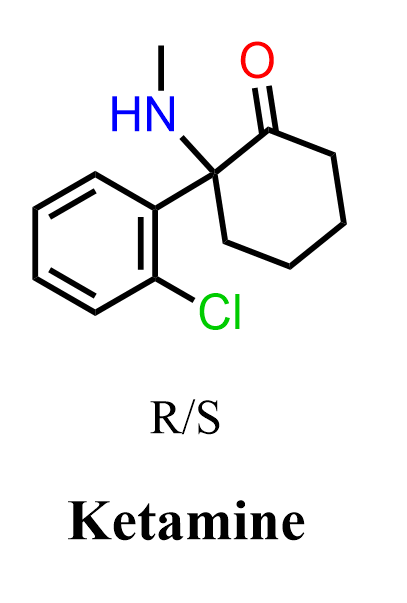
 Ketamine is a dissociative anesthetic widely used in anesthesia and, more recently, in the treatment of depression. It belongs to a class of compounds characterized by an arylcyclohexanone framework, consisting of a substituted phenyl ring attached to a cyclohexanone ring bearing an amino group. Ketamine is classified as an NMDA receptor antagonist, which is responsible for its anesthetic and dissociative effects. It is also used recreationally for its hallucinogenic and dissociative properties, which arise from the same mechanism of action.
Ketamine is a dissociative anesthetic widely used in anesthesia and, more recently, in the treatment of depression. It belongs to a class of compounds characterized by an arylcyclohexanone framework, consisting of a substituted phenyl ring attached to a cyclohexanone ring bearing an amino group. Ketamine is classified as an NMDA receptor antagonist, which is responsible for its anesthetic and dissociative effects. It is also used recreationally for its hallucinogenic and dissociative properties, which arise from the same mechanism of action.
The mechanism of action involves noncompetitive inhibition of NMDA receptors, reducing excitatory neurotransmission in the central nervous system. Structurally, ketamine contains a ketone functional group, a secondary amine, and an aromatic ring, making it a useful example for studying the reactivity of carbonyl compounds and amines. It is typically used as a racemic mixture, although the S-enantiomer shows higher affinity for the NMDA receptor.
In this practice problem, we will discuss the synthesis and mechanism of ketamine, focusing on key transformations of carbonyl compounds, including Grignard reactions, imine formation, and ring expansion rearrangements.
As with all examples in this section, this discussion is presented solely for educational purposes and is not intended to represent a practical or real-world method of preparation.
Practice
Identify the reagents and intermediates in the following synthesis of Ketamine, and draw a plausible mechanism for the last two steps of the conversion.
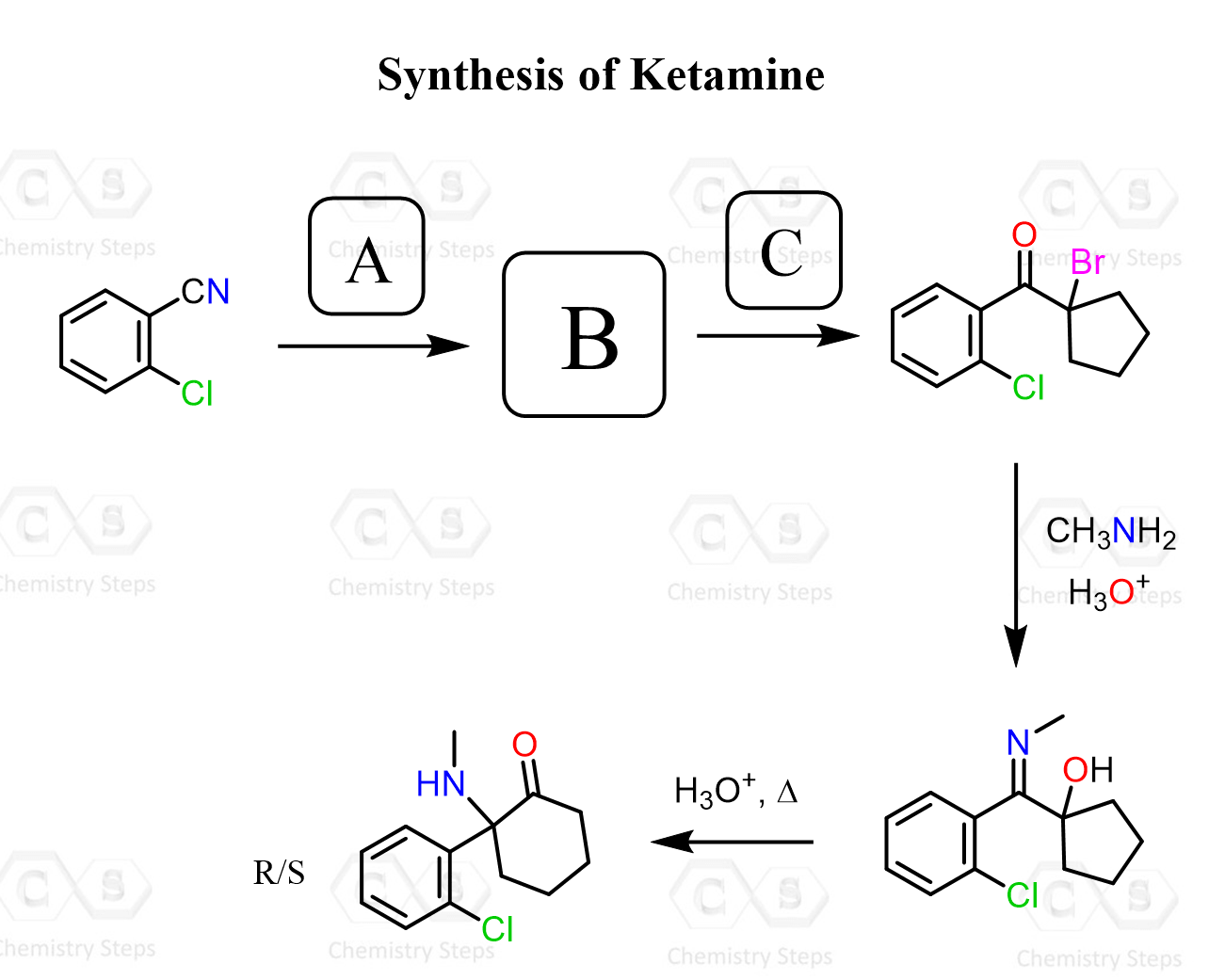
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to the answers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps, and the powerful set of Organic Chemistry 1 and 2 Summary Study Guides.
