Angle Strain and Torsional Strain
Cycloalkanes are less stable than their open-chain analogs. For example, butane is more stable than cyclobutane, and propane is far more stable than cyclopropane. It may visually be obvious why they are less stable without going much into the details-why would the molecule be happy if you bend it like that?
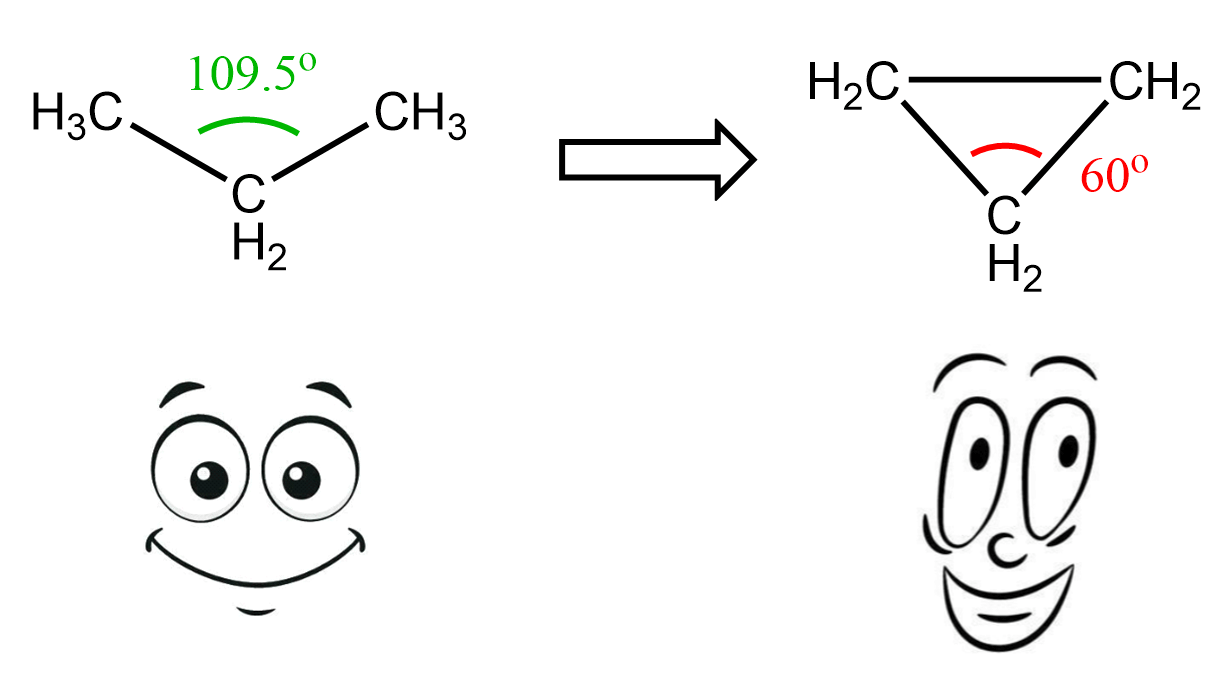
And that is essentially the answer to why cycloalkanes are less stable than open-chain alkanes. It is even more