Let’s determine the relationship between the molecules in each of the following two pairs of molecules:
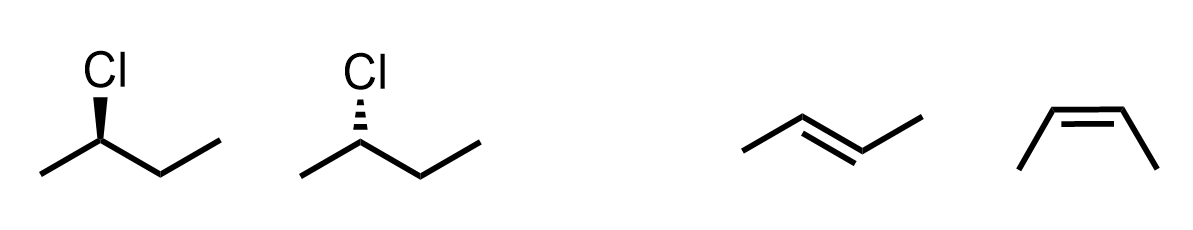
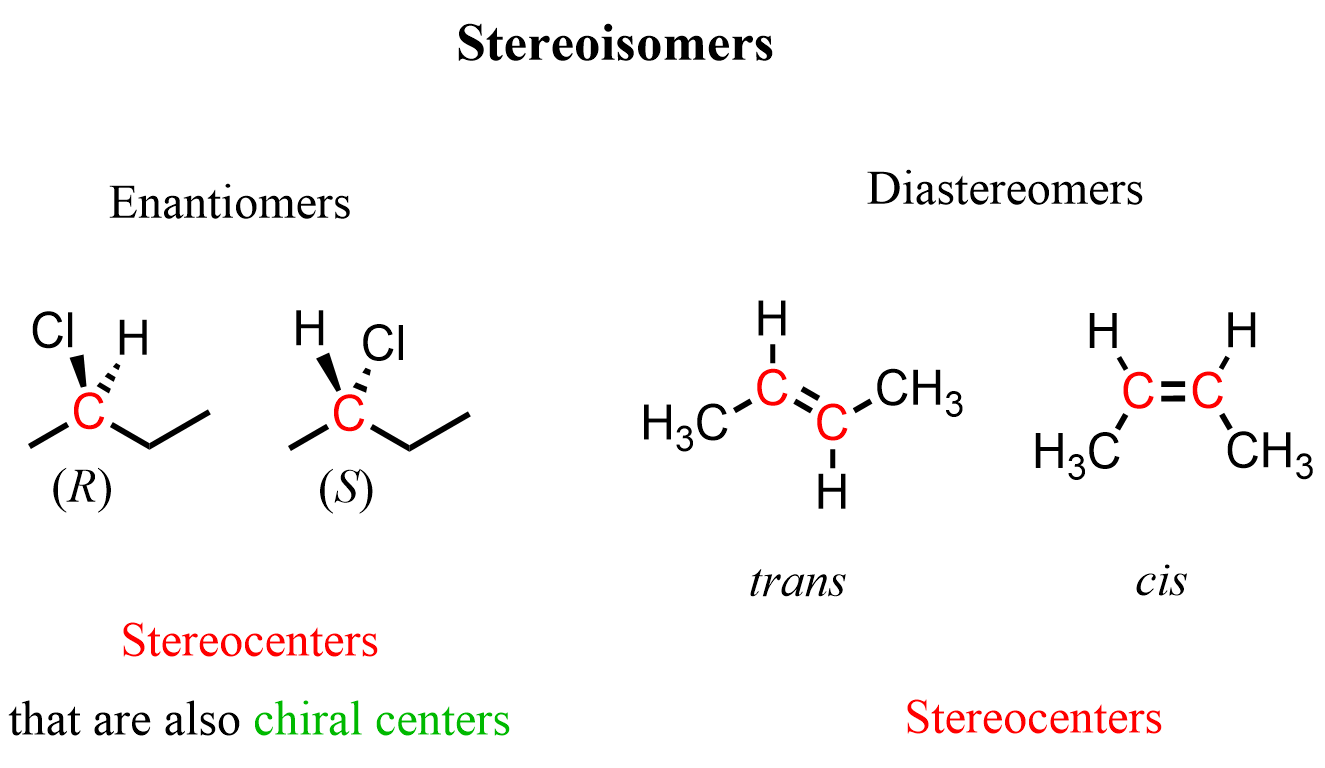
They have the same chemical formula and connectivity of atoms, which, however, have different spatial arrangements. Therefore, they are stereoisomers:
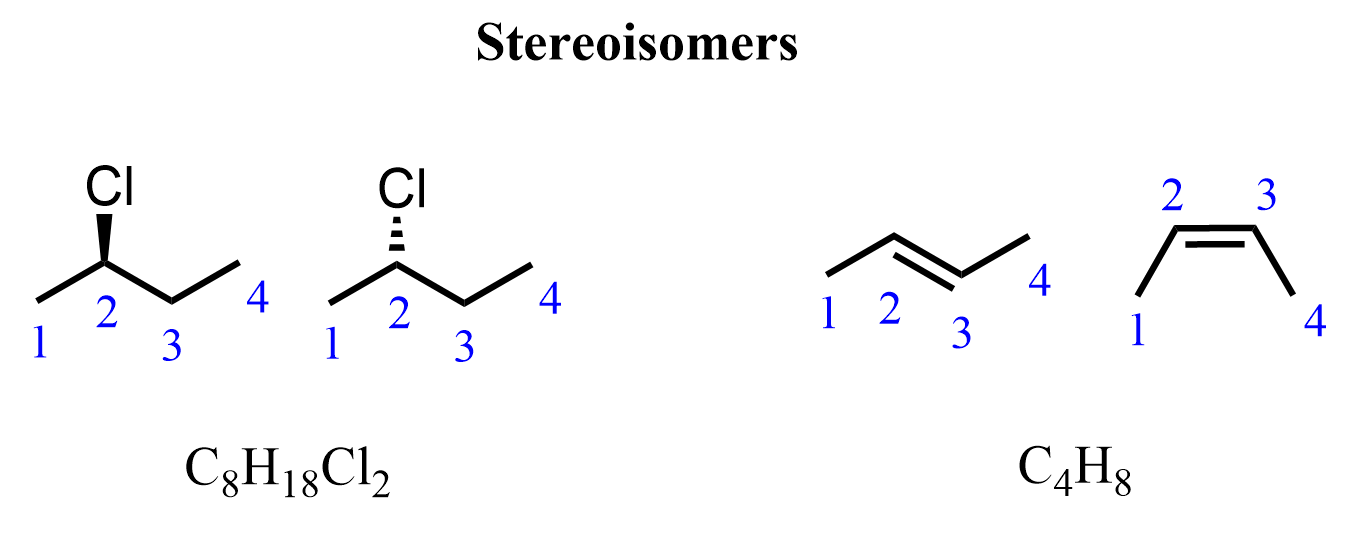
