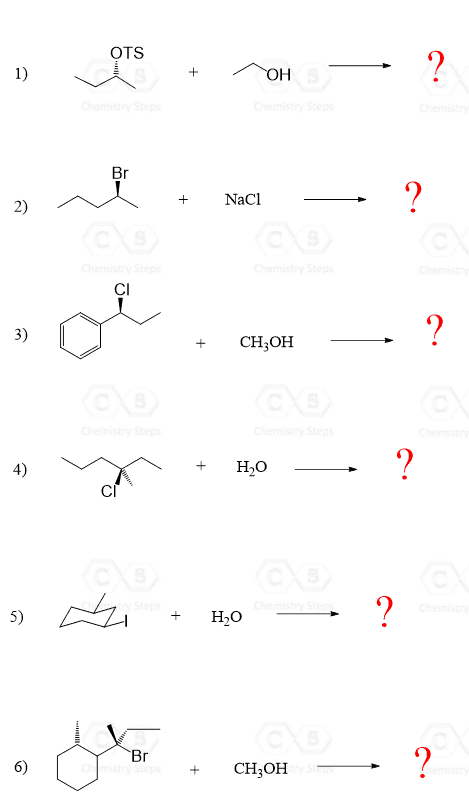
Alkyl halides react with water by SN1 mechanism forming alcohols. Let’s put together some examples of these hydrolysis reactions of alkyl halides and address some important concepts such as the mechanisms, stereochemistry, the relationship of products, and different types of rearrangements:

Let’s mention one thing that is common in most (and all) of the reactions between alkyl halides and water: Water is a poor nucleophile and a weak base; therefore, it only does SN1 or E1 reactions on alkyl halides. The only exception would be primary halides, which cannot undergo SN1 reaction as the intermediate primary carbocation is too unstable.