evident if we recall that in alkanes and cycloalkanes the carbons are sp3-hybridized, which means their ideal bond angle is 109.5o. A glimpse of memories from geometry class should remind us that the angles in a triangle are 60o, and 90o in a square:
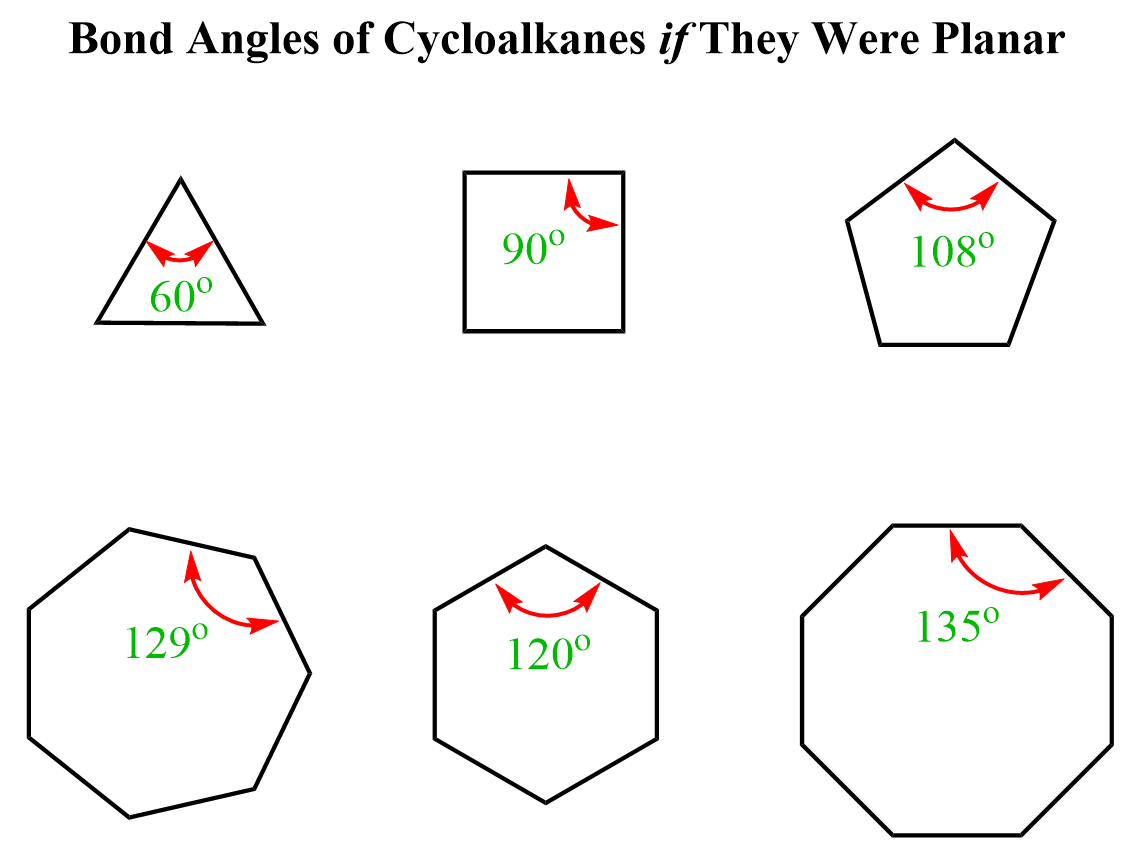
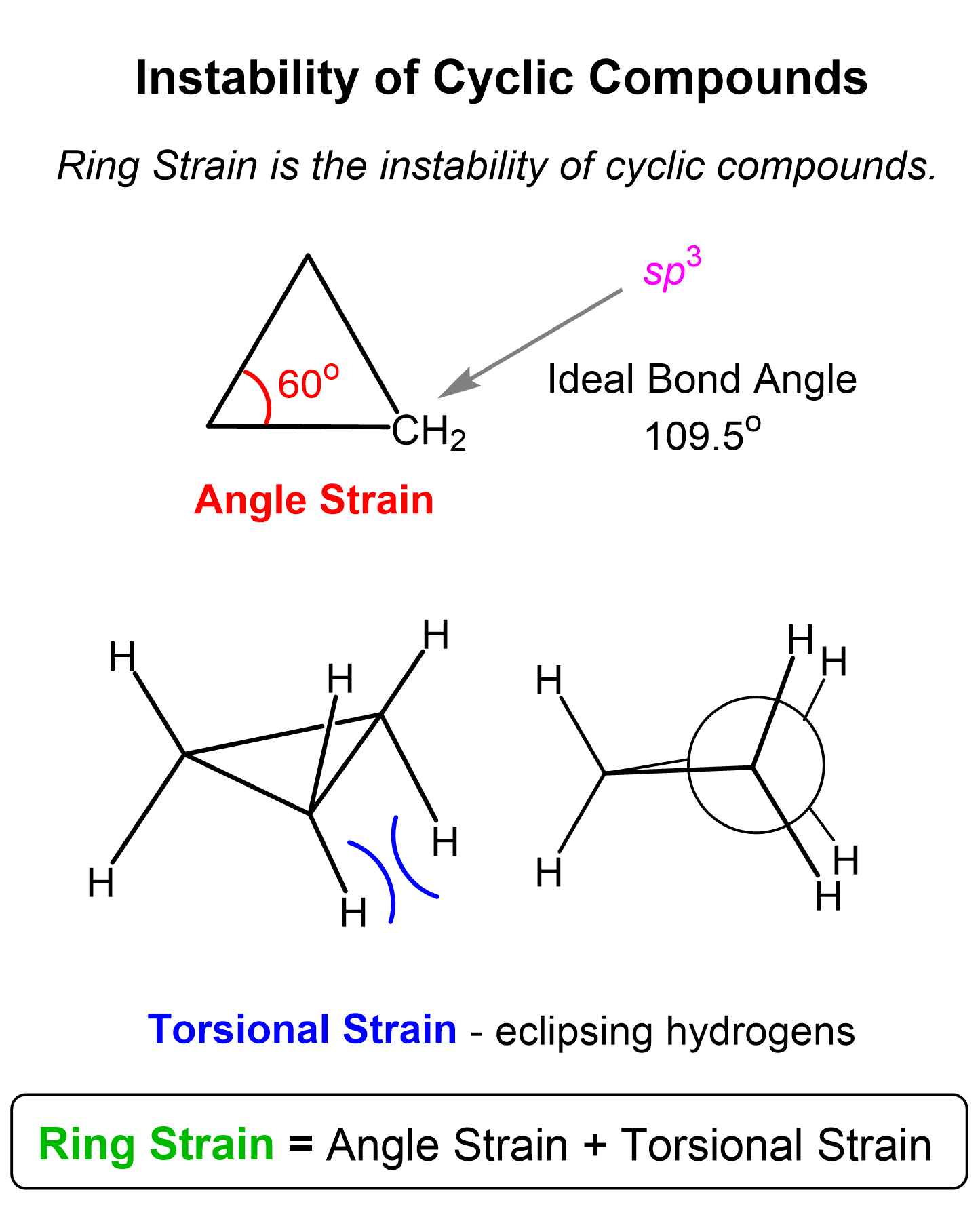
So, we are bending the angles, thus making the molecule unstable. This instability, caused by the deviation from the ideal tetrahedral angle, is called angle strain.
