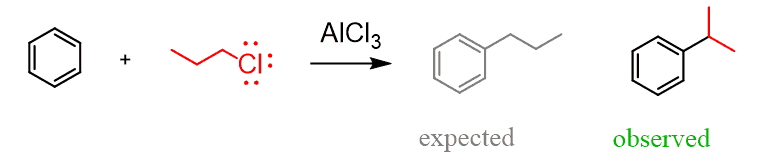
We have seen, in the Friedel-Crafts alkylation reaction, that rearrangements do not allow for the preparation of primary alkyl benzenes:
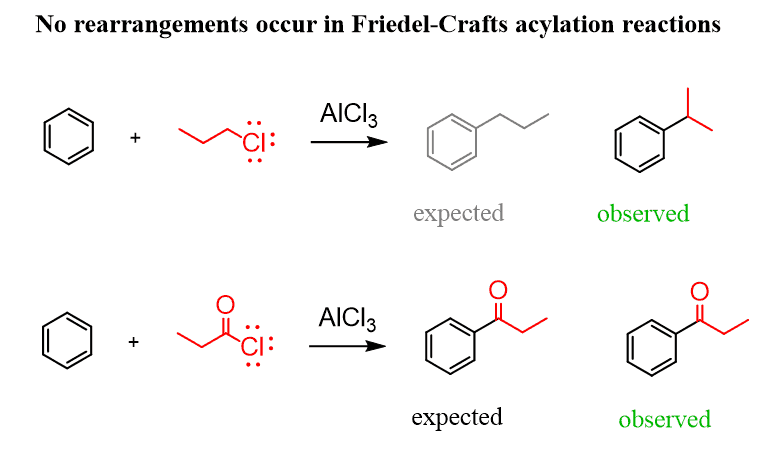
To overcome this limitation, you can first prepare the corresponding aryl ketone by the Friedel-Crafts acylation and then reduce the carbonyl to the alkyl group. The difference here is that, unlike the alkyl carbocations formed in the Friedel-Crafts alkylation, the acyl cation does undergo a rearrangement, and it is possible to perform the synthesis as planned:
The benzylic carbonyl can be reduced by hydrogen using a metal catalyst. Notice that other ketones are reduced to alcohols, and only aryl ketones can be reduced to a methylene group by catalytic hydrogenation (H2 + Pt or Pd/C):
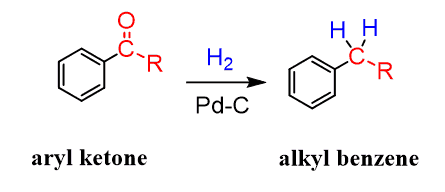
Wolff-Kishner and Clemenson Reduction
The two most common reactions for performing this reduction homogeneously are the Clemenson and Wolff-Kishner reactions:
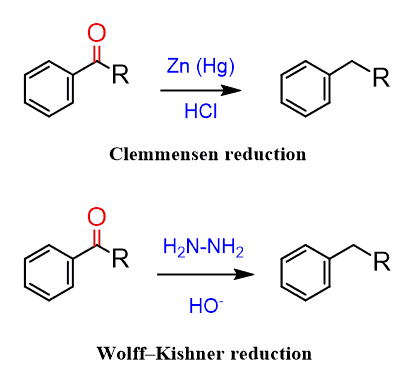
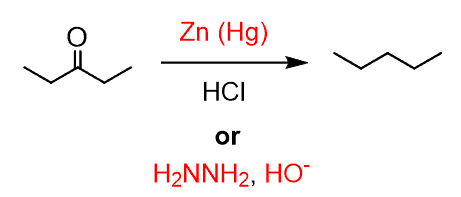
These reactions are general ways of reducing carbonyls and are not restricted to benzylic carbonyls only:
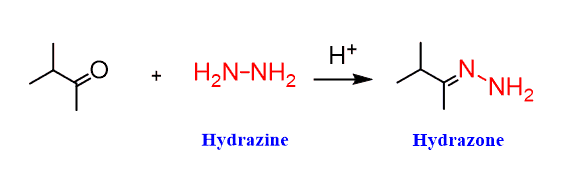
In the Wolff-Kishner reaction, hydrazine (H2N-NH2) reacts with the ketone just like in any reaction of amines with carbonyl compounds, forming an imine (Schiff base). In this case, a hydrazone is formed, which is an analog of imines:
The base and high temperature are used for cleaving the nitrogens according to the following mechanism:

Can you draw the mechanism for this reaction?