Enantiomers in SN1 Reactions
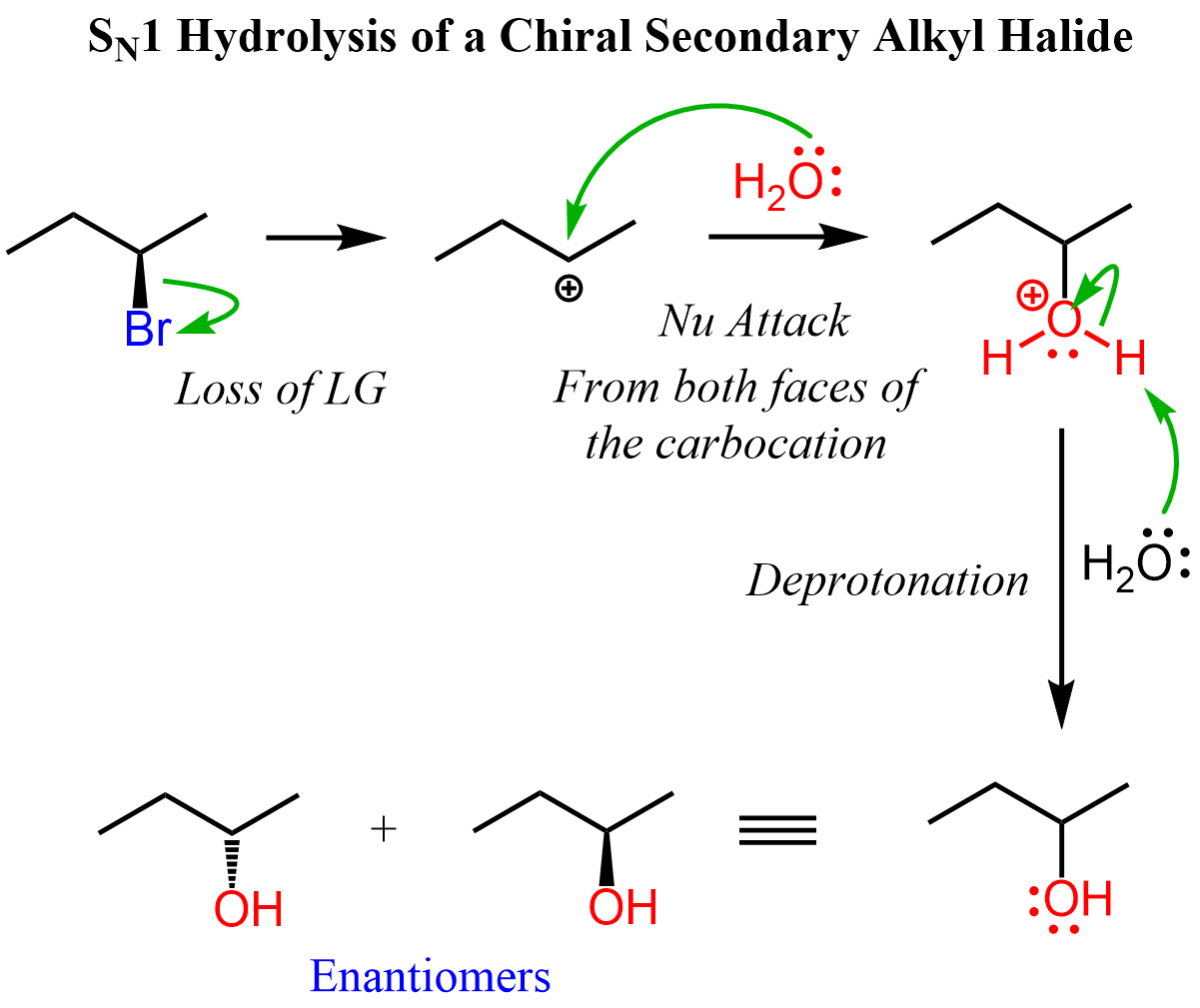
If the leaving group in the substrate is part of a chirality center, then SN1 substitution will give a mixture of enantiomers. The hydrolysis of (R)-2-bromobutane in reaction 1 is such an example. In the first step, a secondary butyl carbocation is formed which is attacked from both faces of the planar carbon giving a racemic mixture of enantiomers:
Diastereomers in SN1 Reactions
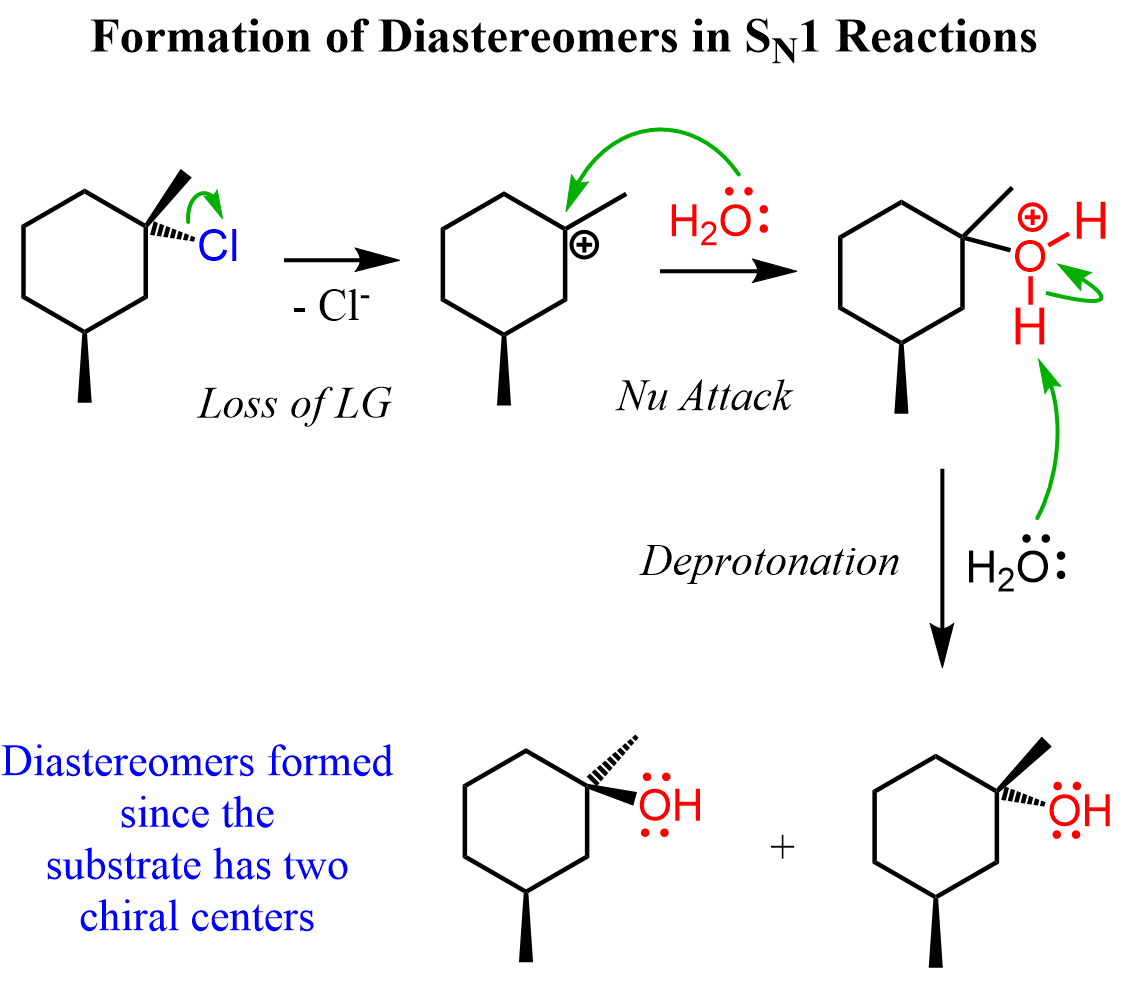