The deviation from the ideal bond angle causes a poor overlap of the orbitals, making the C-C sigma bond, which explains the bond angle strain. Recall that sigma bonds are formed by linear head-on overlap of the bonding orbitals, which is impossible for cyclopropane and cyclobutene because the angle between the carbon atoms is far smaller than 109.5o:

The angle strain, however, is not the only factor making cycloalkanes unstable. For example, if we make a 3D representation of cyclopropane, we can see that all the hydrogens are right in front of each other. This is what we call an eclipsed arrangement, which, we know from Newman projections that is not the optimal orientation because of torsional interactions. Torsional interactions are repulsive forces caused by the bonding electrons of the eclipsed atoms. For example, this short video demonstrates the torsional and steric interactions in the eclipsed conformations of butane:
The angle strain and torsional strain together make what is called ring strain:
The pattern of cycloalkanes being less stable than their linear counterparts is true for all the cycloalkanes. However, the more severe the deviation from the ideal tetrahedral angle and the number of eclipsing hydrogens, the more unstable the cycloalkane. As expected, the most unstable is cyclopropane, so let’s discuss it and other cycloalkanes to find ways of minimizing this unfavorable energetic uphill.
Cyclopropane
Cyclopropane is the smallest cycloalkane, which makes the angles more strained than in other cycloalkanes. Importantly, it has no way of avoiding being planar and making more room for the eclipsed hydrogen atoms:
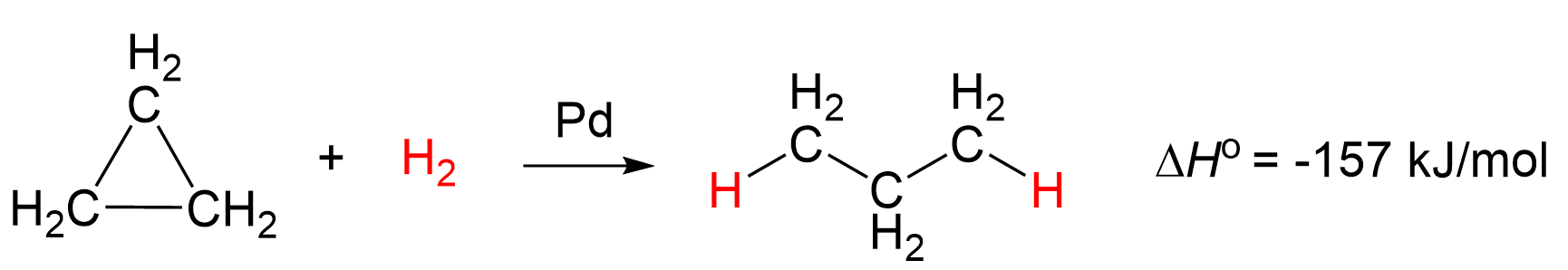
Therefore, it is the least stable cycloalkane, and for comparison, the bond dissociation energy of a C–C bond in cyclopropane is about 65 kcal/mol (272 kJ/mol), much lower than the 90 kcal/mol (377 kJ/mol) in a typical alkane like ethane.
Cyclobutane
Unlike cyclopropane, the for-membered ring gets some flexibility and avoids being planar, which significantly reduces torsional strain, thus the overall ring strain. In its slightly bent geometry, also known as a puckered conformation, the bond angle is 88o. This is smaller than the 90o of the planar geometry, but it is still energetically more favorable than having all the C-H bonds eclipsing:
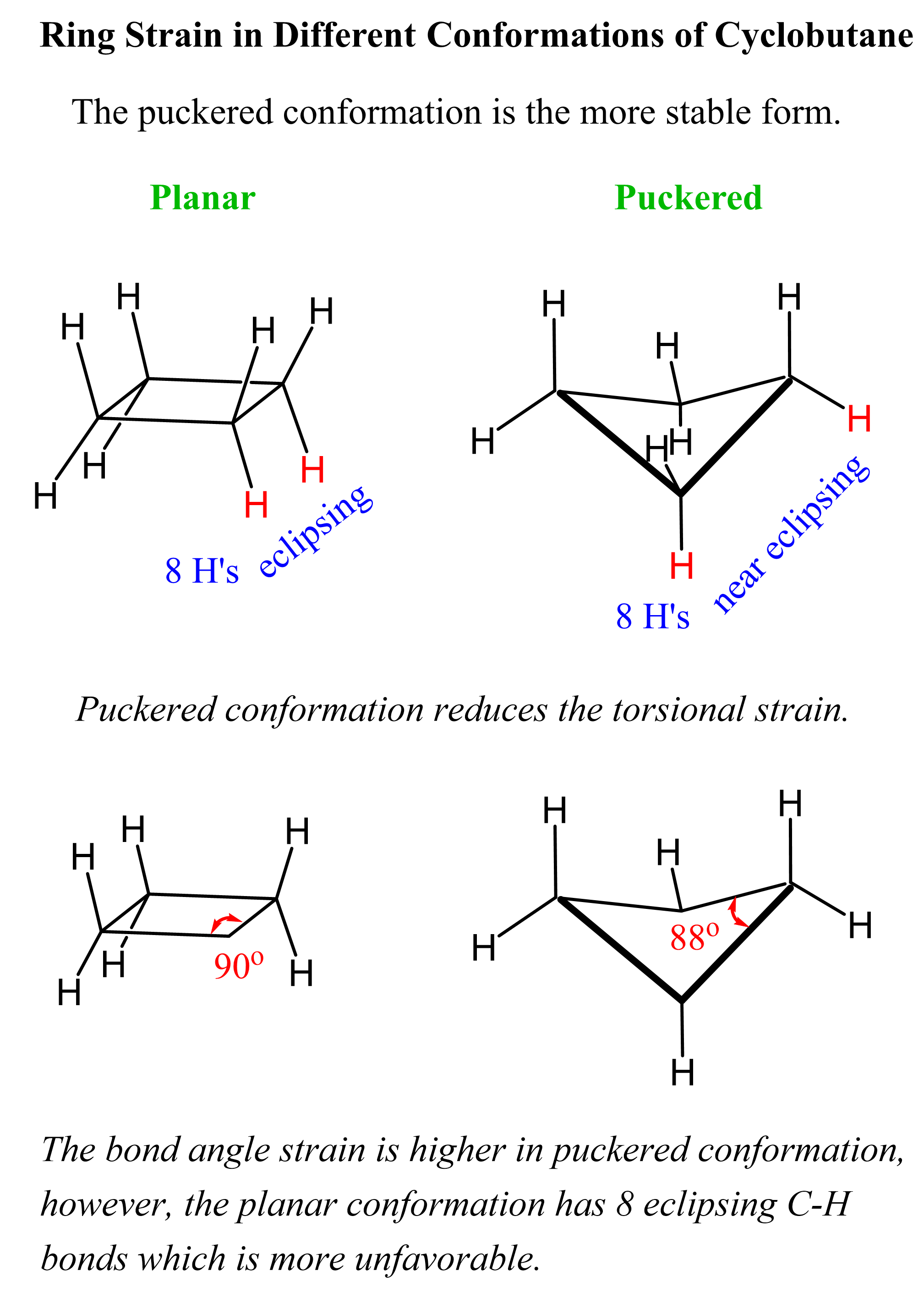
Cyclopentane
At first glance, cyclopentane seems like it should be very stable. As we have seen above, the internal bond angles of a pentagon are 108°, which may seem like it must be lacking any instability because of the nearly perfect match to the tetrahedral angle of 109.5o. However, it turns out that it is impossible to avoid any torsional interactions even in the most optimal geometry of cyclopentane known as the envelope conformation.
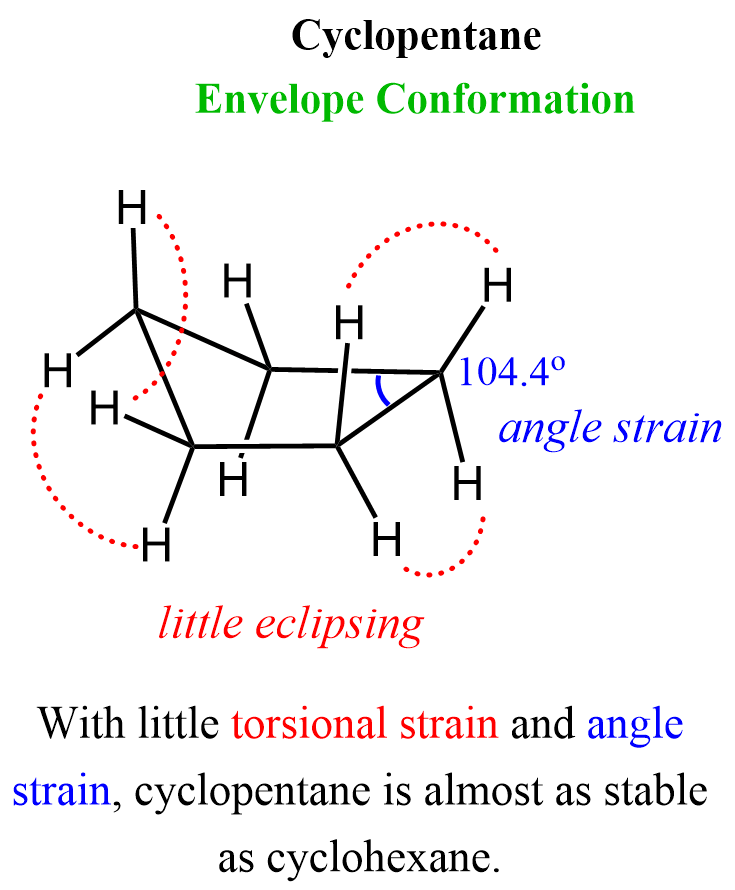
Overall, cyclopentane is fairly stable. It has some residual ring strain, but much less than cyclopropane or cyclobutane. The flexibility of its ring allows it to balance between minimizing torsional and angle strain, which gives it moderate stability – less than cyclohexane, but more than the smaller rings.
Cyclohexane
Cyclohexane is the most common ring structure found in both natural and synthetic compounds, and the reason for this is its exceptional stability. Unlike smaller cycloalkanes we have seen so far, it avoids both angle strain and torsional strain by adopting the most stable conformation known as the chair conformation.