Let’s clarify that they are stereoisomers because the highlighted carbon atoms have certain configurations that cannot be changed via rotation about single bonds. The first one is a pair of stereoisomers with R and S configurations, and the second one is a pair of cis and trans stereoisomers:
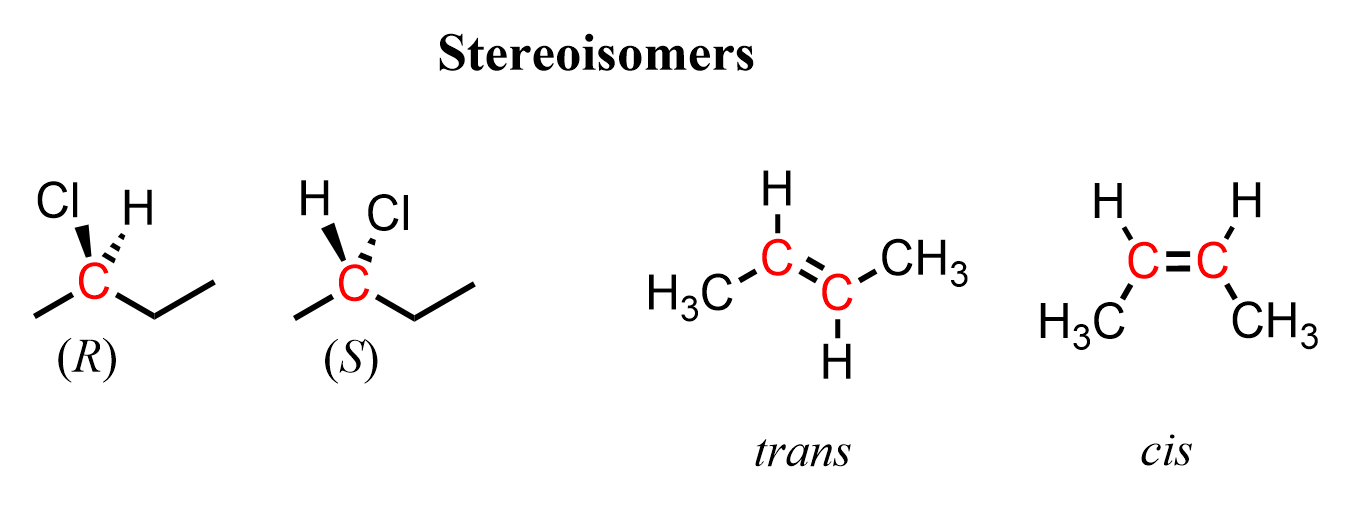
So, in both cases, the highlighted carbons are stereocenters or teratogenic centers because their different configurations result