In the previous post, we learned about the electrophilic aromatic substitution reactions and went over the mechanisms of halogenation, nitration, and sulfonation of the benzene ring.
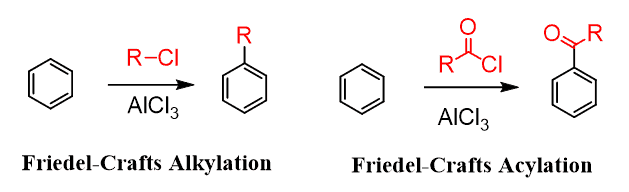
Following an identical mechanism, two reactions, discovered by Charles Friedel and James Crafts in 1877, allow for making a new carbon–carbon bond with the aromatic ring. We can show the following general equations for the Friedel–Crafts alkylation and Friedel–Crafts acylation:
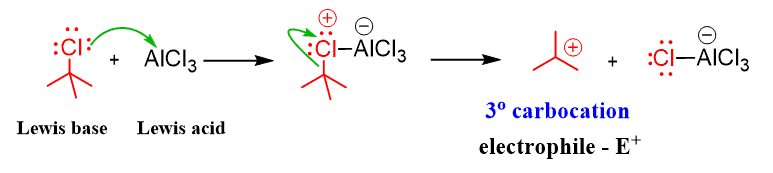
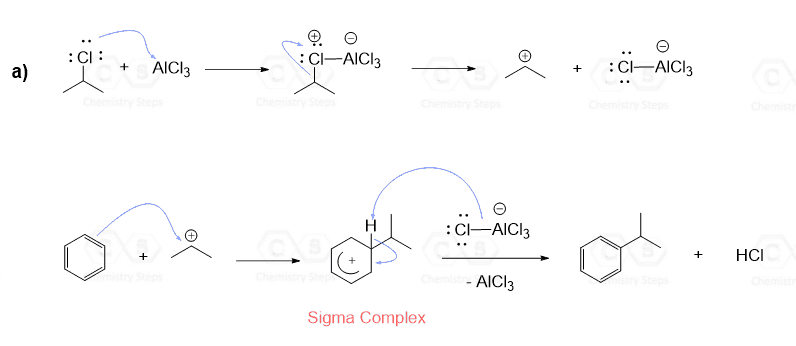
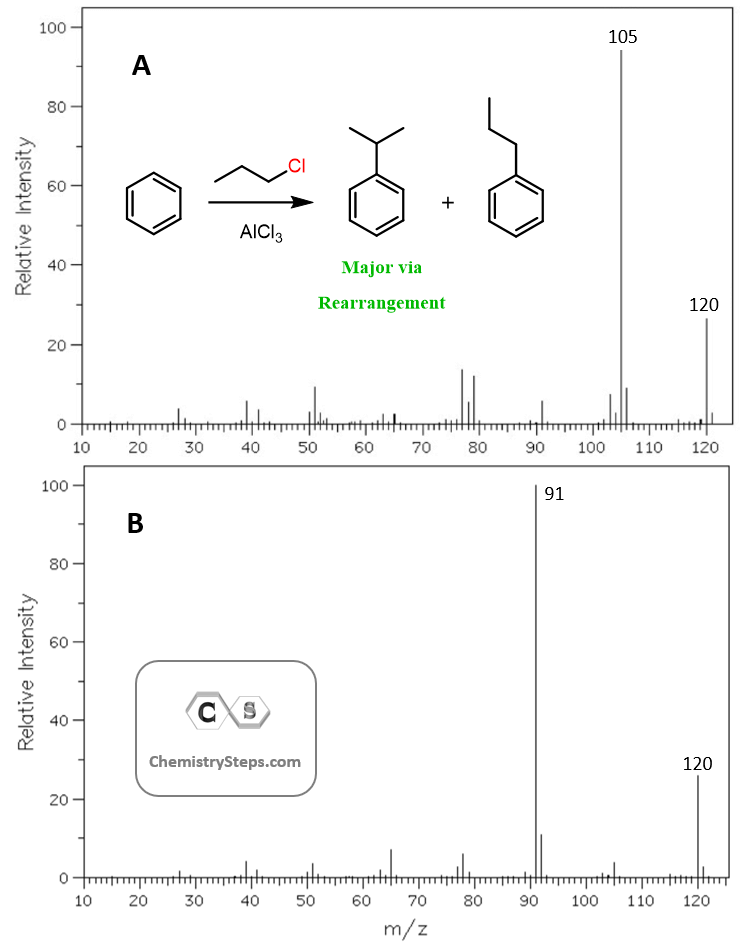
As an example, let’s show the mechanism for the reaction of t-butyl chloride with benzene. The first step is a Lewis acid-base reaction where the activator AlCl3 takes a lone pair from Cl and eventually breaks it off from the carbon. As a result, a 3o carbocation is formed which is a strong electrophile and can be attacked by the aromatic ring:
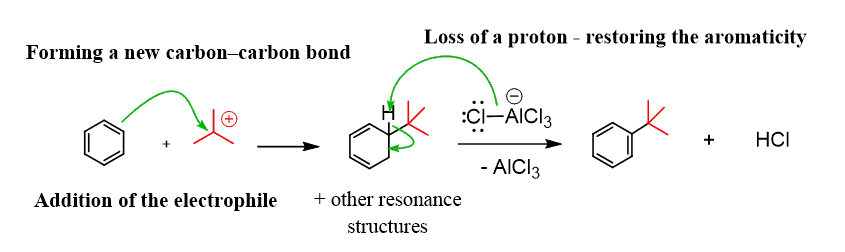
The alkylation of benzene goes according to the general mechanism of the electrophilic aromatic substitution reactions:
Rearrangements in Friedel–Crafts alkylation
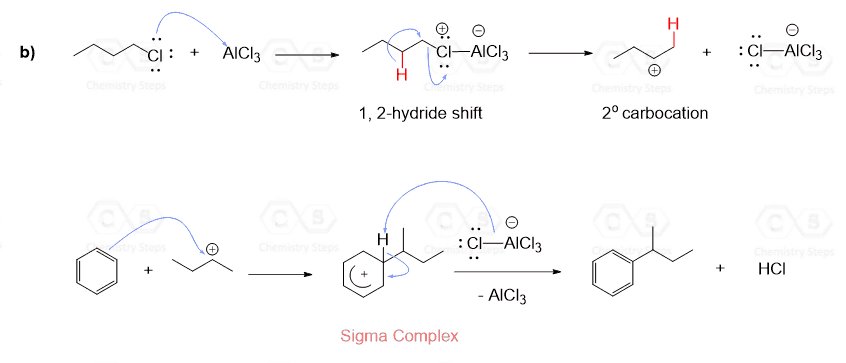
We saw that the Friedel–Crafts alkylation involved a carbocation, and remember, carbocations can undergo rearrangement reactions by hydride or methyl shift.
For example, how do we explain the following reaction, where we start with a primary alkyl halide, but the carbon connected to the aromatic ring in the final product is a secondary carbon?
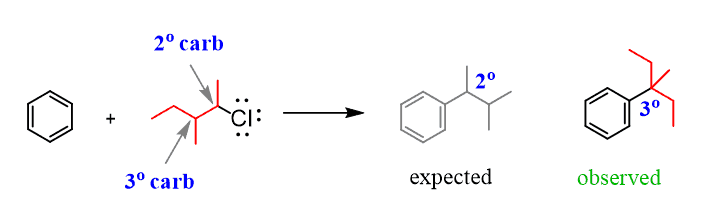
The following mechanism, showing a rearrangement of a secondary carbocation to a more stable tertiary carbocation, explains this observation:
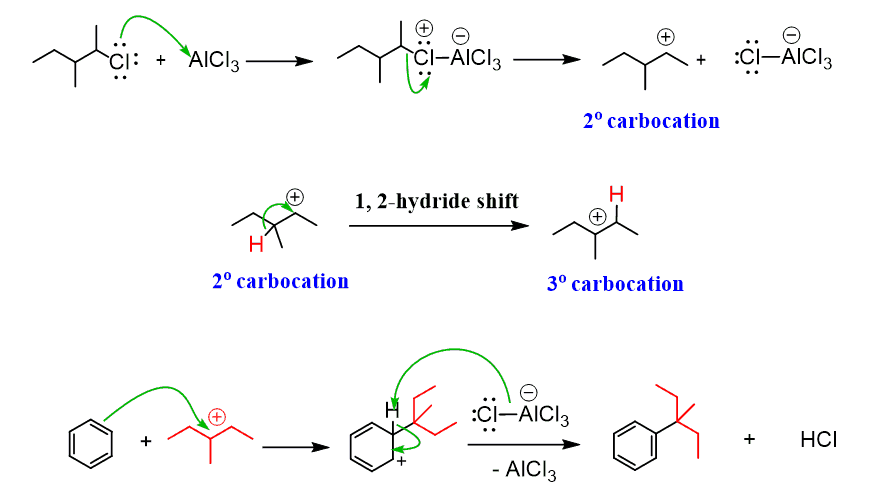
Rearrangements affect the Friedel–Crafts alkylation even when primary alkyl halides are used and no free carbocations are initially formed:
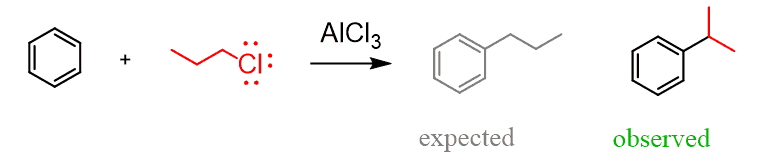
The rearrangements of primary carbocations are surprising because they shouldn’t be formed in the first place. And this is correct, primary carbocations are generally not observed, and instead there a complex is formed in the Lewis acid-base reaction has a very electrophilic carbon connected to the Cl, and it still promotes a 1,2-hydride shift:
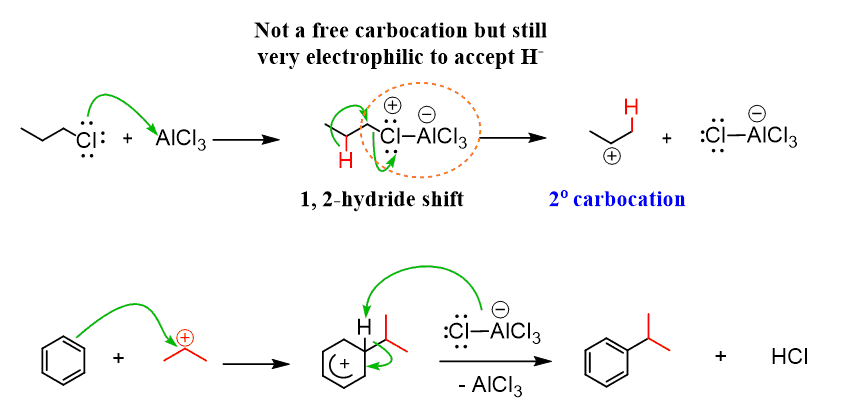
This is similar to what we have seen in the dehydration of primary alcohols:

Whenever possible, rearrangements are going to occur, and a mixture of alkylated rings will be obtained. The ratio can be optimized based on the conditions, such as the concentrations and temperature.
With this said, when is it possible to avoid rearrangements in Friedel–Crafts alkylation?
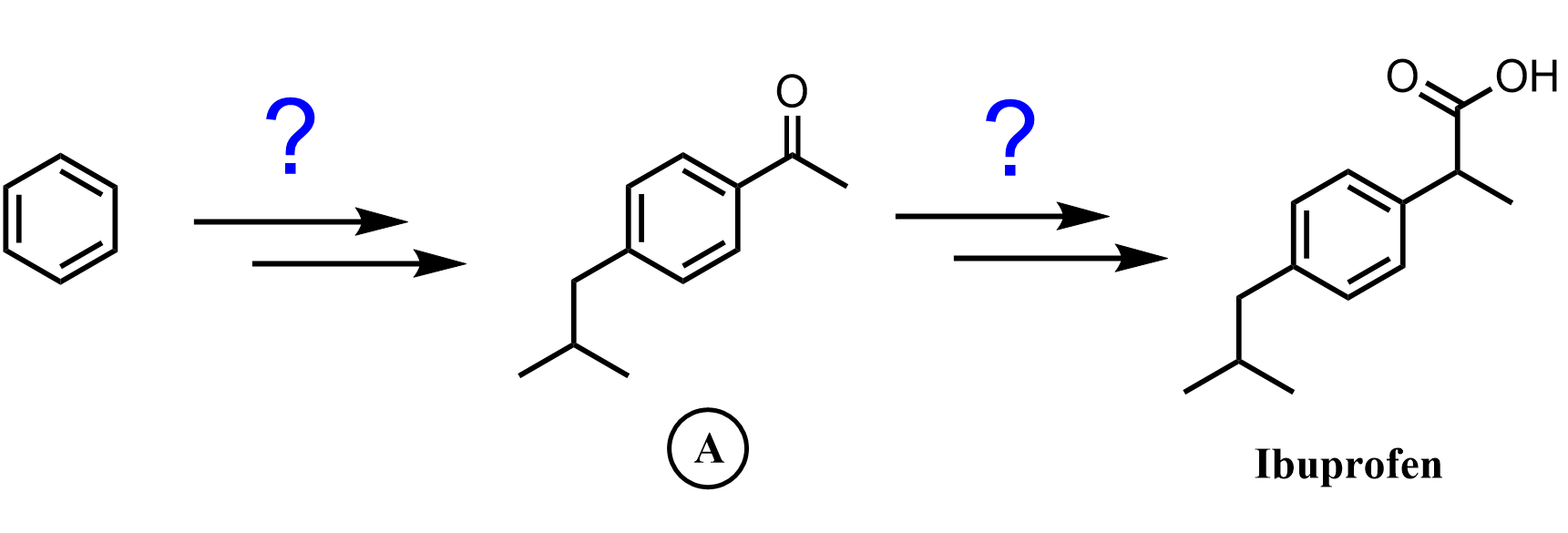
One way of obtaining the desired alkyl arene without worrying about rearrangements is the use of Friedel-Crafts acylation followed by a reduction of the carbonyl group. In Friedel-Crafts acylation, instead of alkyl halides, acetyl chlorides are used, which results in the formation of benzylic ketones.
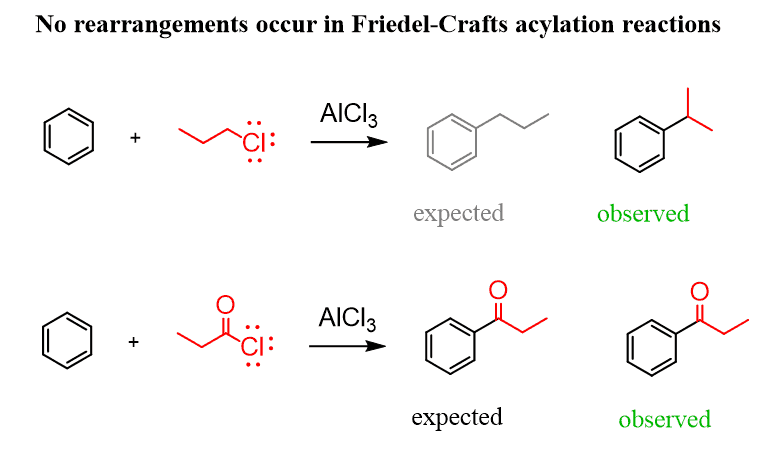
The difference here is that, unlike the alkyl carbocations formed in the Friedel-Crafts alkylation, the acyl cation does undergo a rearrangement, and it is possible to perform the synthesis as planned:
The benzylic carbonyl can be reduced by hydrogen using a metal catalyst. Notice that other ketones are reduced to alcohols, and only aryl ketones can be reduced to a methylene group by catalytic hydrogenation (H2 + Pt or Pd/C):
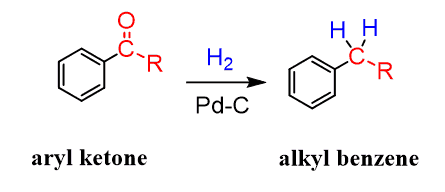
For simple alkyl groups and those not prone to undergoing a rearrangement, there is, of course, no need to opt for this route.
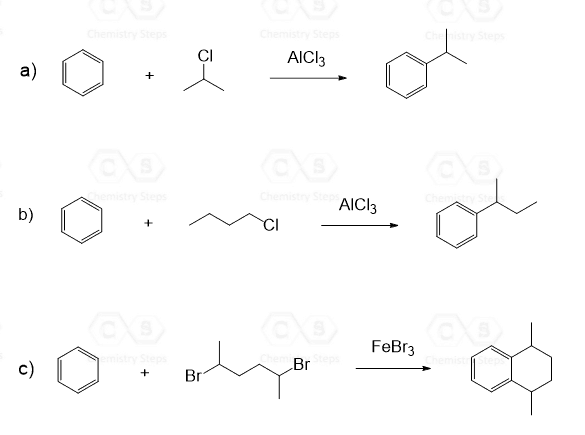
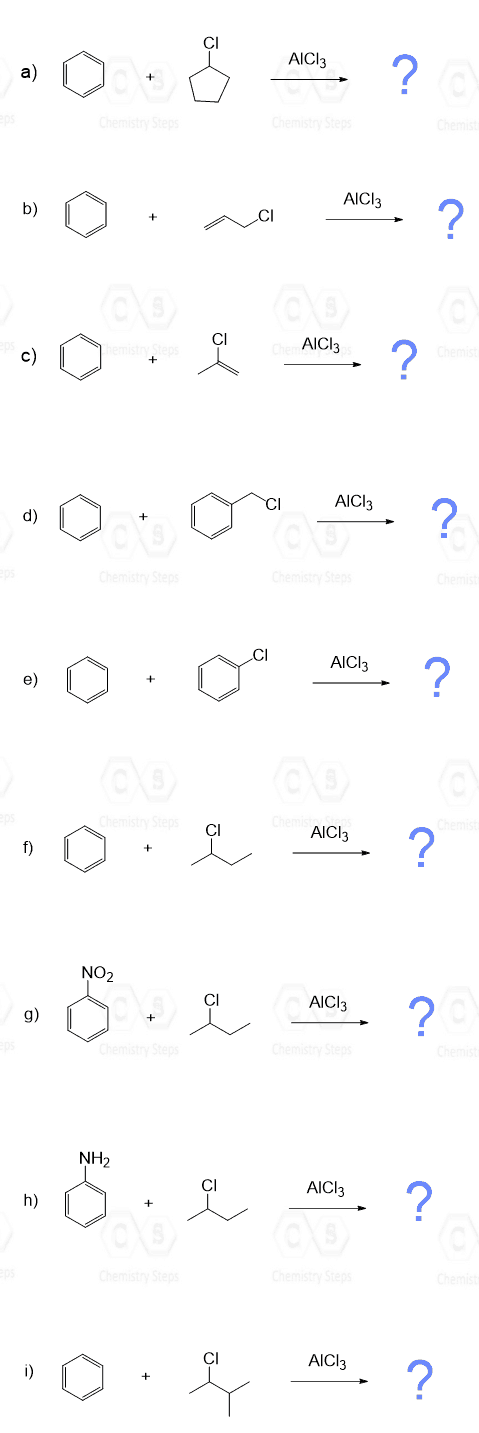
These will be, for example, tertiary alkyl halides, as we have seen, plus the methyl or ethyl alkyl halide, since they cannot rearrange into more stable carbocations:
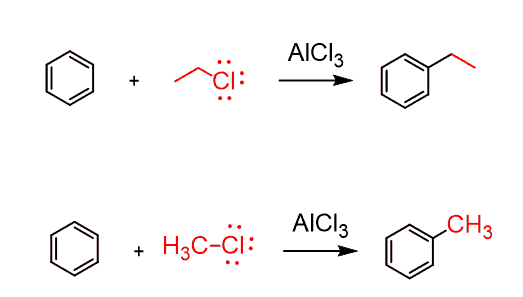
Limitations of Friedel–Crafts Alkylation
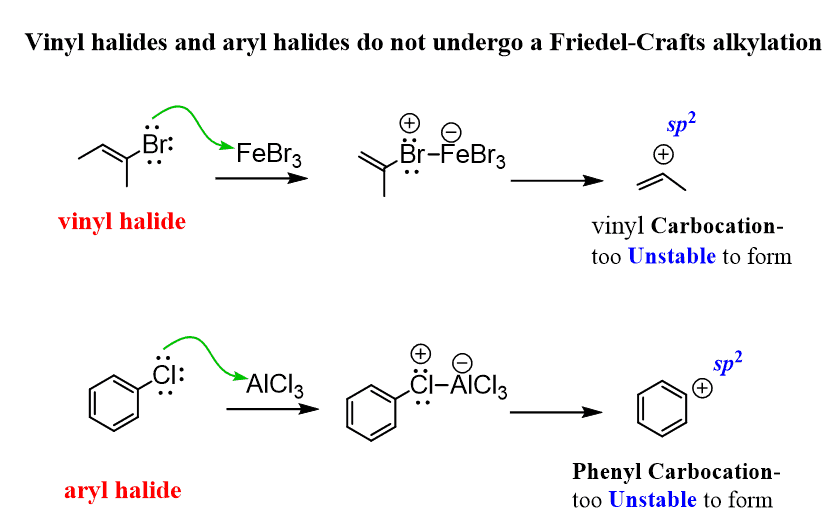
For a successful Friedel–Crafts alkylation, the halogen of the alkyl halide must be connected to an sp3 hybridized carbon atom because carbocations with the positive charge on an sp2 carbon are unstable and do not form readily. Therefore, vinyl halides and aryl halides do not undergo a Friedel–Crafts alkylation:
Friedel-Crafts alkylations and acylations are the slowest in electrophilic aromatic substitution reactions, so they do not work when a strongly deactivating group is present:

These are not the only limitations of electrophilic aromatic substitutions that you need to be aware of, so check the post “Limitations of Electrophilic Aromatic Substitution” for more details and examples on this topic.
Other functional groups in Friedel–Crafts Alkylation
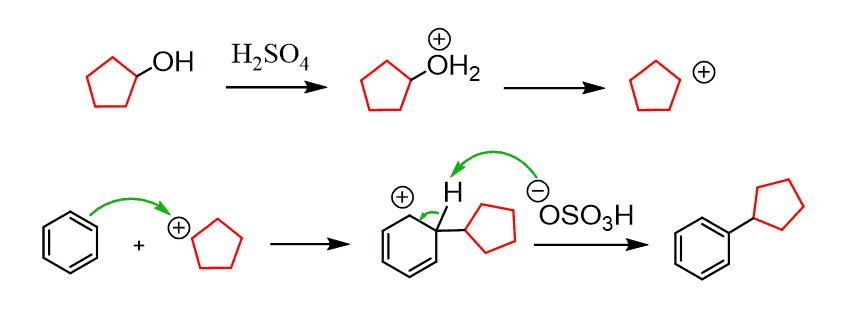
In theory, any functional group that can be converted into a strong electrophile can be used in the Friedel–Crafts Alkylation. For example, alcohols can be converted into carbocations in the presence of H2SO4, just like in E1 elimination reactions:
Alcohols can also be activated with Lewis acids such as BF3.
Alkenes are converted into carbocations in the presence of dilute acids (remember the acid-catalyzed hydration of alkenes), which serve as electrophiles in Friedel–Crafts alkylation reactions:
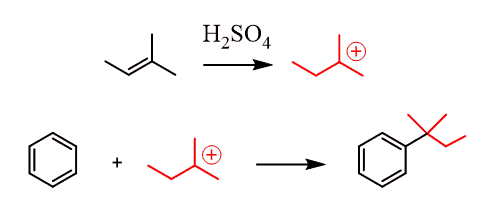
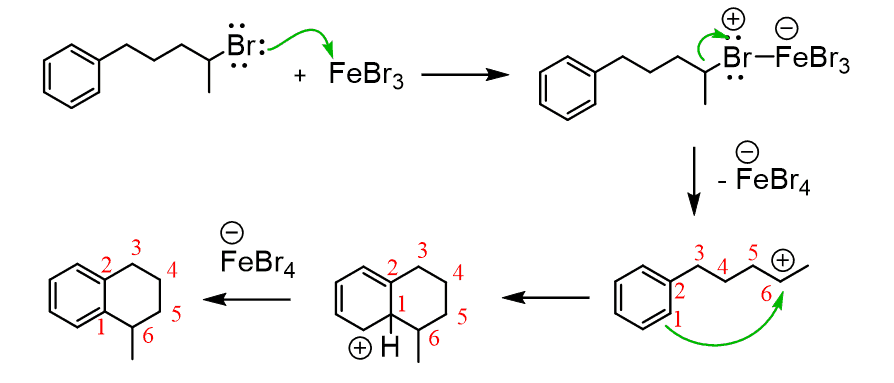
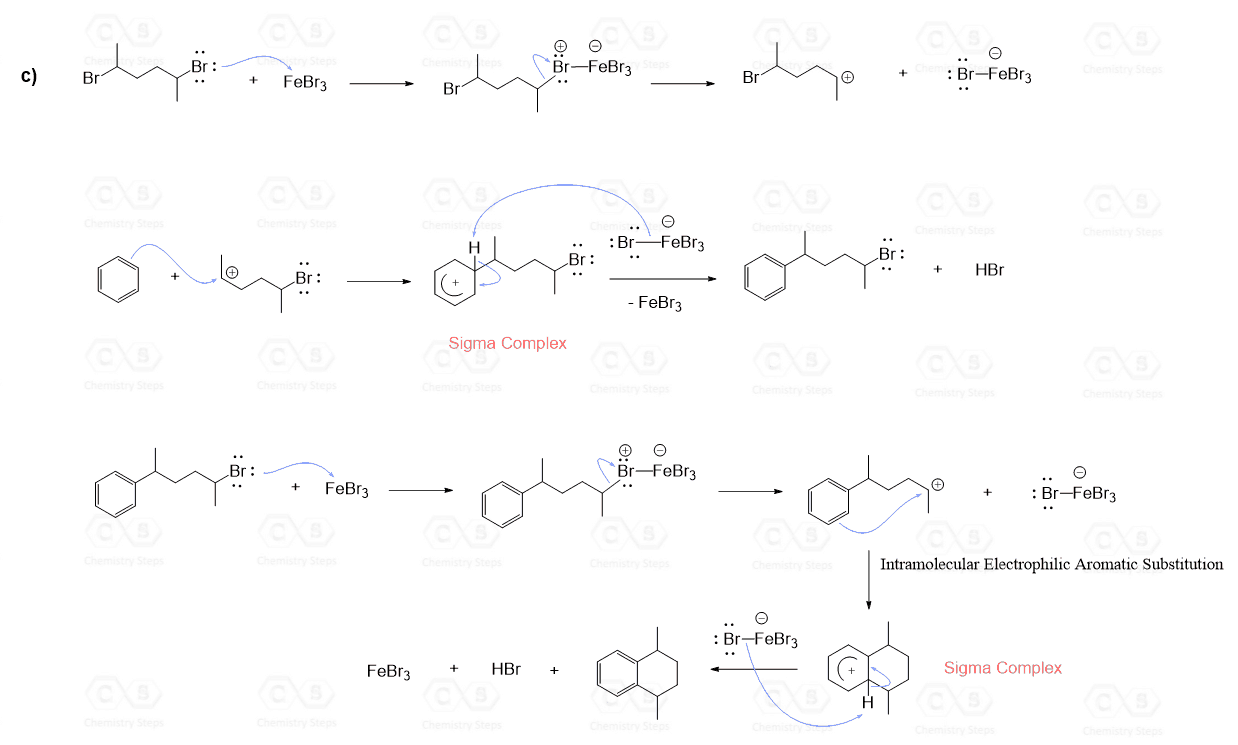
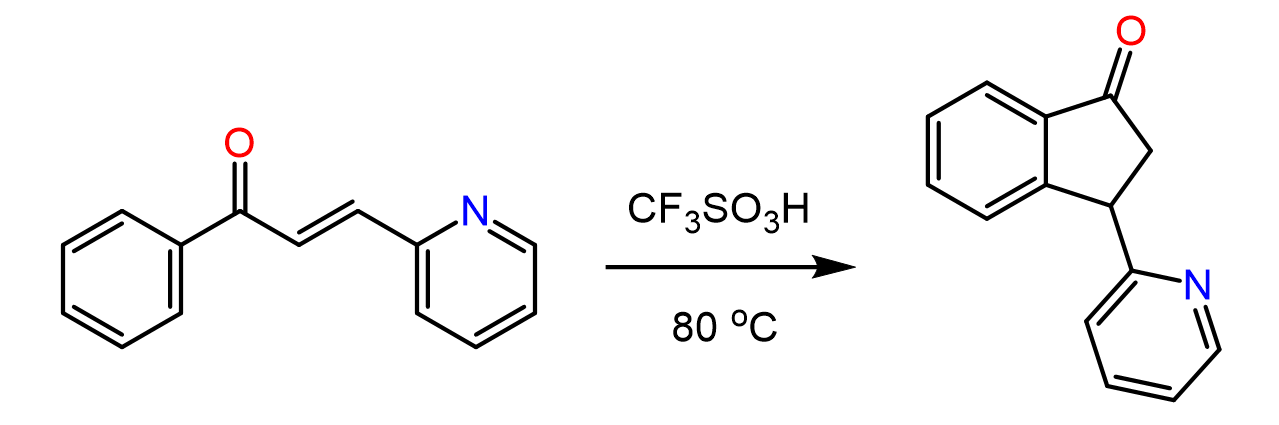
Intramolecular Friedel–Crafts Alkylation
Aromatic compounds that contain a functional group that can be converted into an electrophile can undergo an intramolecular Friedel-Crafts–Crafts alkylation:
![]()
Check this 60-question, Multiple-Choice Quiz with a 1.5-hour Video Solution covering the naming and electrophilic aromatic substitution reactions.




This is very important since I was able to solve many questions. Let’s enjoy together and make interesting questions.