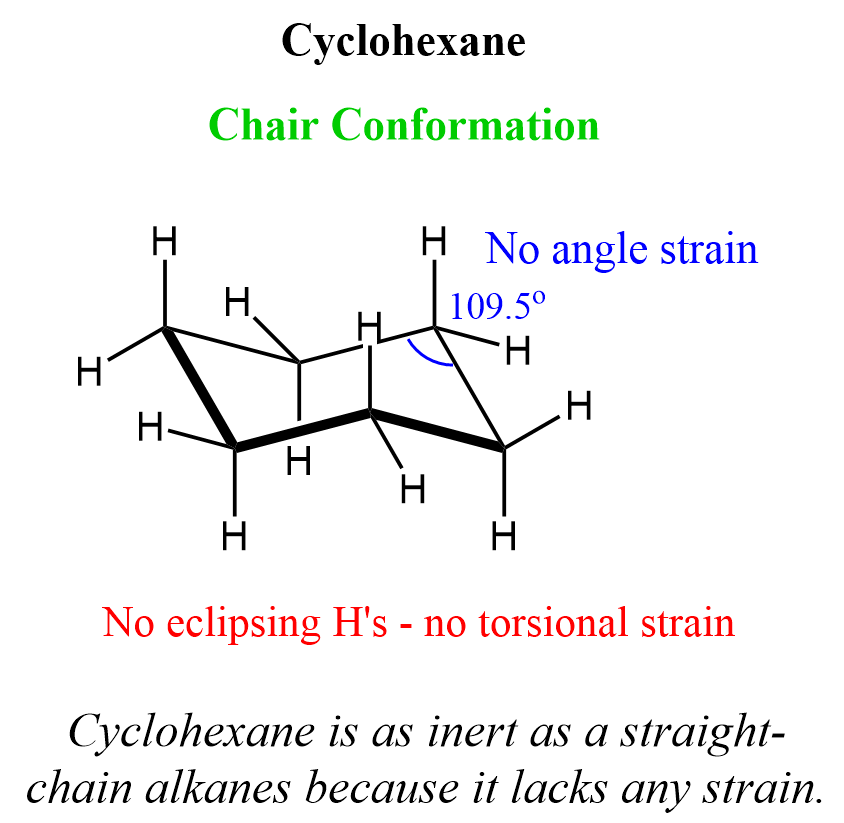
In this geometry, the carbon atoms are positioned so that the bond angles are very close to the ideal tetrahedral angle of 109.5° (sometimes reported as 111.4°), and all the C−H bonds are staggered, which eliminates any eclipsing interactions. We can see this in Newman projections of the chair conformation:
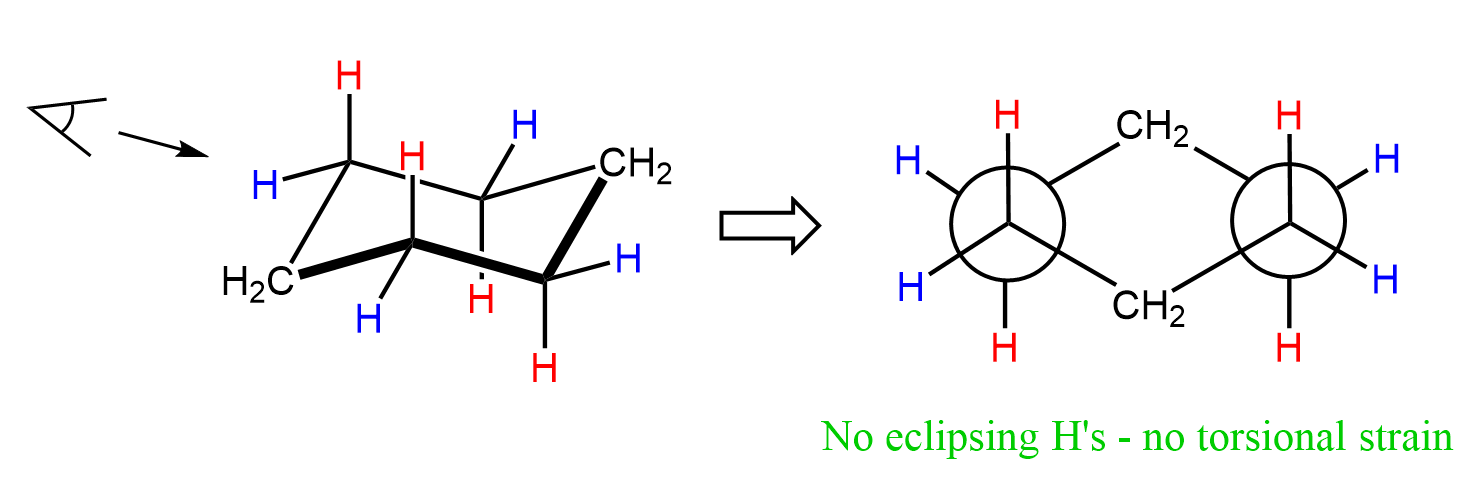
Let’s take a look at the conformational analysis of cyclohexane. On both ends of the energy diagram, we have two equivalent chair forms. In between these forms, the molecule adopts several other conformations, which are all higher in energy, thus less stable than the chair conformation.
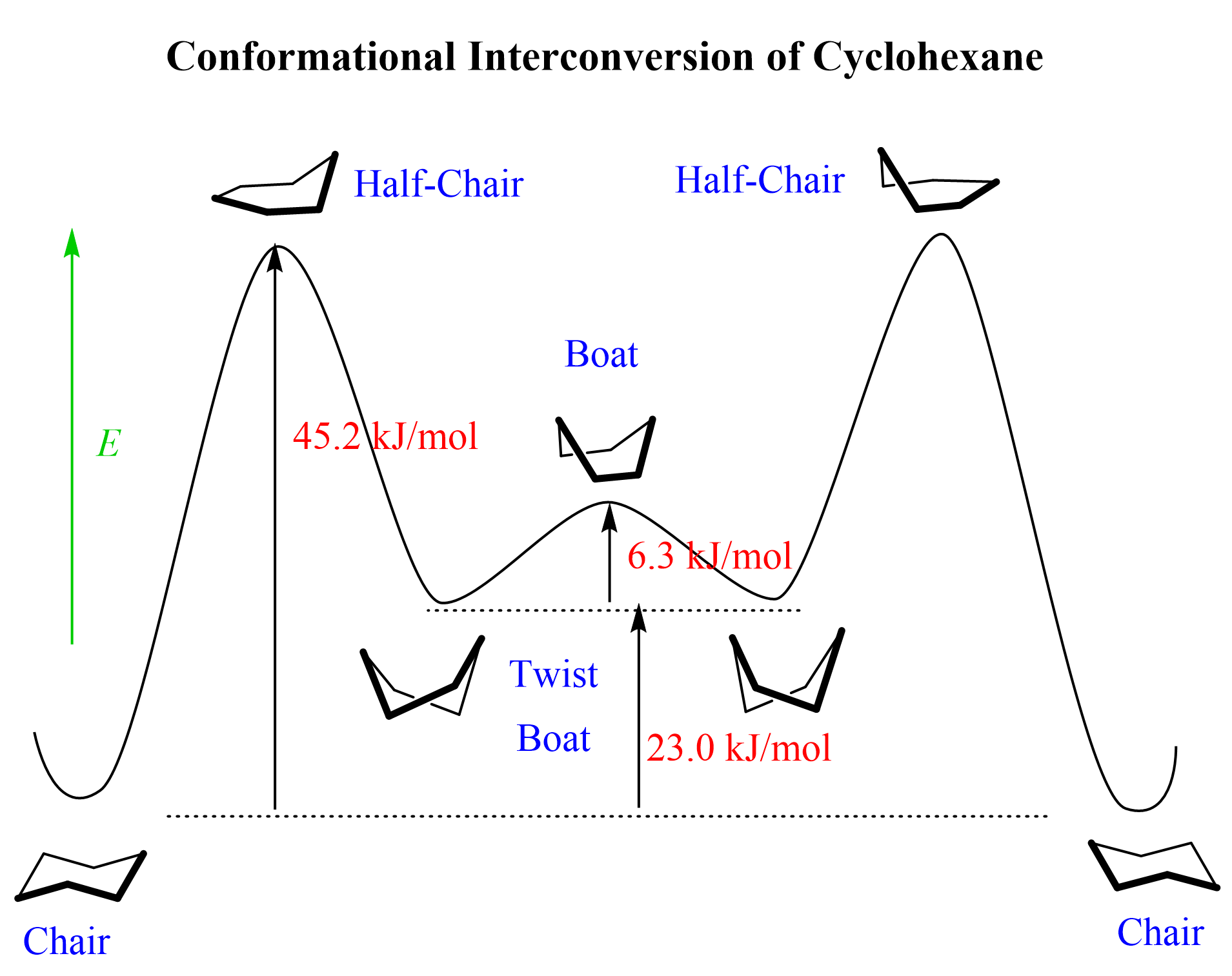
The process of interchanging between two chair conformations is called ring-flip, which happens via rotation about the C-C single bonds:
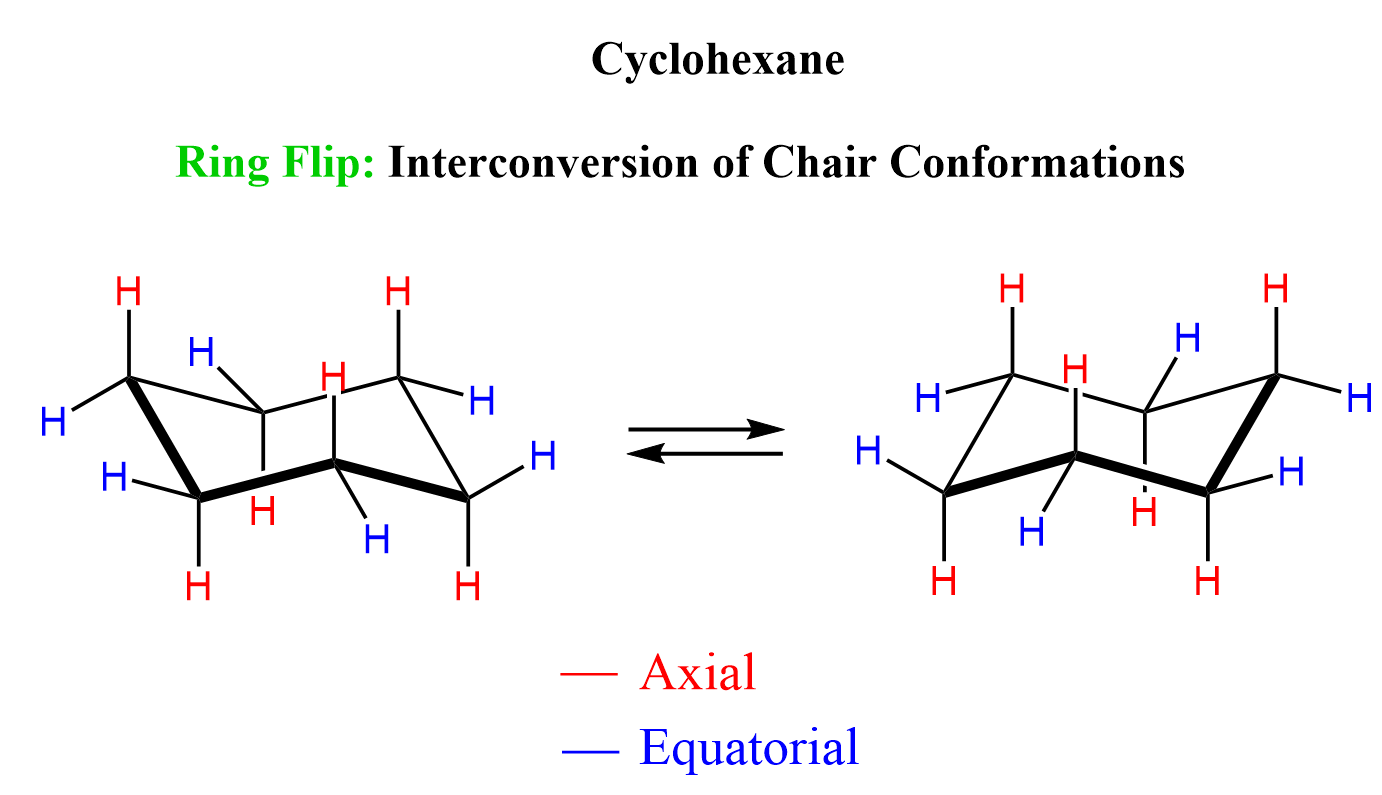
The color-coded hydrogens don’t represent the same atom. I did that to rather illustrate their positions as axial or equatorial in each chair conformation.
