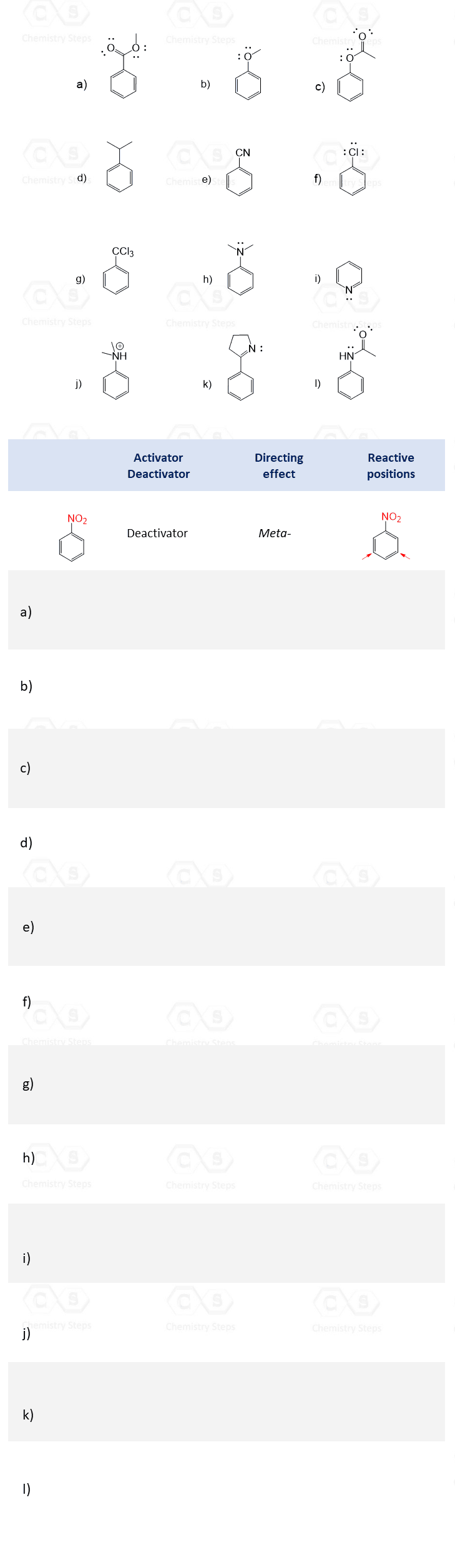
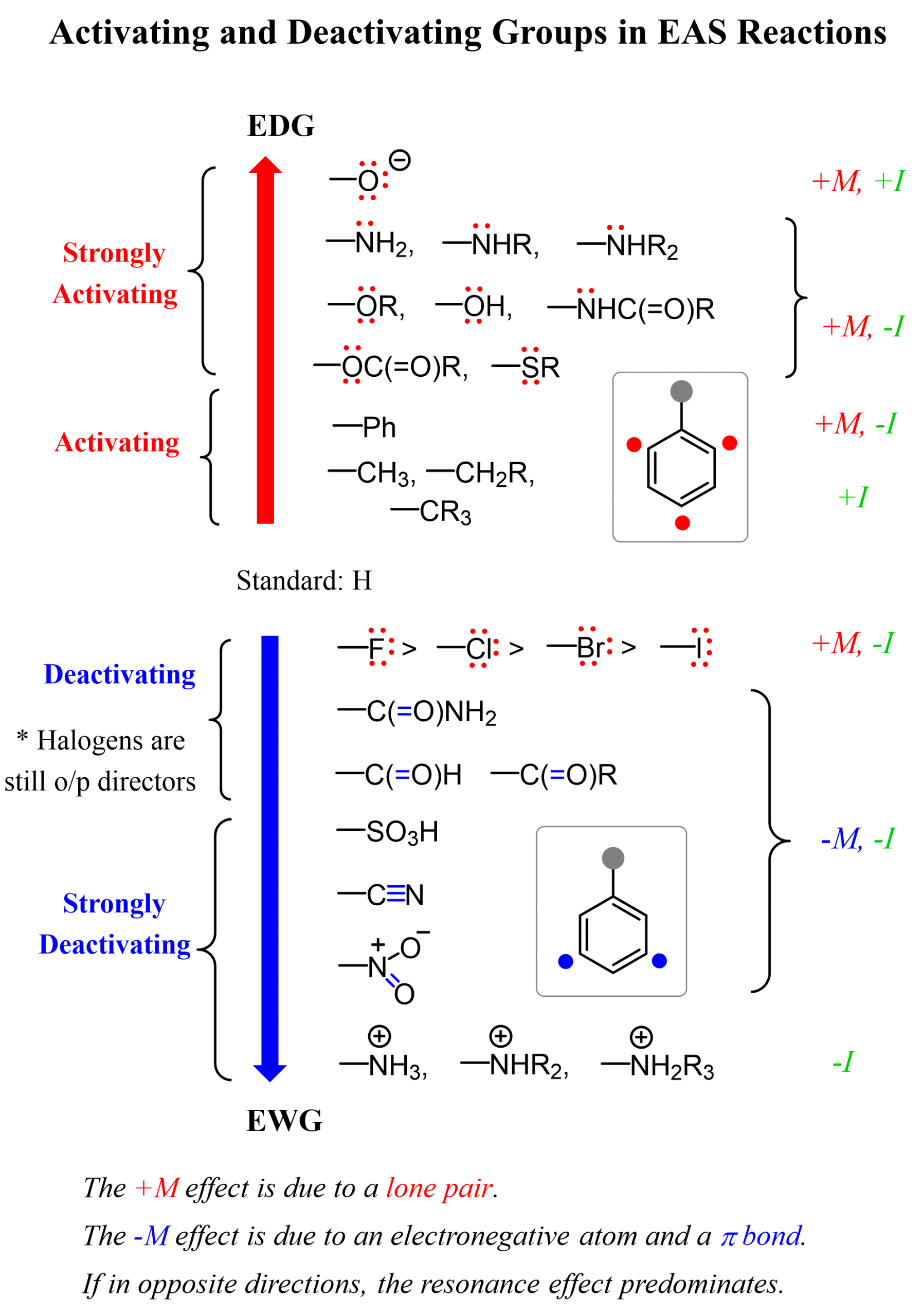
In the previous post, we talked about the ortho-, meta-, and para-directors in electrophilic aromatic substitution (EAS). Substituents on a benzene ring influence both the reactivity of the ring and the position where new groups enter.
- Activating groups (such as -OH, -OCH₃, -NH₂) donate electron density into the ring, making it more reactive and directing incoming substituents to the ortho and para positions.