in stereoisomers. In addition to being a stereocenter, the carbon atoms in the first pair are also a chiral center or chirality, as it is connected to four different groups.
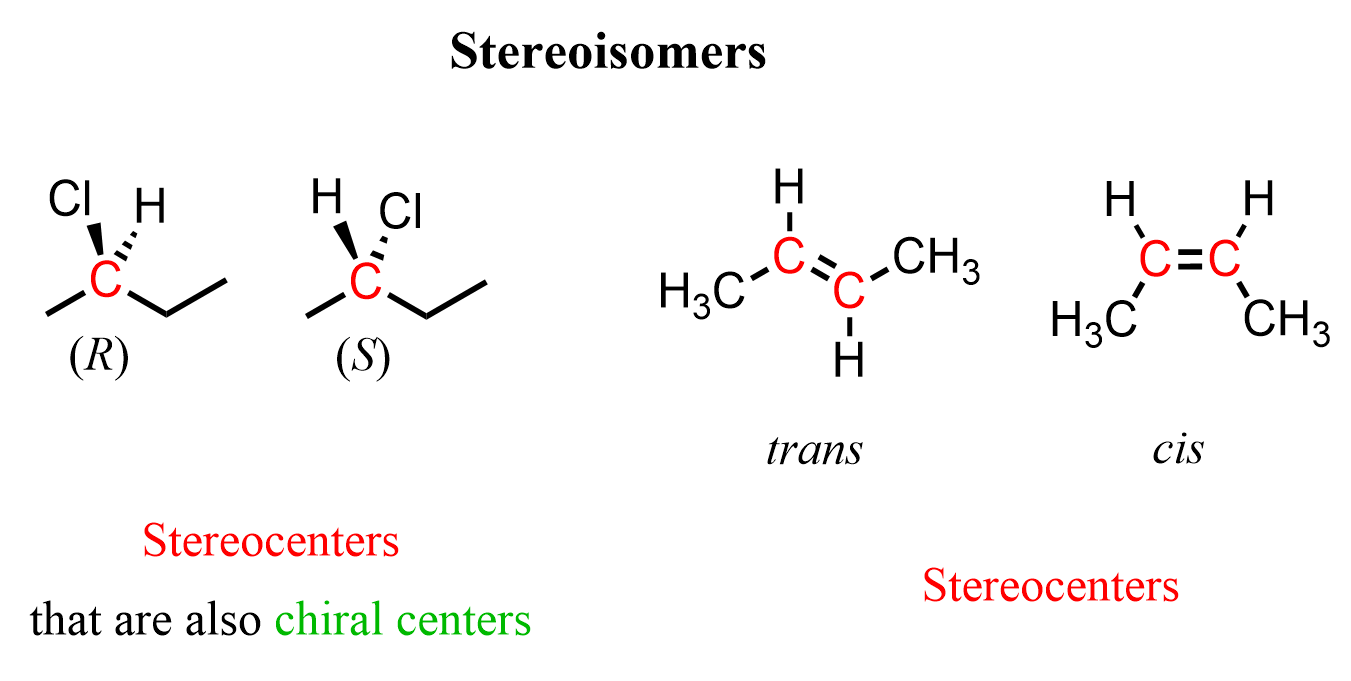
Therefore, a chiral center is a particular type of stereocenter. Any chiral center is a stereocenter, but not every stereocenter is a chiral center, as we have seen in the example of cis and trans butene.
To narrow it further, we should recognize that (R)– and (S)-2-chlorobutanes are enantiomers, and the cis and trans butanes are diastereomers. Remember, enantiomers are nonsuperimposable mirror images, whereas diastereomers are stereoisomers that are not mirror images. You can read more about the difference between enantiomers and diastereomers here.
Let’s also add a few important notes about stereocenters and chiral centers. We know that carbon is chiral only when connected to four different atoms. Similarly, a double-bonded carbon is a stereocenter only if it is connected to three different atoms. Moreover, in order to be stereogenic, both carbon atoms of the double bond must be connected to different substituents.
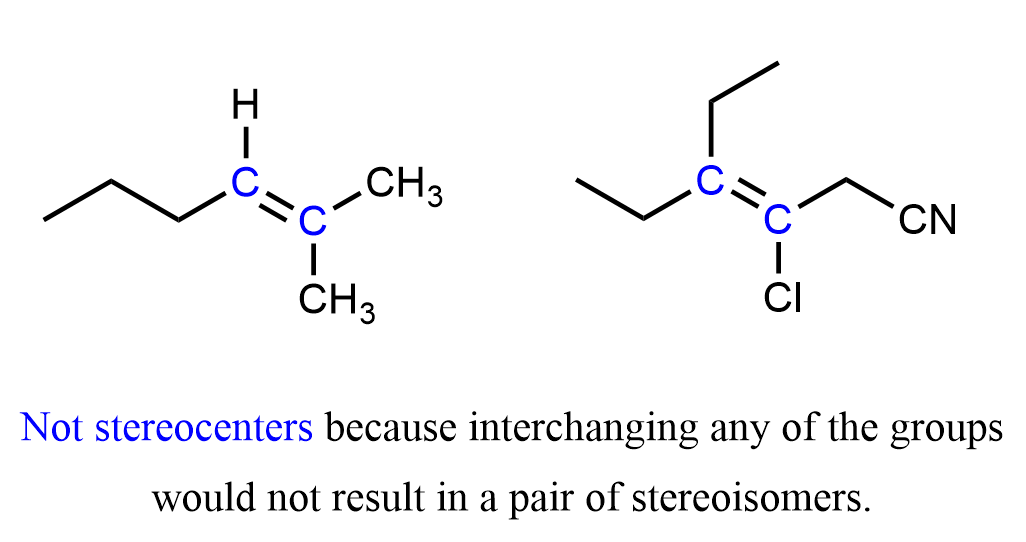
The first molecule is not stereoisomeric because one of the C=C carbons has two identical substituents (methyl groups). Similarly, in the second alkene, the two ethyl groups on the left carbon also prevent the possibility of cis-trans isomerism.
The simplest such example would be any terminal alkene because of the two hydrogens on the double bond.
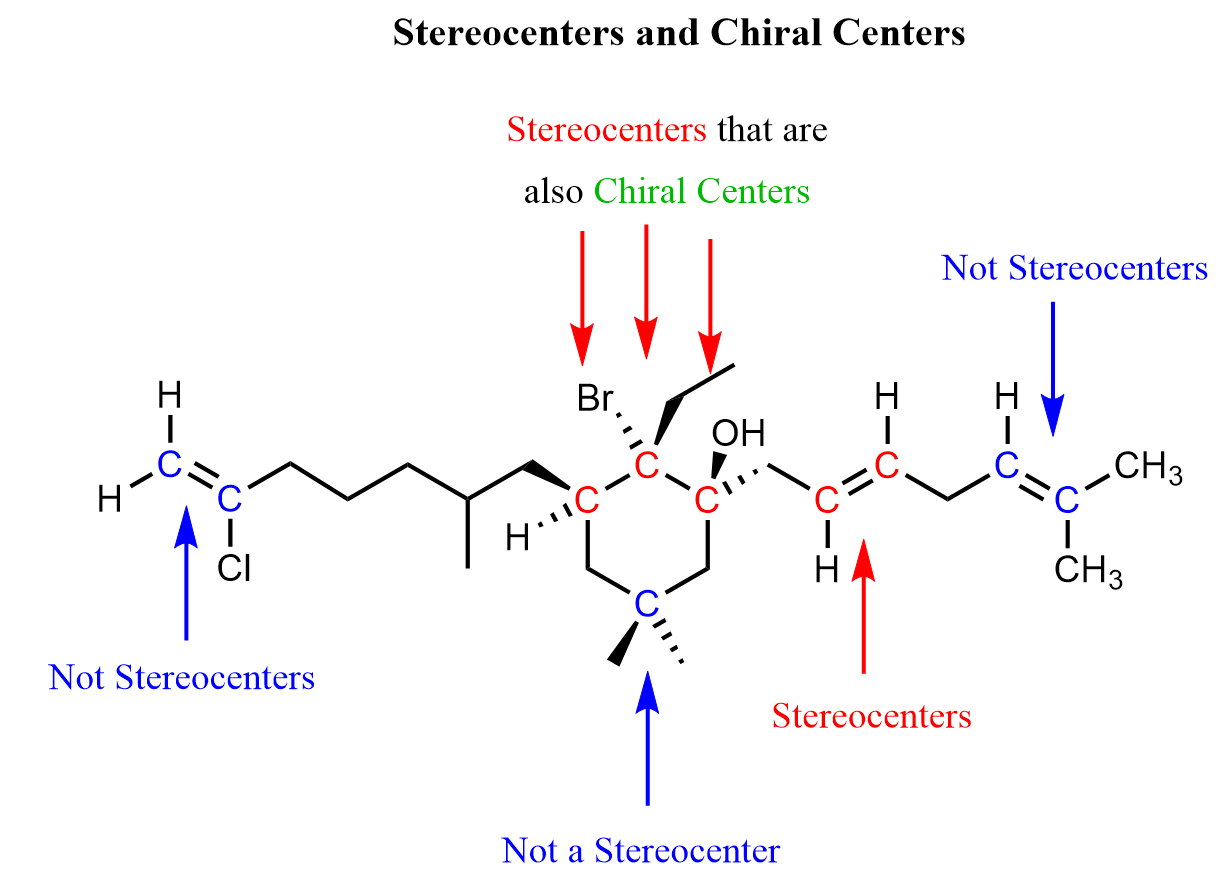
To summarize what we discussed about stereocenters and their particular type of chiral centers, here is a molecule that contains stereocenters as well as carbon atoms that may only appear to be so, but they are not because they do not have a sufficient number of different atoms connected to them:
Check Also
- How to Determine the R and S Configuration
- The R and S Configuration Practice Problems
- What is Nonsuperimposable in Organic Chemistry
- Chirality and Enantiomers
- Diastereomers-Introduction and Practice Problems
- Cis and Trans Stereoisomerism in Alkenes
- E and Z Alkene Configuration with Practice Problems
- Enantiomers vs Diastereomers
- Enantiomers Diastereomers the Same or Constitutional Isomers with Practice Problems
- Configurational Isomers
- Optical Activity
- Specific Rotation
- Racemic Mixtures
- Enantiomeric Excess (ee): Percentage of Enantiomers from Specific Rotation with Practice Problems
- Symmetry and Chirality. Meso Compounds
- Fischer Projections with Practice Problems
- R and S Configuration in the Fischer Projection
- R and S configuration on Newman projections
- R and S Configuration of Allenes
- Converting Bond-Line, Newman Projection, and Fischer Projections
- Resolution of Enantiomers: Separate Enantiomers by Converting to Diastereomers
- Stereochemistry Practice Problems Quiz