Being able to draw chair conformations of cyclohexane and identify the axial and equatorial positions is a core skill in organic chemistry. We also have separate articles covering ring flip, conformational stability, and 1,3-diaxial interactions in substituted cyclohexanes, so feel free to explore those as well.
Summarizing the Stability of Cycloalkanes
The stability of cycloalkanes varies depending on the size of the ring, and this is largely due to the presence or absence of ring strain. As we’ve seen, small rings like cyclopropane and cyclobutane suffer from significant angle and torsional strain, making them the least stable. Cyclopentane shows improved stability, though it still has some strain, while cyclohexane stands out as the most stable due to its ability to adopt a strain-free chair conformation.
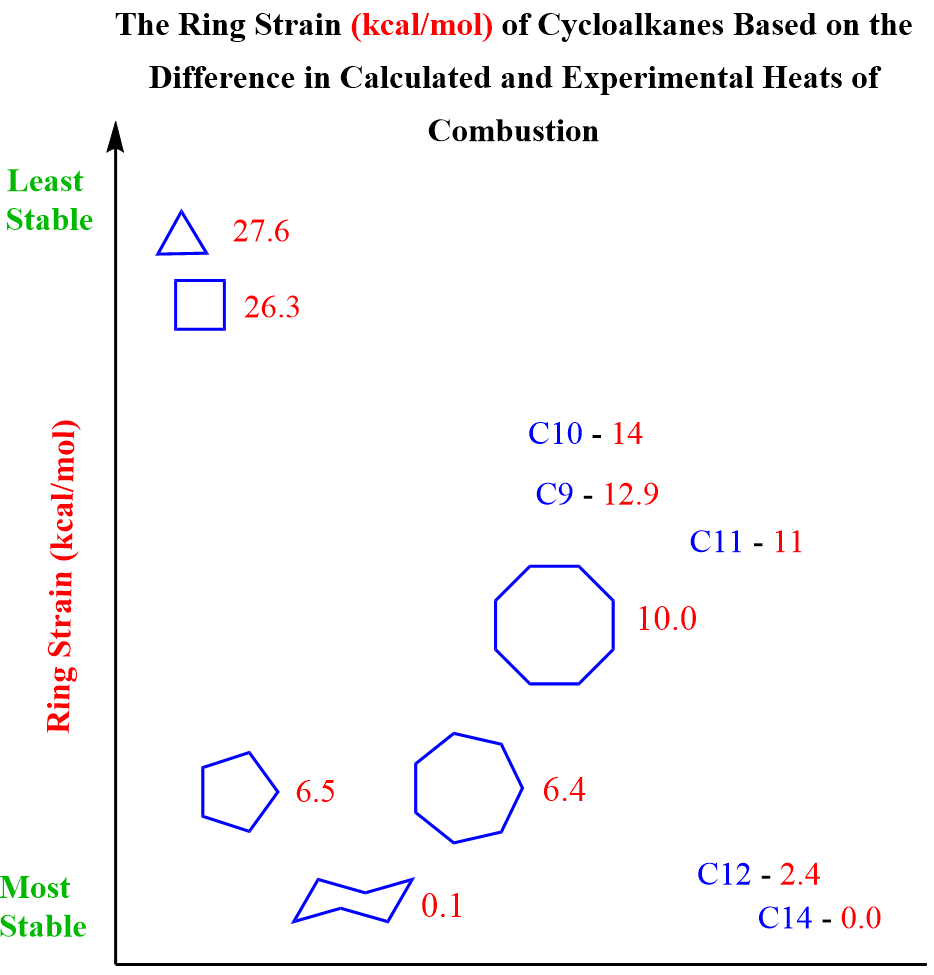
As ring size increases beyond six atoms, strain begins to build up again due to unfavorable conformations and increased torsional strain. The table below summarizes the calculated ring strain energies for cycloalkanes from C3 to C14, offering a clear comparison of their relative stabilities.
How is the Stability of Cycloalkanes Calculated?
