In the previous post, we talked about the standard valences and formal charges in organic chemistry. Today, we will focus on the number of lone pairs of electrons. While determining this is extensively covered in the Lewis structures and VSEPR theory, it may get tricky when formal charges need to be considered as well. For example, how many lone pairs does the oxygen have in the following molecule?
![]()
In general, there are two approaches you can use to determine the number of lone pairs. The first one, which is also what you should eventually aim for, is to learn the common bonding patterns of the elements in the second row and recognize the number of lone pairs and formal charges based on those. The following table summarizes these patterns, addressing the common bonding and formal charge combinations:

Depending on how long you have been studying organic chemistry, it may be easy for you to recognize that the oxygen needs two lone pairs to satisfy the octet rule. And according to the table, the oxygen should have two lone pairs of electrons when bonded to two atoms. This combination satisfies the octet rule without a formal charge. Therefore, the correct Lewis structure would be as follows:
![]()
A Formula to Determine the Number of Lone Pairs
The second approach is to use the formula for the formal charge and determine the number of lone pairs just like solving an equation with one unknown. We know from the previous post, that the formal charge can be calculated by this formula:
FC= V – (N + B)
Where:
V – number of valence electrons
N – number of nonbonding electrons
B – number of bonds
Notice that this is not the only formula for calculating the formal charge; however, I figured it was the best variation acceptable to my students.
So, for our molecule, we would use 0 for the formal charge, 6 for the number of valence electrons since oxygen is in group 6, 2 for the number of bonds, and keep the N as the unknown.
Therefore, we will have:
0 = 6 – (N + 2), N = 4
And four non-bonding electrons means two lone pairs, which is what we got when using the table.
Determining the Number of Lone Pairs when there is a Formal Charge
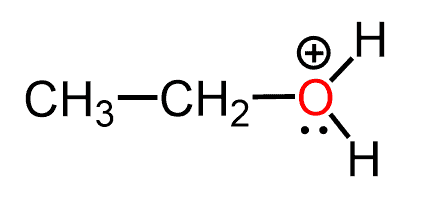
Let’s now consider this question when there is one more hydrogen bonded to the oxygen. How many lone pairs does the oxygen have in the following molecule?
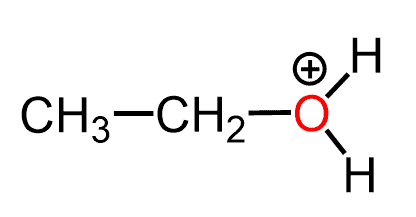
What we notice first is the positive charge on the oxygen which is going to affect the number of lone pairs.
The positive charge indicates that one of the lone pairs that was initially there was used for making the extra bond with the hydrogen and therefore, instead of the normal two lone pairs we expect only one in this case. Which we also find in the table, under the +1 charge of for oxygen:
Let’s also look at some examples with nitrogen. How many lone pairs would you expect to have on the nitrogen in the following molecule?

Nitrogen is usually surrounded by three covalent bonds and a lone pair in its standard, no-formal charge combination. The positive charge here indicates that it cannot have lone pairs since that would exceed the octet around the nitrogen; four bonds and a lone pair, which is a total of 10 electrons:

While exceeding the octet is not impossible, in fact, the octet rule applies only to the elements in the second row, it is still very important in organic chemistry because carbon and the other elements in the second row do obey the rule. Nitrogen is also in the second row and therefore, it cannot have 10 electrons with any bond, lone pair, and formal charge combination.
Looking at these two examples with oxygen and nitrogen, we notice that a positive formal charge implies that a lone pair was on the atom before an additional bond was formed using these lone pairs. So, in our molecule, we can say that there is no lone pair on the nitrogen because it participated in the formation of the fourth bond around the nitrogen.
Number of Lone Pairs with Negative Partial Charge
Contrary to the positive charge, a negative partial charge accounts for an additional one pair. For example, carbon is normally associated with four bonds and no lone pairs. However, when bonded with metals, the carbon is often said to be negatively charged because of the large difference in the electronegativity values, even though these bonds are often covalent.
Later, in your organic chemistry class, you will see lots of structures where the lone pair is not shown, but the atom is negatively charged. It will be presumed that you recognize the presence of the lone pair(s) based on the formal charge.
Let’s try to figure out how many lone pairs we should expect in the following structure:
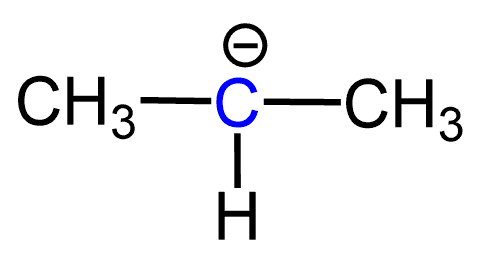
First, remember that carbon “likes” four bonds, and most of the time, carbon atoms are going to have four bonds and no formal charge. A negatively charged carbon atom should immediately tell you about a lone pair of electrons. In this case, since the carbon has only three bonds and a negative charge, it must also have a lone pair.
This can also be confirmed by using the formula:
FC= V – (N + B)
-1 = 4 – (N + 3)
N = 2, thus, one lone pair of electrons
Therefore, we have the following structure with a lone pair for this carbanion:

To summarize, when asked to determine the number of lone pairs, remember that electrons are associated with a negative charge, so if the atom is negatively charged, consider the possibility of having a lone pair(s). Likewise, a partial positive charge should be a hint that the atom does not have as many bonds (electrons) as it should.