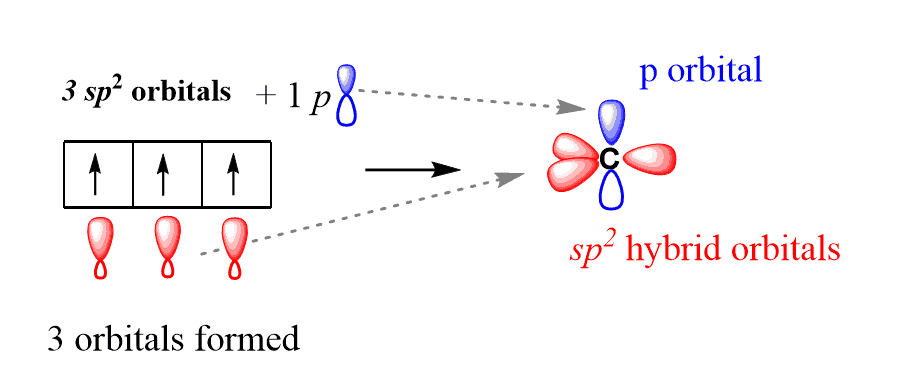
When the excited state carbon atom is formed, the sp3 hybridization is not the only option for mixing the orbitals. The sp2 hybridization occurs when the s orbital is mixed with only two p orbitals, as opposed to the three p orbitals in the sp3 hybridization. So, three orbitals are mixed, and the outcome is three hybrid orbitals, which are called sp2 hybrid orbitals.

The resulting 3 sp2 orbitals are then arranged in a trigonal planar geometry (120o). An important difference here, compared to the sp3 hybridization, is the left-out (unhybridized) p orbital that did not participate in the hybridization. This orbital is placed at 90o to the plane of the trigonal planar arrangement of the three sp2 orbitals:
Carbon hybridization in Ethylene—C2H4
Two sp2 hybridized carbon atoms can make a sigma bond by overlapping one of the three sp2 orbitals and bond with two hydrogens each and two hydrogens make sigma bonds with each carbon by overlapping their s orbitals with the other two sp2 orbitals.

This makes three bonds for each carbon and one p orbital left. Remember, the standard valency of carbon is four, and it likes to have four bonds.
This fourth bond is formed by the side-by-side overlap of the two 2p orbitals on each carbon. This type of bonding by a side-by-side overlap of the orbitals is called a π bond.
So, the two carbons in ethylene, which is the first member of the alkene family, are double-bonded. In each double bond, there is one sigma and one π bond.
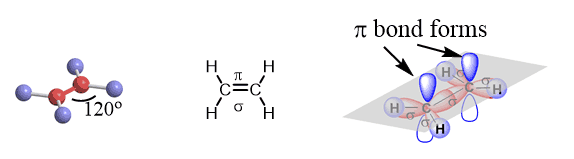
Here are some key parameters about the sp2 hybridization and double bonds that you need to know:
* All the atoms on the double bond are in one plane.
* The angle between atoms is 120o.
* The angle between the plane and p orbitals is 90o.
The sp hybridization
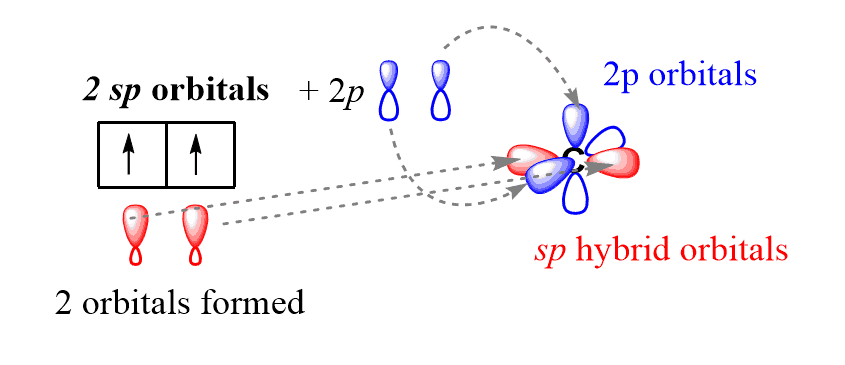
In sp hybridization, the s orbital of the excited state carbon is mixed with only one out of the three 2p orbitals. It is called sp hybridization because two orbitals (one s and one p) are mixed:

The resulting two sp hybrid orbitals are then arranged in a linear geometry (180o) and the two unhybridized 2p orbitals are placed at 90o:
Let’s see how this happens in acetylene – C2H2. The two carbon atoms make a sigma bond by overlapping the sp orbitals.
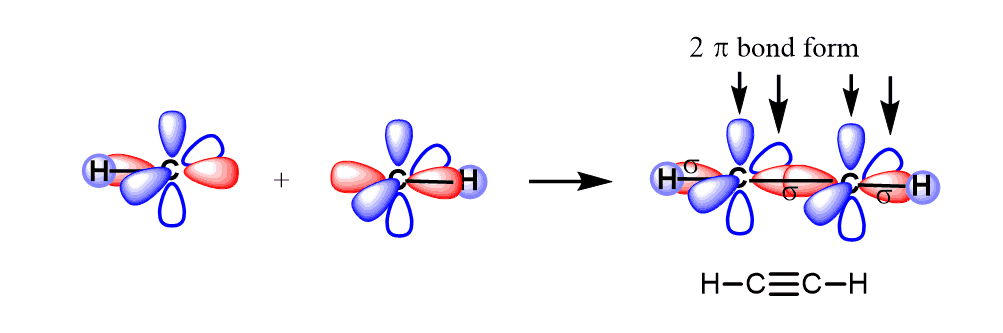
One hydrogen bonds to each carbon atom by overlapping its s orbital with the other sp orbital.
The two p orbitals of each carbon overlap to make two π bonds.
The key parameters of the sp hybridization and triple bond:
* All the atoms have linear geometry.
* The angle between atoms is 180o.
* In a triple bond, there is one σ (sigma) and two π (pi) bonds.
Hybridization of other elements
As a final note, everything we have discussed above is not pertinent to carbon only. The hybridization theory works with the same principle for all the other important elements in organic chemistry, such as oxygen, nitrogen, halogens, and many others.
In the next post, we will discuss how to quickly determine the hybridization of any atom in an organic molecule.
![]()















I think the link for the answers is broken.
John,
Is it for all of them or there is a specific answer you were looking at?
Hi,
Why is the bond angle for number 8 120 when the molecular geometry is linear?
Thanks!
Hi there,
Good question. Perhaps the terminology bong angle is not the most appropriate when there is only one atom connected, as it must be 0 by necessity. The accurate description here would be interorbital angle implying the angle between the bonding orbital(s) and the ones accommodating the lone pairs. In other words, the angle in this example and in number 12 applies to the electron geometry. For the oxygen in number 8, the lone pairs are two sp2 orbitals, which are at 120o to the C=O bond.