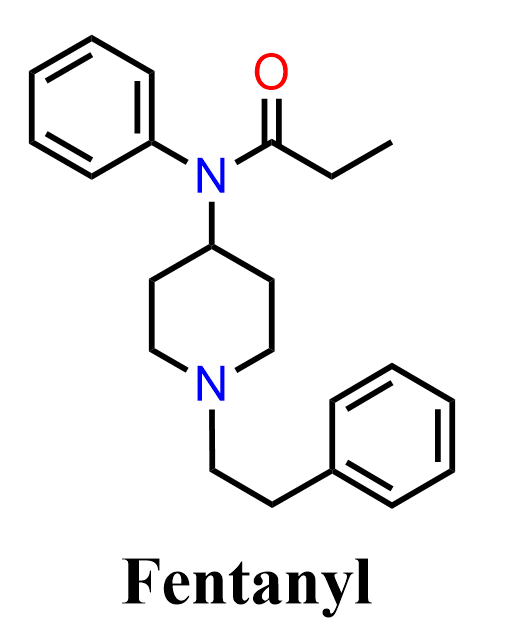
 Fentanyl is a potent synthetic opioid widely used for pain management and anesthesia. Structurally, it is a phenylpiperidine derivative, featuring a substituted aniline ring, a piperidine heterocycle, and an amide linkage connecting the key fragments. Its biological activity arises from strong agonism at opioid receptors. Due to its extremely high potency, even small dosing errors can be dangerous, and it is also widely associated with misuse and overdose in illicit settings.
Fentanyl is a potent synthetic opioid widely used for pain management and anesthesia. Structurally, it is a phenylpiperidine derivative, featuring a substituted aniline ring, a piperidine heterocycle, and an amide linkage connecting the key fragments. Its biological activity arises from strong agonism at opioid receptors. Due to its extremely high potency, even small dosing errors can be dangerous, and it is also widely associated with misuse and overdose in illicit settings.
Because of its structure, fentanyl is commonly discussed in organic chemistry in the context of aromatic substitution, amide formation, heterocycle functionalization, and C–N bond-forming reactions.
In the following practice problem, we will focus on the organic synthesis of fentanyl rather than its pharmacological properties. Specifically, we will examine how the phenylpiperidine framework can be constructed starting from simple aromatic compounds, using key transformations including amide coupling, nucleophilic substitution, and functional group interconversions.
As with all examples in this section, this discussion is presented solely for educational purposes and is not intended to represent a practical or real-world method of preparation.
Practice
Add the missing intermediates and reagents in the following synthesis of Fentanyl.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to the answers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps, and the powerful set of Organic Chemistry 1 and 2 Summary Study Guides.
