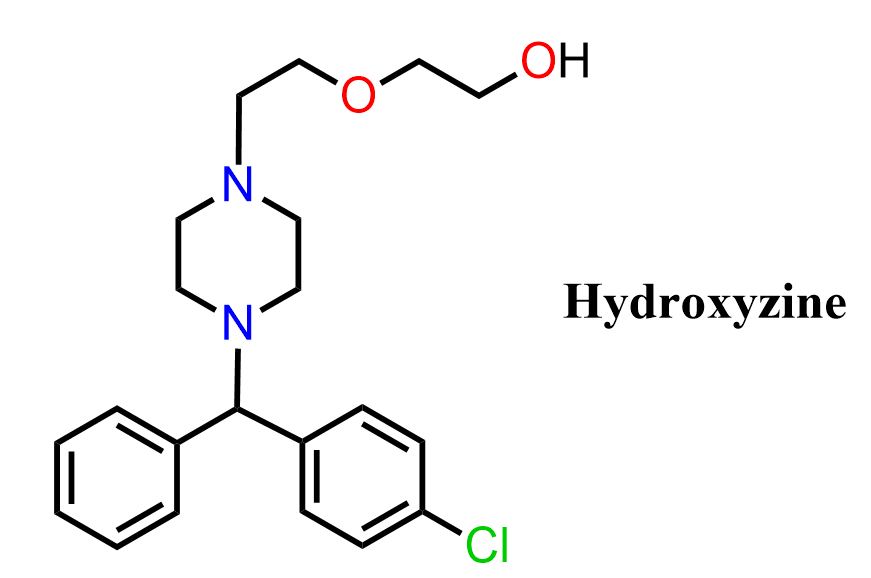
 Hydroxyzine is a first-generation antihistamine widely used in the treatment of anxiety, nausea, and allergic conditions. It belongs to a class of drugs characterized by a piperazine ring connected to a substituted phenyl group and a hydroxyethyl side chain. Hydroxyzine is classified as a piperazine derivative and displays strong antagonism at H1 histamine receptors, contributing to its sedative and anti-allergic effects.
Hydroxyzine is a first-generation antihistamine widely used in the treatment of anxiety, nausea, and allergic conditions. It belongs to a class of drugs characterized by a piperazine ring connected to a substituted phenyl group and a hydroxyethyl side chain. Hydroxyzine is classified as a piperazine derivative and displays strong antagonism at H1 histamine receptors, contributing to its sedative and anti-allergic effects.
The mechanism of action involves competitive inhibition of histamine at H1 receptors, reducing the effects of histamine in smooth muscle, endothelium, and the central nervous system. Additional interactions with serotonin and adrenergic receptors may contribute to its anxiolytic properties. Structurally, hydroxyzine contains a piperazine ring, a substituted phenyl group, and a hydroxyethyl side chain, making it a useful example for exploring functional group transformations in multistep synthesis.
In this practice problem, we will discuss how to synthesize hydroxyzine starting from benzene, illustrating the application of electrophilic aromatic substitution, nucleophilic substitution, and Friedel-Crafts alkylation.
As with all examples in this section, this discussion is presented solely for educational purposes and is not intended to represent a practical or real-world method of preparation.
Practice
Identify the reagents and intermediates in the following synthesis of Hydroxyzine.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to the answers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps, and the powerful set of Organic Chemistry 1 and 2 Summary Study Guides.