- Deactivating groups (such as carbonyls, -NO₂, -CF₃, -CN) withdraw electron density, making the ring less reactive. Most of the time, this results in directing substitution to the meta position, except halogens, which are deactivating yet ortho/para-directing due to their lone-pair resonance.
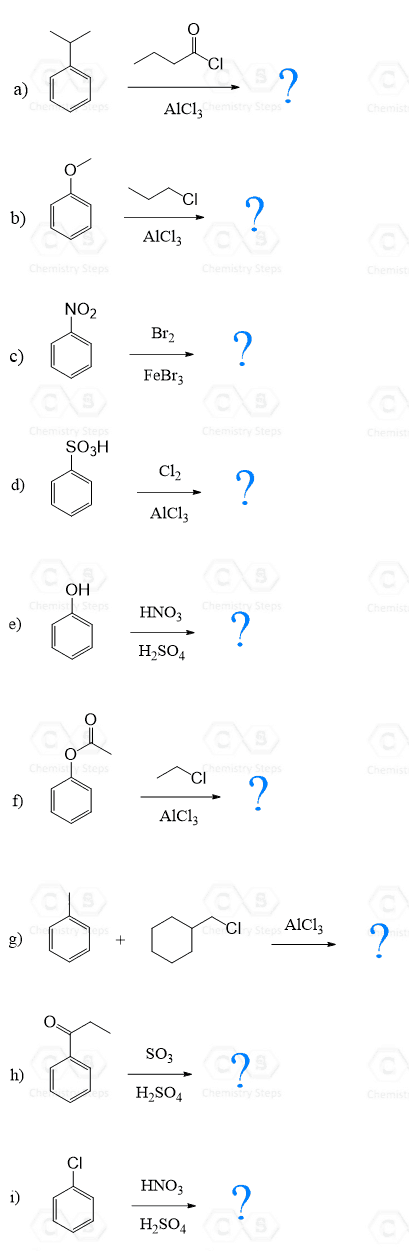
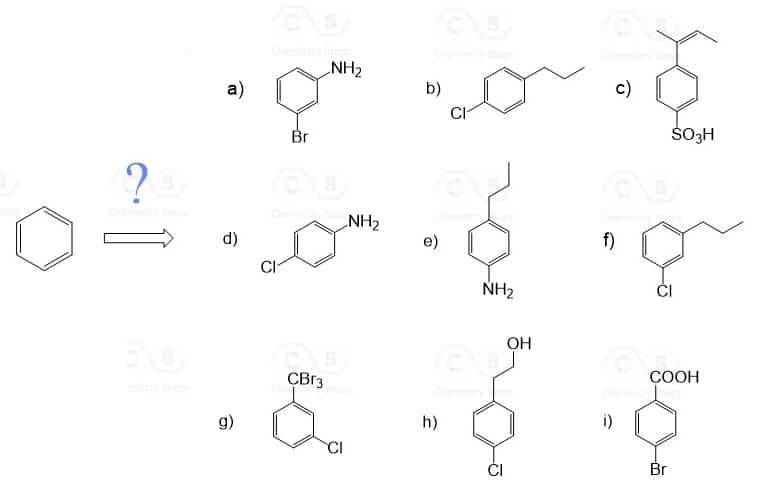
Here are some practice problems on ortho, para, and meta substitution to strengthen your understanding of substitution patterns.