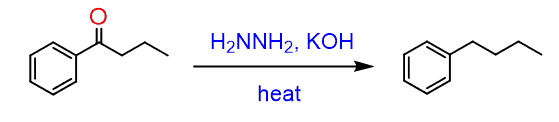
Without looking at the other one 🙂
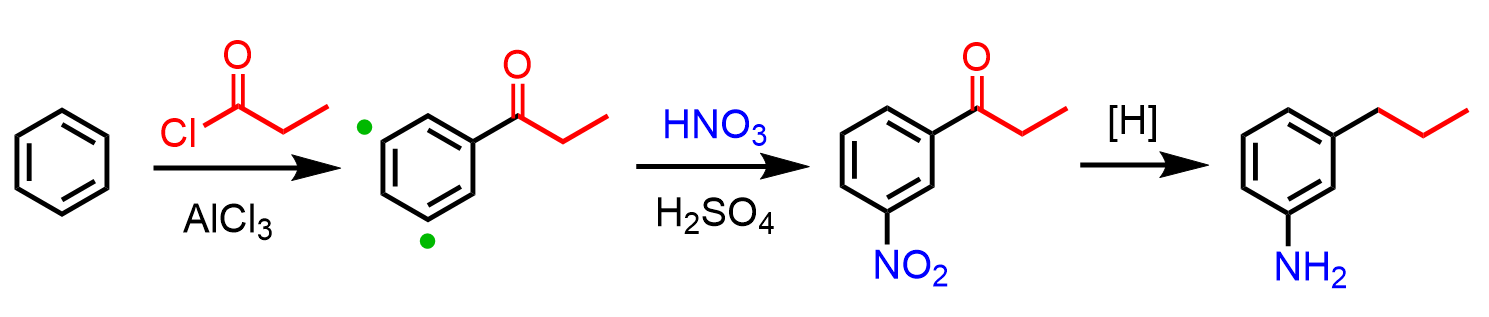
An important application of this strategy is the synthesis of a benzene derivative with meta-oriented substituents. For example, how can we synthesize 3-propylaniline from benzene?
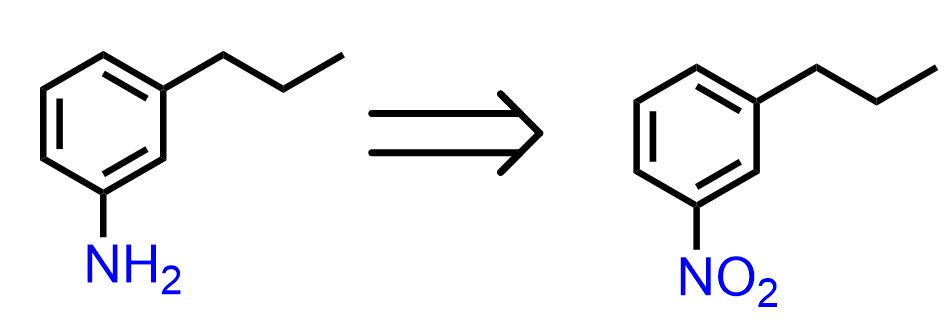
So, the next question is, how do we synthesize the 1-nitro-3-propylbenzene? Nitrobenzene is a meta director, so can it be prepared by a Friedel-Crafts alkylation of nitrobenzene?
