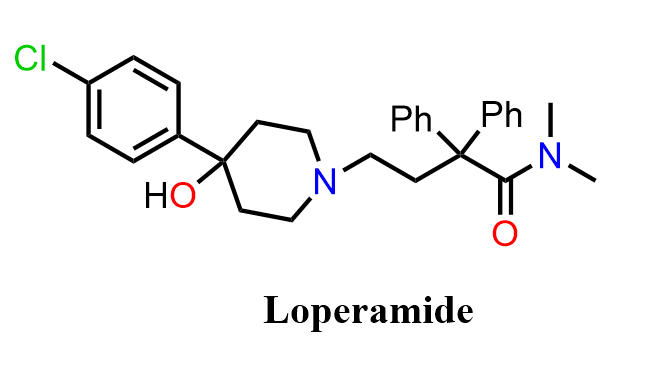
 Loperamide is an antidiarrheal medication best known under the brand name Imodium. It works by activating μ-opioid receptors in the intestinal tract, which slows down gut movement and reduces the frequency of bowel movements. Unlike other opioids, Loperamide does not cause central nervous system effects because it does not easily cross the blood–brain barrier, making it effective for treating diarrhea without producing typical opioid side effects.
Loperamide is an antidiarrheal medication best known under the brand name Imodium. It works by activating μ-opioid receptors in the intestinal tract, which slows down gut movement and reduces the frequency of bowel movements. Unlike other opioids, Loperamide does not cause central nervous system effects because it does not easily cross the blood–brain barrier, making it effective for treating diarrhea without producing typical opioid side effects.
From a synthetic standpoint, Loperamide is an excellent multi-chapter exercise in organic chemistry. Its preparation incorporates several key transformations, including Electrophilic Aromatic Substitution (EAS) to build and functionalize the aromatic core, unique SN2 substitutions for installing the tertiary amine side chain, a Grignard reaction, and ring-opening reactions of epoxides. The route also features Fischer esterification and nucleophilic acyl substitution steps—fundamental reactions covered in the chapter on carboxylic acid derivatives. Together, these transformations make the synthesis of Loperamide a rich example of how diverse organic reactions converge in the preparation of a clinically important drug.
Practice
Add the missing reagents and intermediates in the synthesis of Loperamide, and draw plausible mechanisms for the reaction with the epoxide and the final step of the synthesis, which involves intermediate K.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to the answers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps, and the powerful set of Organic Chemistry 1 and 2 Summary Study Guides.