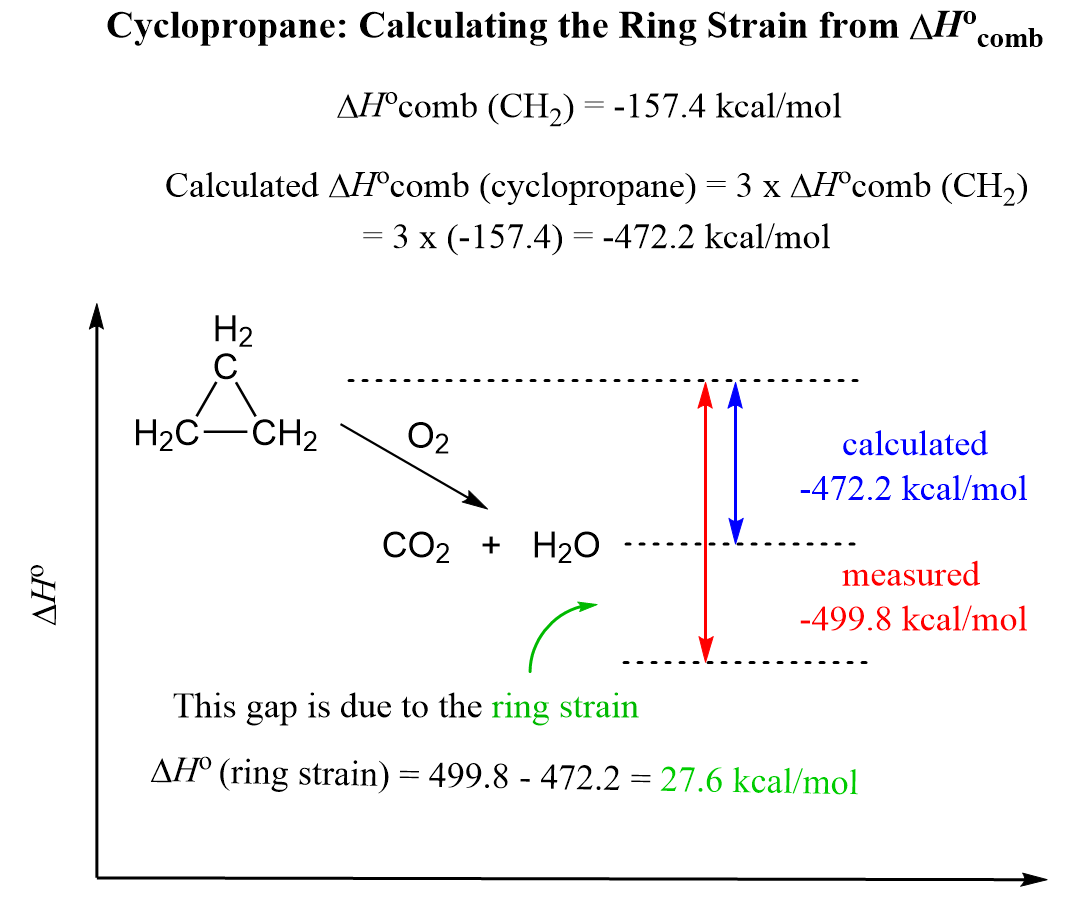
Ring strain is calculated by comparing the experimental heat of combustion of a cycloalkane to the theoretical value expected if it had no strain, meaning if it were like a straight-chain alkane only with both ends connected. So, for cyclopropane, we’d simply multiply the heat of combustion of the CH₂ group (157.4 kcal/mol) by 3.
Calculated ΔHocomb (cyclopropane) = 3 x ΔHocomb (CH2) = 3 x (-157.4) = -472.2 kcal/mol
Once again, this is the calculated heat of combustion, ignoring any ring strain. Next, we compare it with the experimental data – the actual heat of combustion, which, for cyclopropane, is -499.8 kcal/mol. They don’t match, and this difference can only be due to the ring strain; therefore:
Ring strain = ΔHocomb (actual) – ΔHocomb (calculated)
Ring strain (cyclopropane) = 499.8 – 472.2 = 27.6 kcal/mol (115.5 kJ/mol)
In similar experiments, the ring strain, or the absence thereof, was determined for other cycloalkanes, and the data are summarized in the table above.
Check Also
- Naming Alkanes by IUPAC Nomenclature Rules Practice Problems
- Naming Bicyclic Compounds
- Naming Bicyclic Compounds-Practice Problems
- How to Name a Compound with Multiple Functional Groups
- Primary, Secondary, and Tertiary Carbon Atoms in Organic Chemistry
- Constitutional or Structural Isomers with Practice Problems
- Degrees of Unsaturation or Index of Hydrogen Deficiency
- The Wedge and Dash Representation
- Sawhorse Projections
- Newman Projections with Practice Problems
- Staggered and Eclipsed Conformations
- Conformational Isomers of Propane
- Newman Projection and Conformational Analysis of Butane
- Newman Projection of Chair Conformation
- Gauche Conformation
- Gauche Conformation, Steric, Torsional Strain Energy Practice Problems
- Ring Strain
- Steric vs Torsional Strain
- Conformational Analysis
- Drawing the Chair Conformation of Cyclohexane
- Ring Flip: Drawing Both Chair Conformations with Practice Problems
- 1,3-Diaxial Interactions and A value for Cyclohexanes
- Ring-Flip: Comparing the Stability of Chair Conformations with Practice Problems
- Cis and Trans Decalin
- IUPAC Nomenclature Summary Quiz
- Alkanes and Cycloalkanes Practice Quiz