As we have mentioned earlier in the article, this reaction does not work because Friedel-Crafts do not work on deactivated rings, and in addition, even if the reaction worked, there would’ve been a rearrangement of the primary alkyl halide:
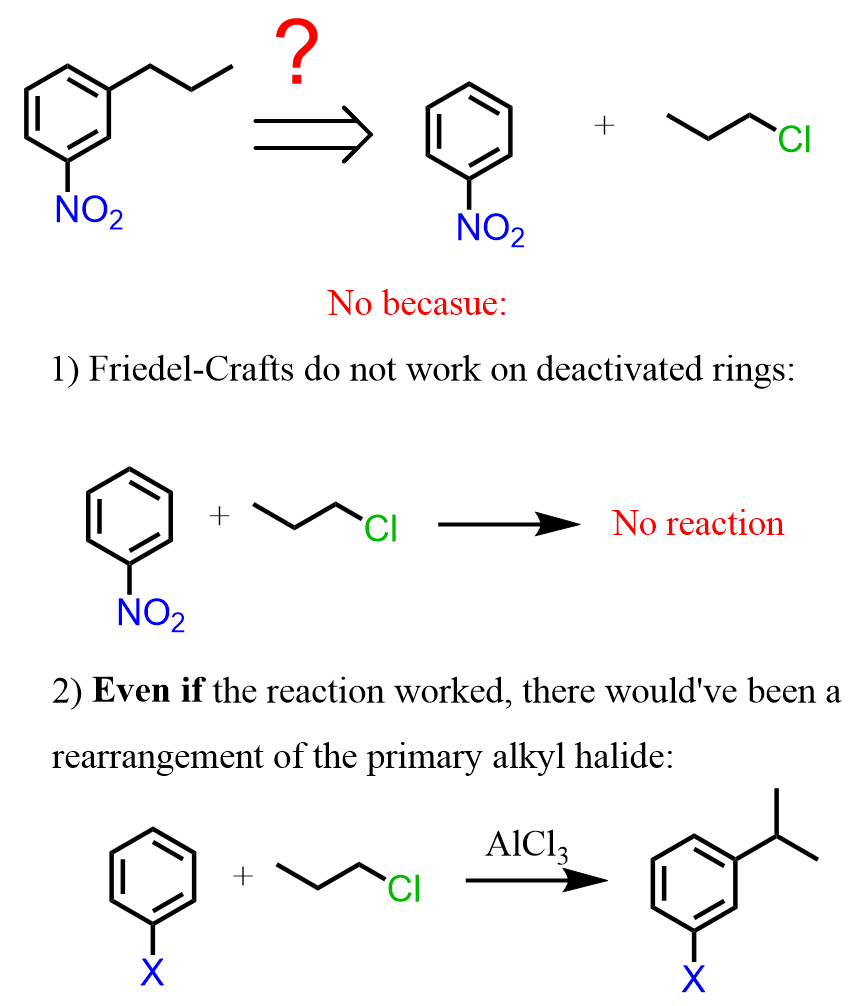
Therefore, the strategy is to nitrate the do the Friedel-Crafts first and then nitrate the ring. The question is, how do we add the nitro group in the meta position if the propyl group is an activator and an ortho, para director?
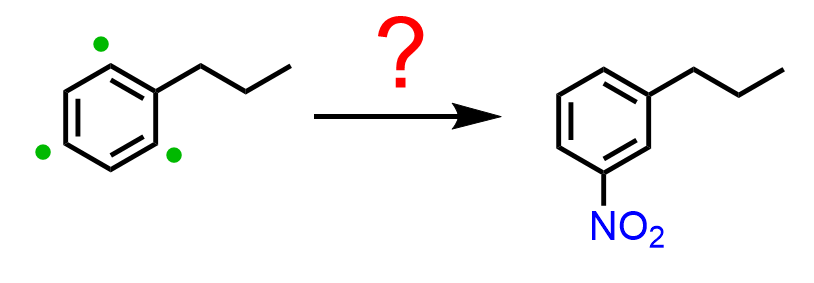
In these cases, Friedel-Crafts acylation followed by a reduction of the carbonyl is used. So, first, we acylate benzene, and the reactive side is now the meta position, which allows us to add the nitro group. The final product with two activators in meta orientation is achieved by reducing the nitro and carbonyl groups: