In an antiperiplanar arrangement, two atoms or groups are positioned on opposite sides of the same plane, typically around a carbon-carbon single bond. This is clearly seen in the Newman projection of butane, where the lowest-energy anti conformation places the two methyl groups 180° apart, making them antiperiplanar.
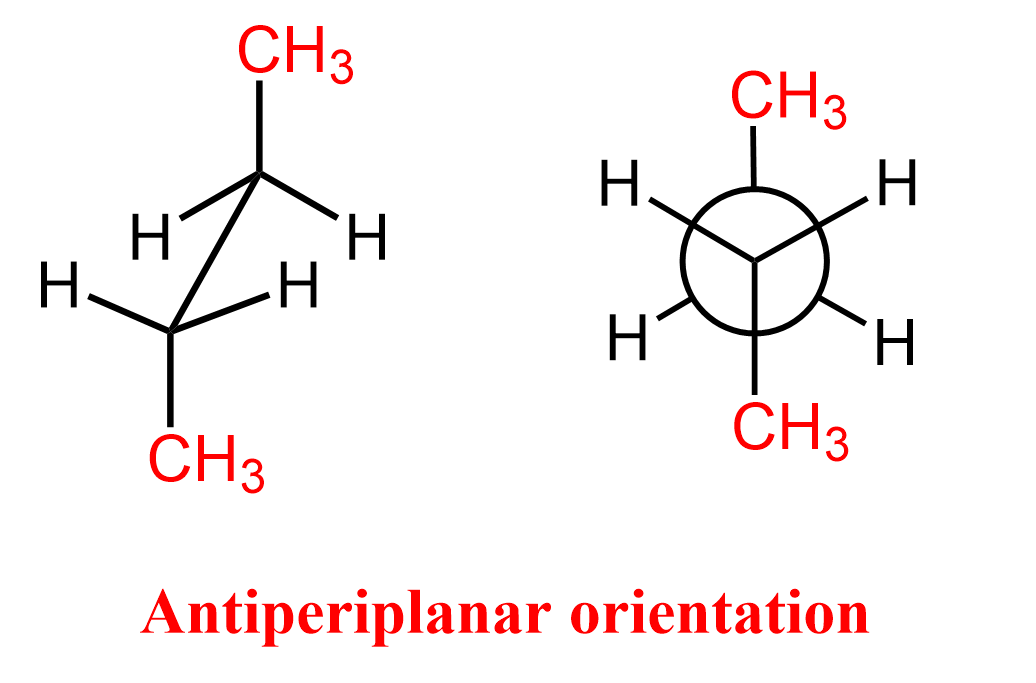
As the bond rotates, other conformations of the molecule, such as gauche, syn periplanar, and antiperiplanar, are possible. The antiperiplanar orientation is a staggered conformation where the dihedral angle between the groups on neighboring carbons is 180°.
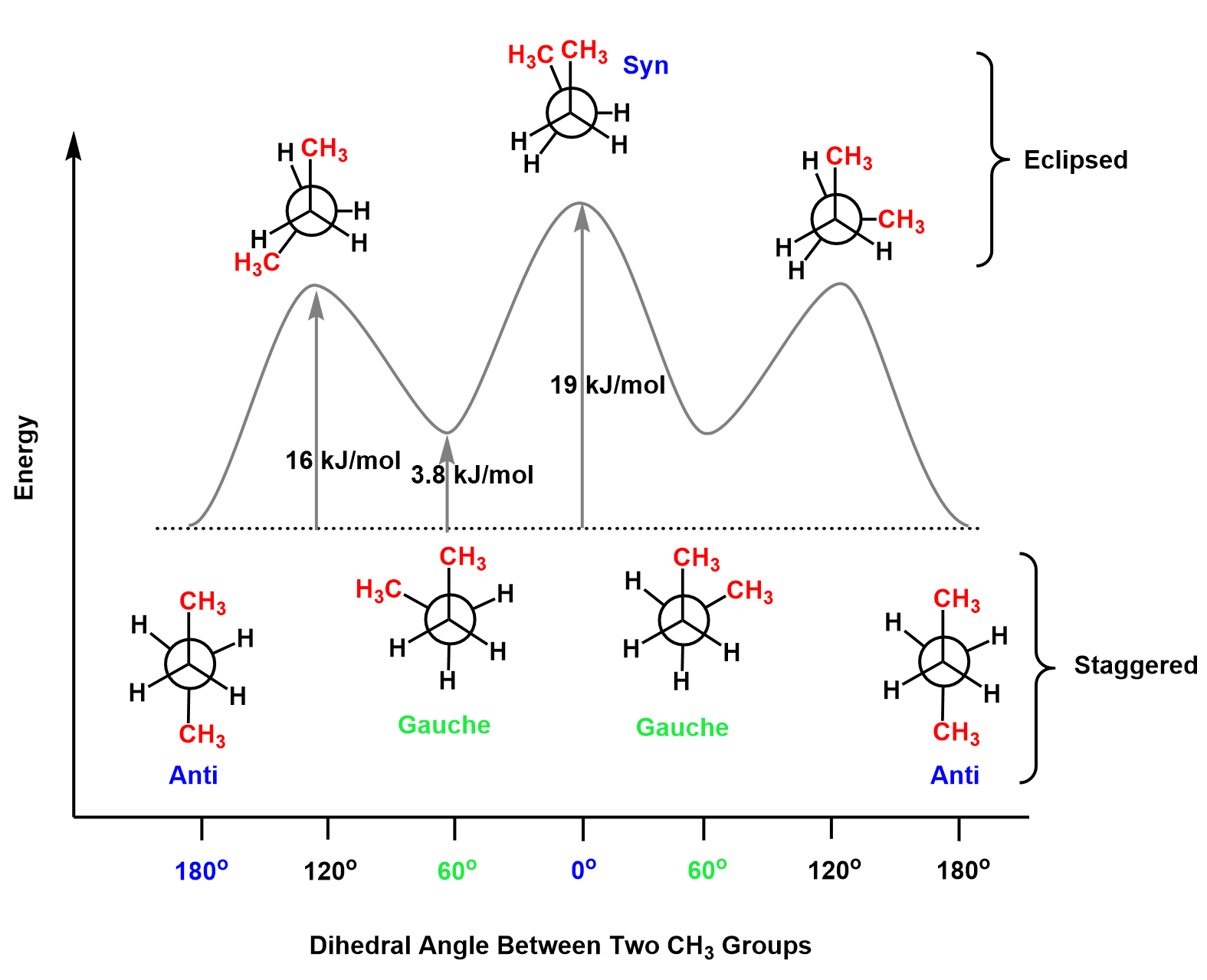
Antiperiplanar Orientation in E2 Eimination
So, what is special about the antiperiplanar arrangement that you need to know for your organic chemistry class? Well, first of all, let’s mention that the term antiperiplanar does not exclusively refer to the methyl groups in butane. We can use it for any atoms or groups that adopt this opposite-side, 180° staggered arrangement.
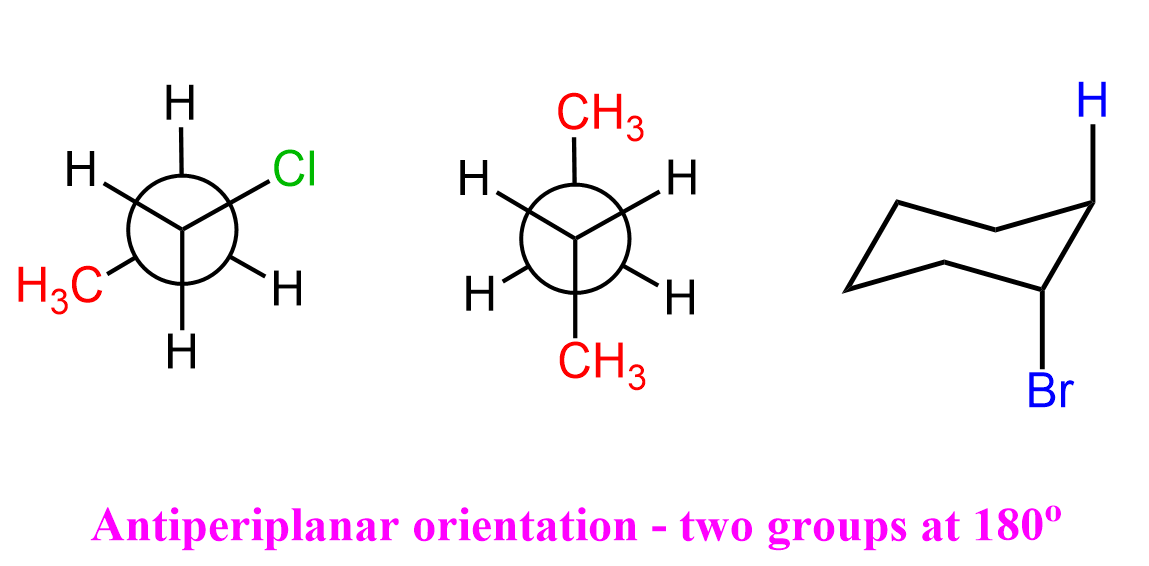
The most important example of two atoms in an antiperiplanar orientation is when a hydrogen atom and a halogen atom are positioned opposite each other. The hydrogen on the adjacent carbon is called the β-hydrogen, and you might recall from substitution and elimination reactions that it is this β-hydrogen that participates in E2 and sometimes E1 eliminations.
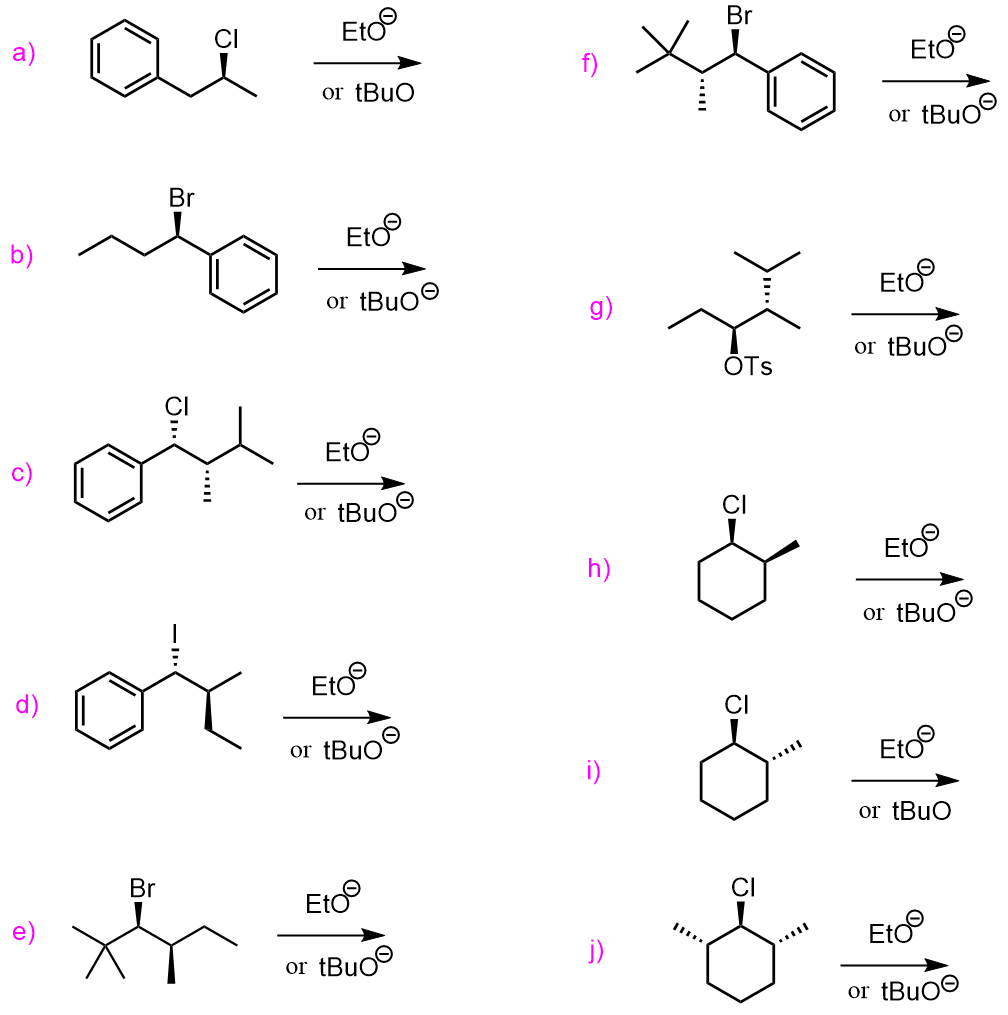
For example, the antiperiplanar alignment of the halogen and the β-hydrogen can be seen in the E2 elimination of 2-bromo-3-methylbutane with potassium tert-butoxide (t-BuOK).
In the antiperiplanar orientation, the C–H bond on the β-carbon is perfectly aligned opposite to the C–Br bond on the α-carbon in the same plane but pointing in opposite directions:
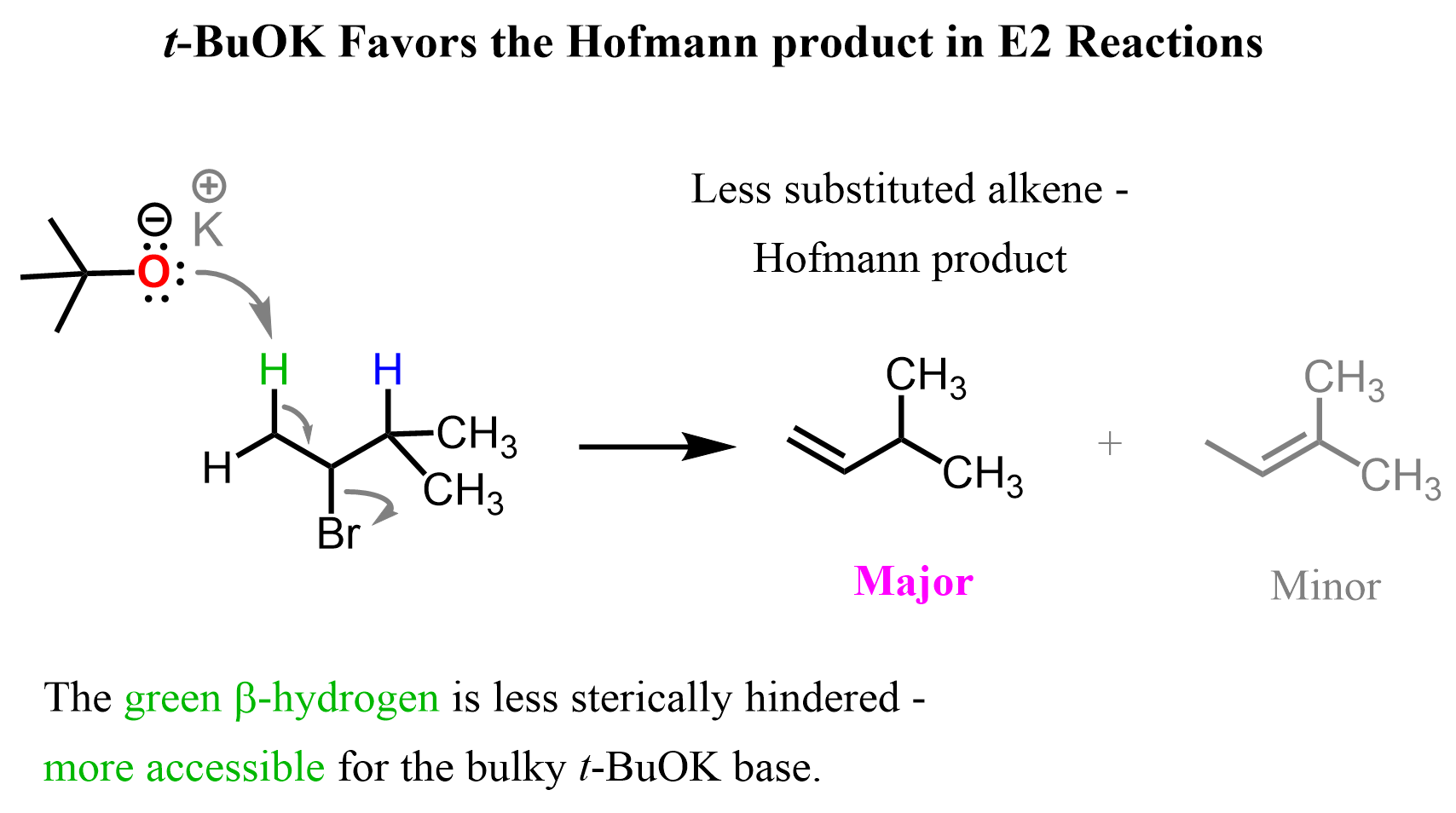
This alignment is the only one that allows the electrons from the breaking C–H bond to be donated directly into the antibonding σ* orbital of the C–Br bond. As t-BuOK abstracts the β-hydrogen, the electrons flow into the σ*(C–Br) orbital, breaking the C–Br bond and forming the double bond in a single concerted step.
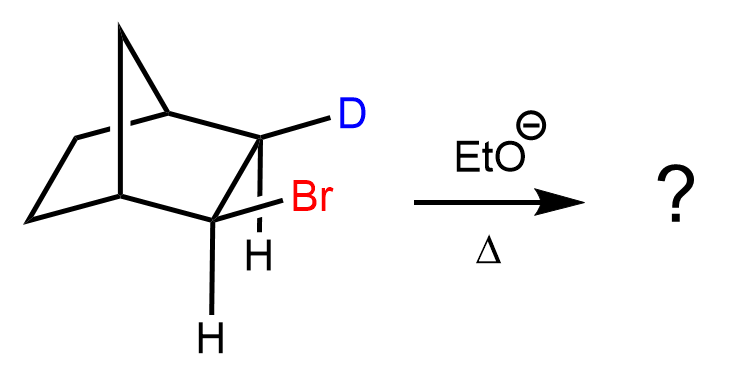
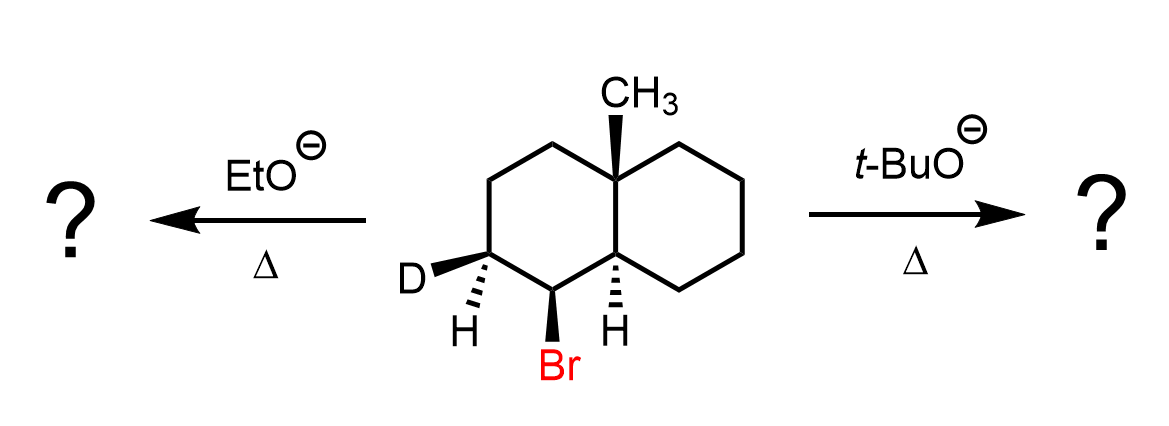
We can also observe this in the E2 elimination of cyclohexane derivatives. In the chair conformation, the antiperiplanar arrangement is perfectly illustrated because it is only possible when both the leaving group and the β-hydrogen occupy axial positions on adjacent carbons. When the leaving group is axial, the β-hydrogen on the neighboring carbon will also be axial but pointing in the opposite direction, creating the required antiperiplanar geometry. This precise alignment allows the C–H bond electrons to overlap with the σ*(C–X) orbital, enabling efficient elimination and double-bond formation.
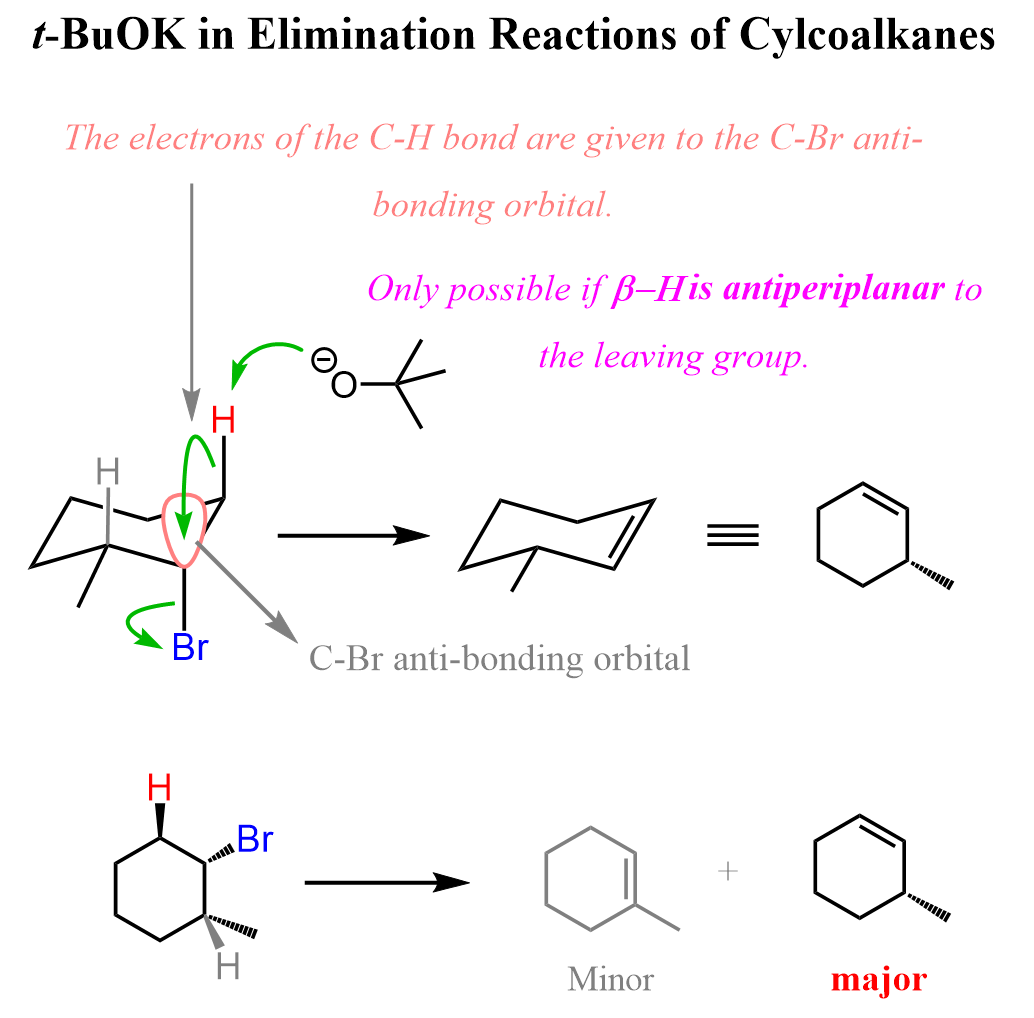
In contrast, if either substituent is equatorial, the geometry needed for orbital overlap is lost, and the E2 elimination is not possible.
Antiperiplanar Orientation and the Stereochemistry of E2 Elimination
The requirement for antiperiplanar orientation dictates the stereochemistry of E2 reactions. This is more pronounced when there is only one beta hydrogen, and the molecule has no choice but to adopt this conformation, even though it is not the most stable, and the resulting alkene is not either.
For example, the following chiral alkyl bromide has only one beta hydrogen, and because of this, when treated with a strong base such as an ethoxide ion, the Z alkene is formed despite the fact that it is less stable than the E isomer:
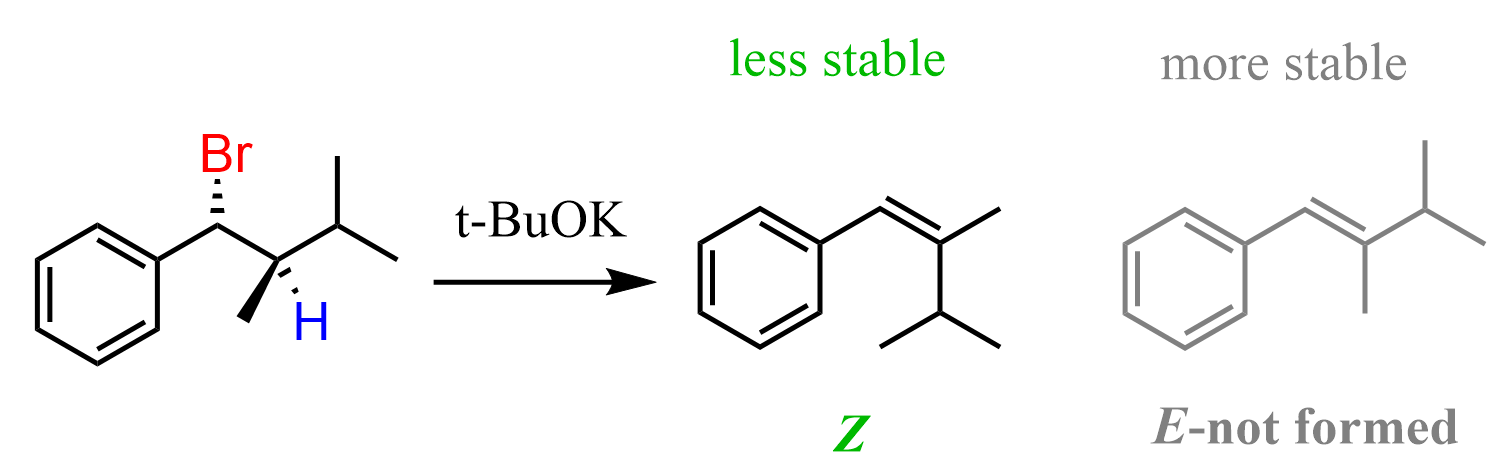
Once again, the reason for this is the requirement of antiperiplanar orientation of the beta hydrogen and the bromine. We can see this better by looking at the Newman projection of this molecule, where the Br and the beta hydrogen are at 180°.
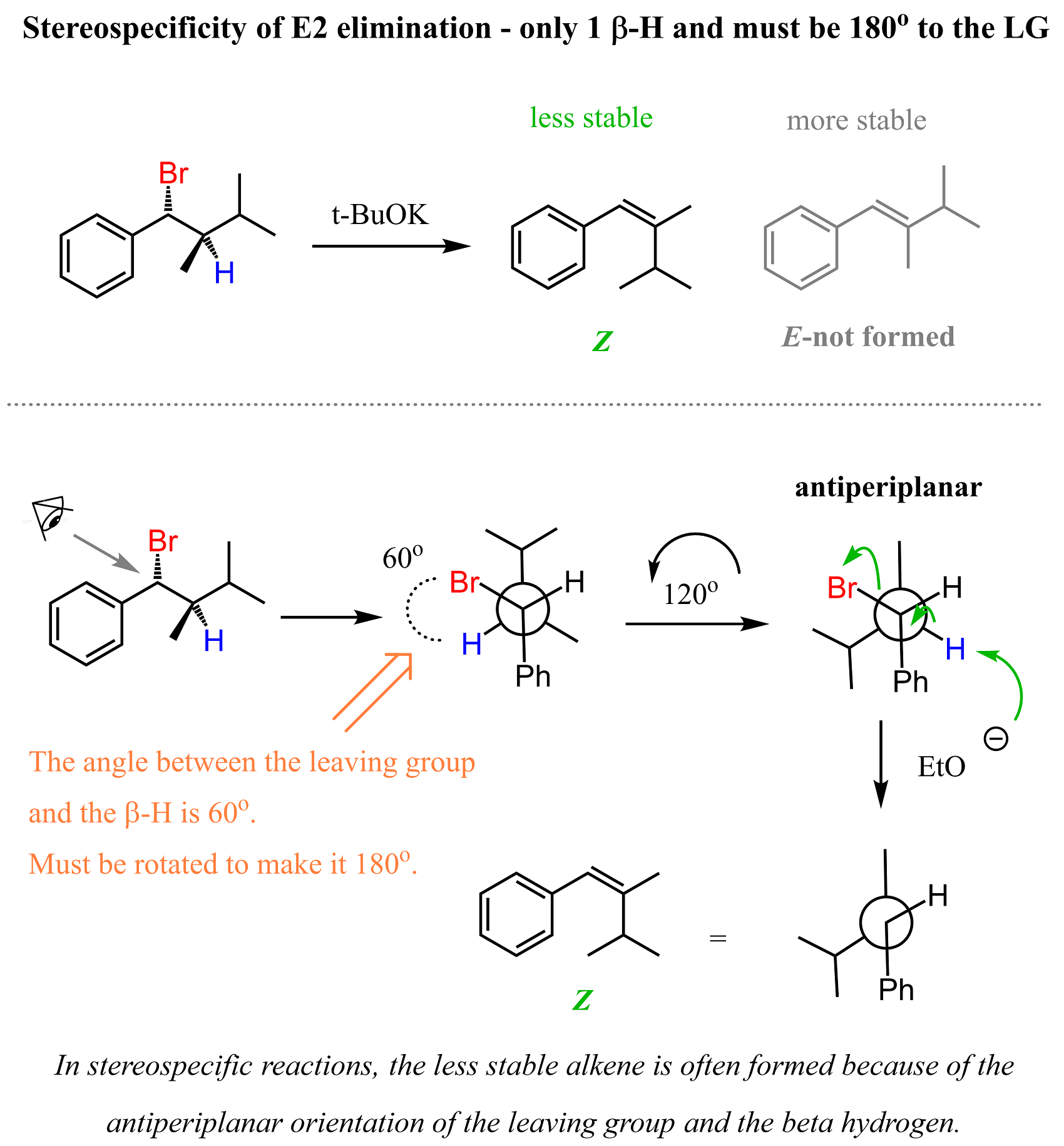
When only one beta hydrogen is present, the E2 reaction is said to be stereospecific, as the stereochemistry (configuration) of the starting material dictates the configuration of the product.
Had the alkyl halide had two beta hydrogens, the more stable E alkene would have formed because, in this case, there are two conformations where the Br and beta hydrogen can be at 180°, and the molecule adopts the more stable one.
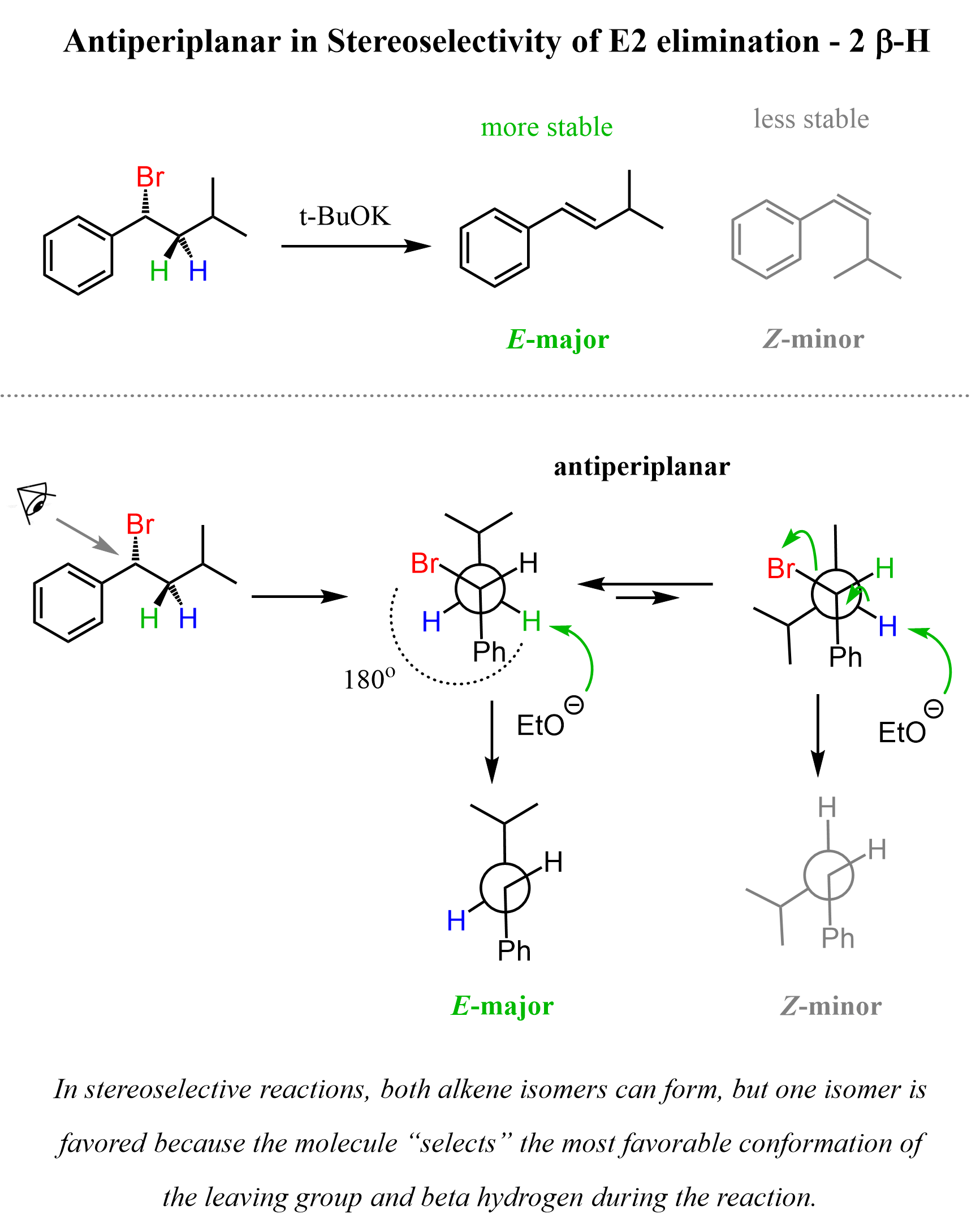
This is called a stereoselective reaction because the molecule “selects” which conformation is better to do the reaction.
Check these two posts on the Stereoselectivity and Stereospecificity of E2 reactions for more examples and practice problems on this topic.