Reactions 2, 3, 4, and 5 are examples of SN1 reactions where the product is formed as a pair of diastereomers. Generally, this happens if the substrate has two chiral centers. One of them is racemized during the substitution, while the other stays intact, thus a mixture of diastereomers is obtained (reactions 2, 4, and 5).
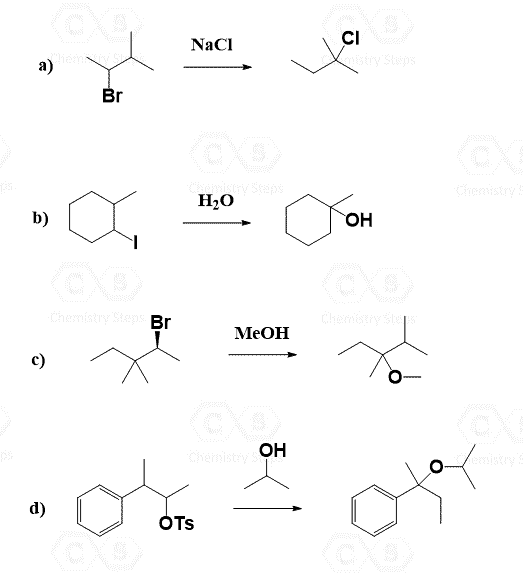
Let’s draw the mechanism of reaction 2. As any other SN1 substitution, it starts with the loss of the leaving group forming a carbocation, and in the second step the nucleophilic attack occurs:
Once you finish drawing the mechanism of reactions 4 and 5, come back so we can discuss what happens in reactions 3 and 4.
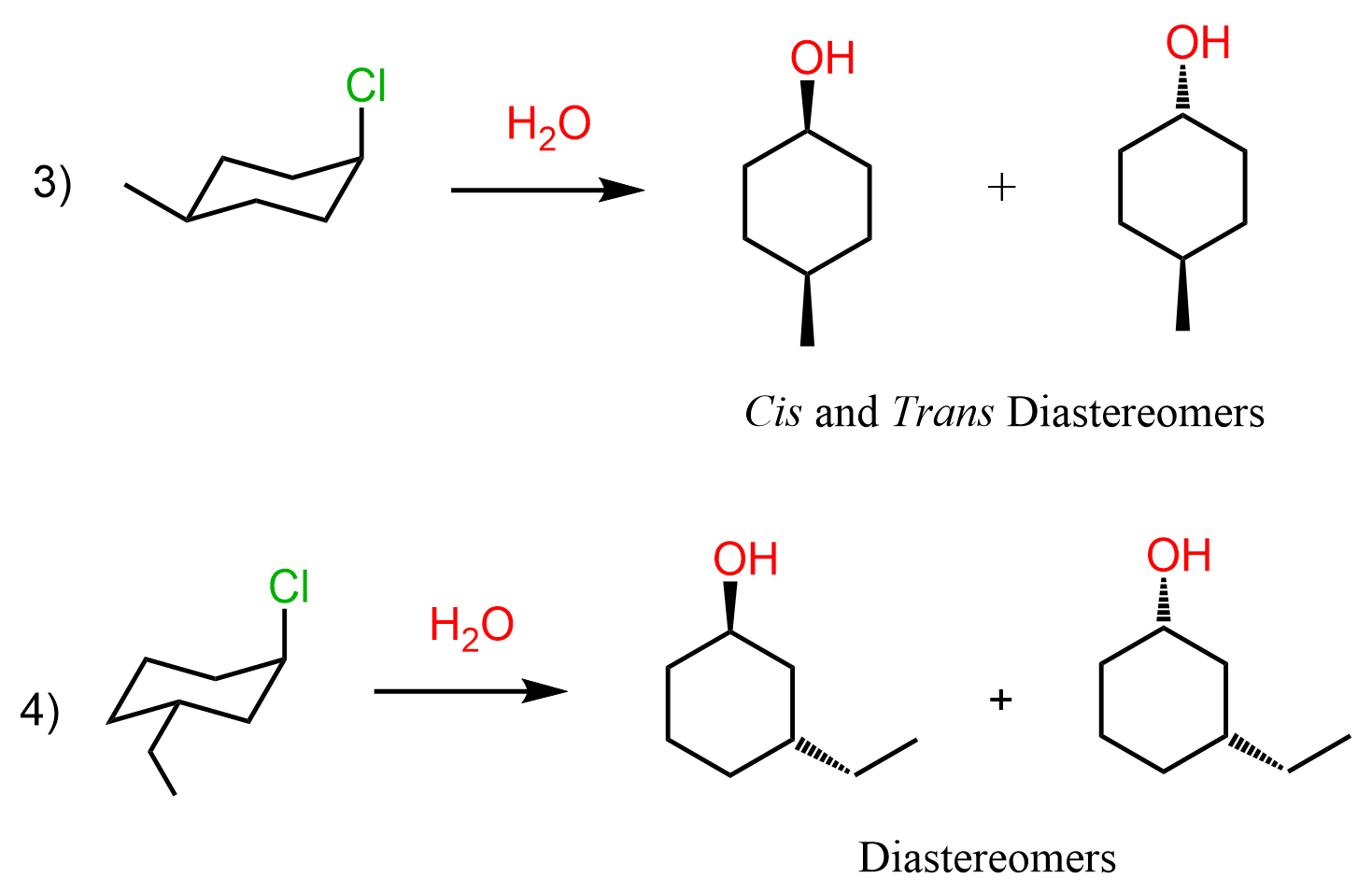
The main difference here is that the first molecule is achiral as it has a plane of symmetry. However, we cannot ignore the stereochemistry of the hydrolysis because there are two alcohols formed in that reaction. These are diastereomers because remember, diastereomers are stereoisomers that are not mirror images. We can look at these alcohols as cis and trans isomers because of the relative orientation of the methyl and OH groups. So, keep in mind that cis and trans isomers are diastereomers even if there are no chiral centers in the given molecules.
Let’s also draw the SN1 mechanism of this hydrolysis reaction:
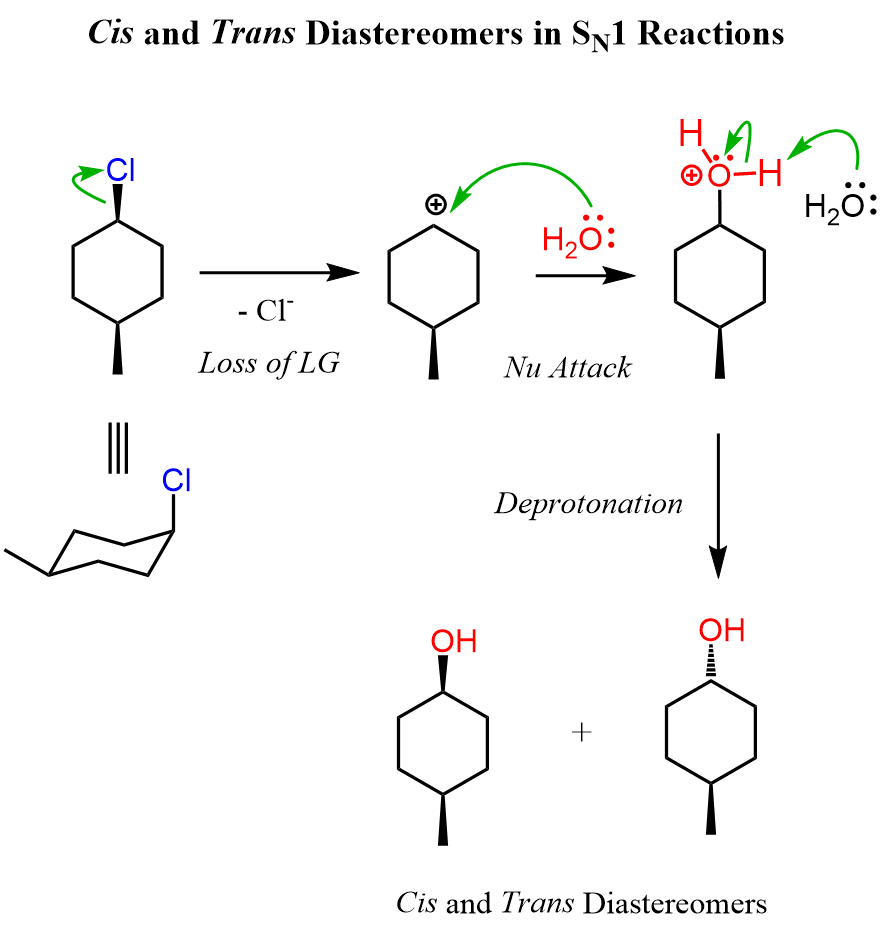
Try to draw the mechanism of reactions 4 and 5 before looking at his one first. Refer to this article for converting cyclohexanes into chair conformations and vice versa.
Rearrangements in SN1 Hydrolysis Reactions
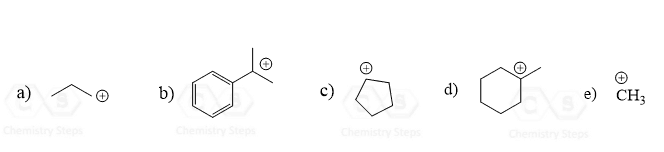
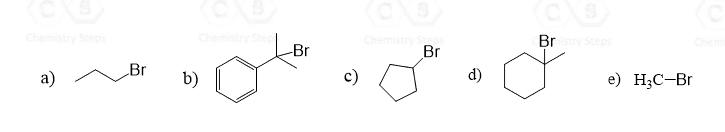
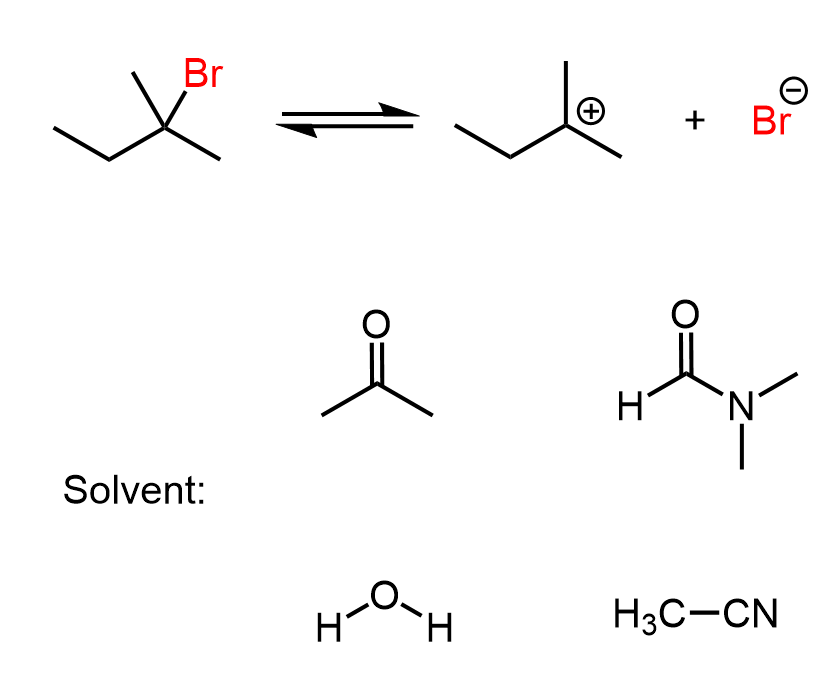
The SN1 mechanism is a unimolecular process where a carbocation is formed in the first step. You need to always check for rearrangement possibilities when dealing with carbocations. Common mistakes when forgetting about rearrangements are the wrong placement of the nucleophile in the product, the leaving group in the substrate, picking the wrong product, etc.
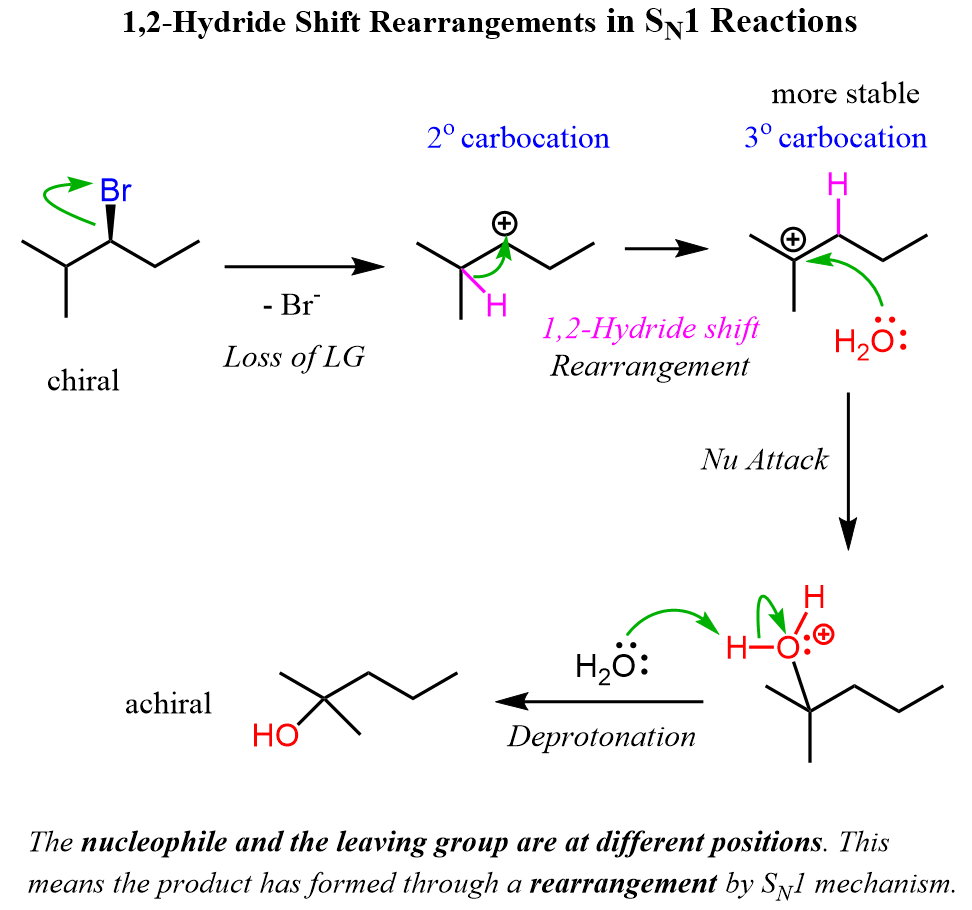
Remember, the main reason for rearrangement is the increased stability of the carbocation. Carbocations get more stable with the number of alkyl groups as they stabilize the positive charge on the carbon atom:

Let’s draw the mechanism of reaction 6 to see how this chiral secondary alkyl halide turns into an achiral tertiary alcohol:

Notice that the chirality of the middle carbon is lost upon the hydride shift because carbocations are sp2-hybridized and only have three atoms connected to the positively charged carbon. Because of symmetrical structures, the nucleophilic attack of water produces an achiral alcohol.
Reaction 7 is very similar to 6, and the only difference is that the chirality here is lost only due to the loss of the leaving group. Try to draw the mechanism before comparing it with what we have below.
Ring Expansion Rearrangements in SN1 Reactions
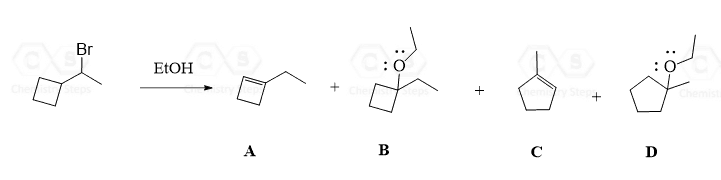
A common type of carbocation rearrangement is the ring expansion of 4 or 5-membered cycloalkanes. The driving force in these reactions is both the increased stability of carbocations (2o-3o) and the ring itself. Remember, 4-membered rings have quite a high ring strain, which is released when expanded to a 5-membered ring. Reaction 8 is an example of ring-expansion rearrangement in an SN1 reaction:
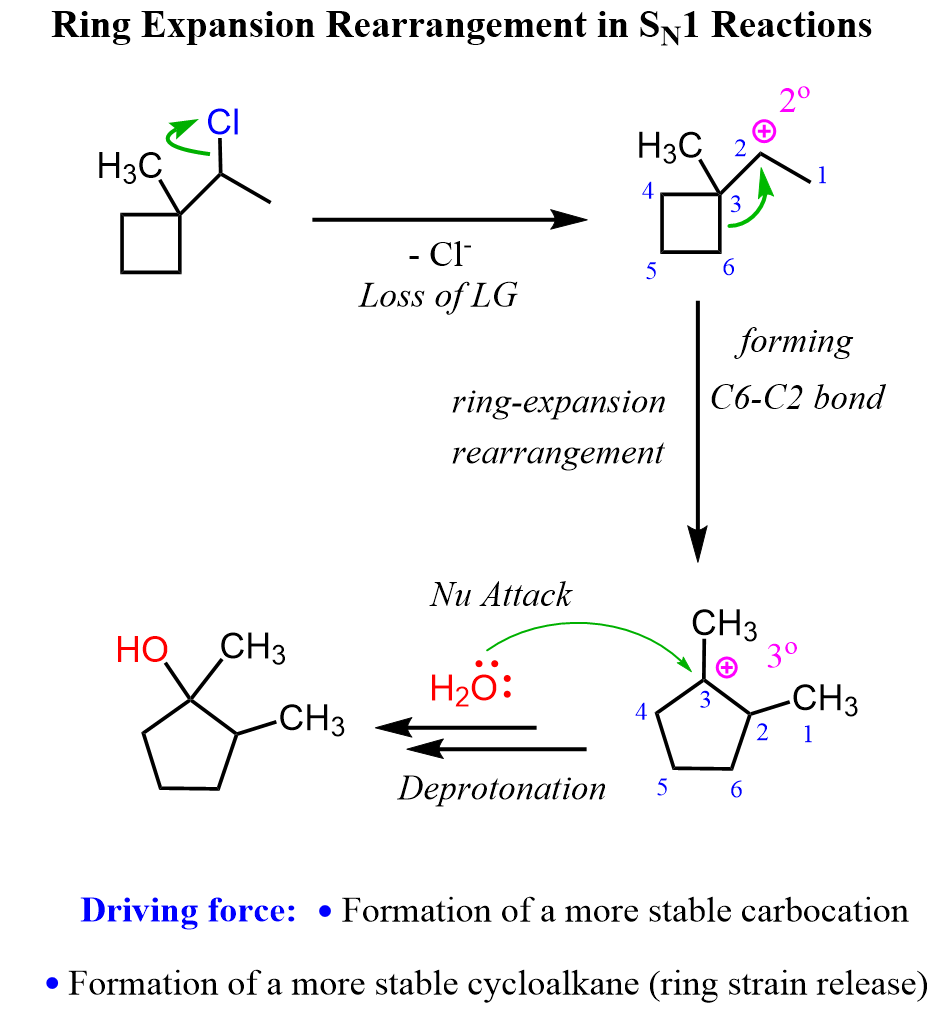
For cyclopentanes, despite being very stable molecules, they are still associated with little ring strain. This, combined with the possibility of rearrangement from secondary to tertiary carbocation, facilitates the ring expansion rearrangement of 5-membered rings to cyclohexenes.
Summary of SN1 Reactions of Water with Alkyl Halides
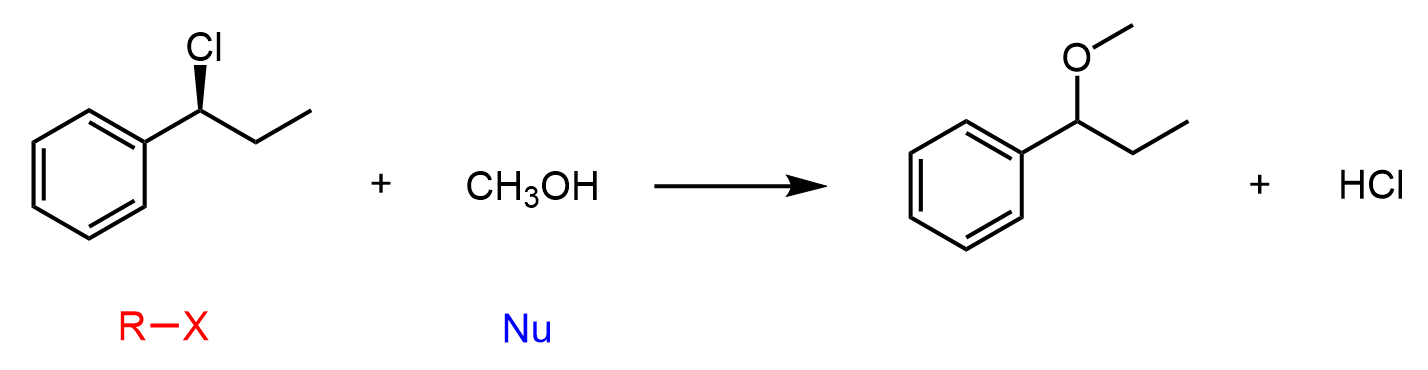
Let’s summarize what we learn. Water is a poor nucleophile and therefore, it reacts with alkyl halides by an SN1 mechanism, forming an alcohol. The same mechanism and pattern would be observed in their reaction with alcohols, as these are also poor nucleophiles. The product of alkyl halides with alcohols is an ether. If the reaction is heated, elimination reactions by the E1 mechanism may be favored.
SN1 reactions proceed via racemization of the chiral center that is part of the reaction. If a new chirality center is formed during the addition of the nucleophile, a pair of enantiomers is obtained. A common exception is when the substrate already contains an additional chiral center that is not part of the reaction. In this case, a pair of diastereomers is formed. Check this article on the most common exceptions in SN1 and SN2 reactions.
Rearrangements will occur whenever possible, so always watch out for those when dealing with unimolecular reactions such as SN1 and E1.