In today’s post, we will discuss the structures of some common Tropane Alkaloids, such as Atropine, Hyoscyamine, Scopolamine, and Cocaine. These naturally occurring compounds share the bicyclic tropane framework and play major roles in pharmacology, medicinal chemistry, and organic synthesis problems.
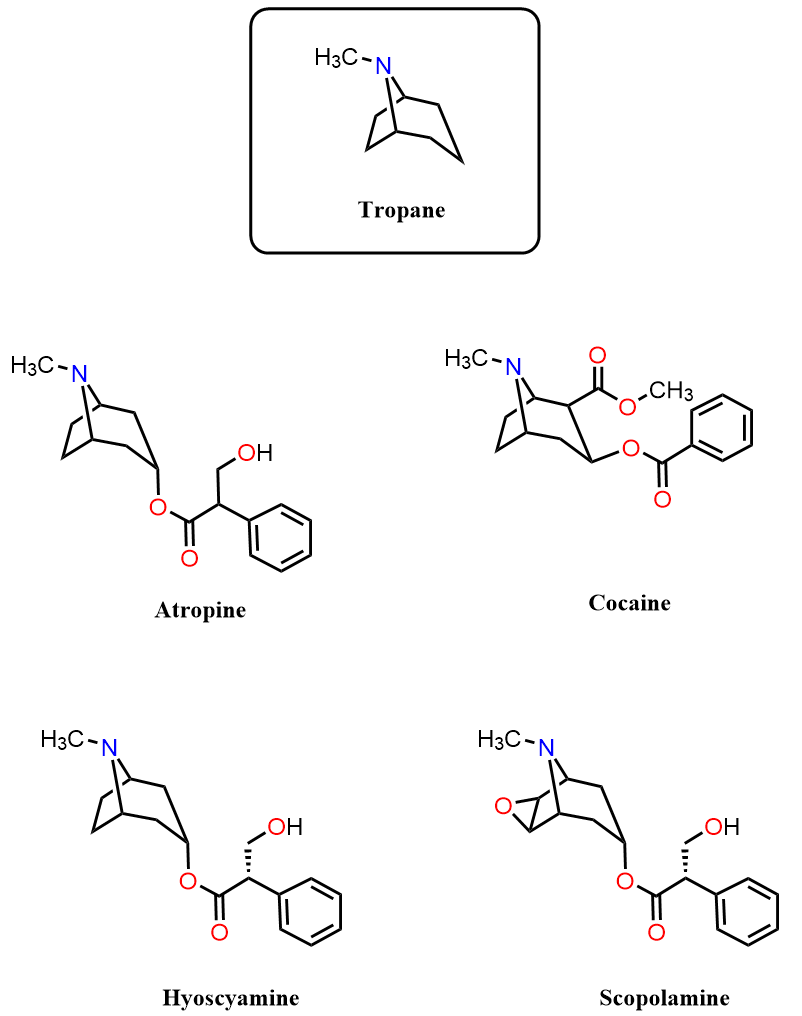
Our main focus will be the synthesis of atropine, one of the most historically significant and chemically interesting tropane alkaloids. We will turn our attention to how atropine is actually prepared by both the classical and more modern synthetic approaches. Before doing that, let’s quickly review the properties of the main tropane alkaloids.
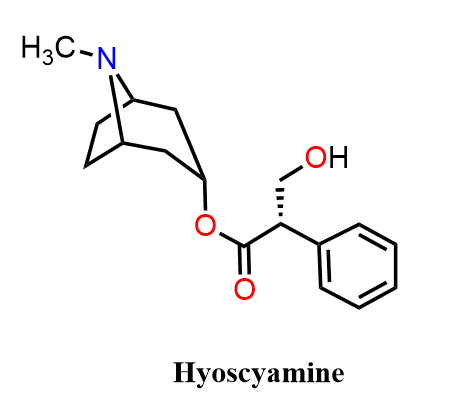 Hyoscyamine is the naturally occurring levorotatory isomer of atropine and is more potent at muscarinic receptors. It is found in plants such as Atropa belladonna and Hyoscyamus niger. It is the levorotary isomer of atropine (the third of the major nightshade alkaloids) and is therefore sometimes referred to as levo-atropine.
Hyoscyamine is the naturally occurring levorotatory isomer of atropine and is more potent at muscarinic receptors. It is found in plants such as Atropa belladonna and Hyoscyamus niger. It is the levorotary isomer of atropine (the third of the major nightshade alkaloids) and is therefore sometimes referred to as levo-atropine.
Clinically, hyoscyamine is used as an antispasmodic in gastrointestinal disorders, to reduce motility and secretions, and to manage symptoms of IBS and peptic ulcers. Because it is enantiomerically pure in nature, it often shows stronger physiological effects than atropine at equivalent doses.
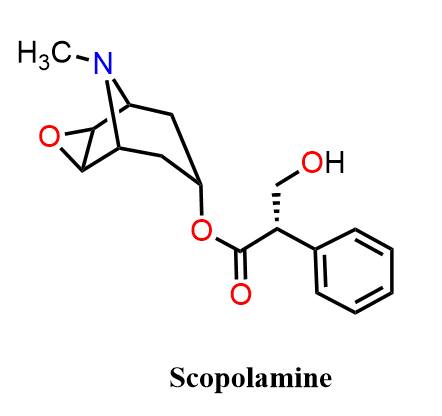 Scopolamine (also known as hyoscine) differs from atropine and hyoscyamine by having an epoxide (bridge) in the tropane ring, which increases its ability to cross the blood–brain barrier. As a result, scopolamine has strong central nervous system depressant effects, making it especially effective for preventing motion sickness, treating postoperative nausea, and reducing excessive salivation. At higher doses, it produces sedation, amnesia, and sometimes hallucinations.
Scopolamine (also known as hyoscine) differs from atropine and hyoscyamine by having an epoxide (bridge) in the tropane ring, which increases its ability to cross the blood–brain barrier. As a result, scopolamine has strong central nervous system depressant effects, making it especially effective for preventing motion sickness, treating postoperative nausea, and reducing excessive salivation. At higher doses, it produces sedation, amnesia, and sometimes hallucinations.
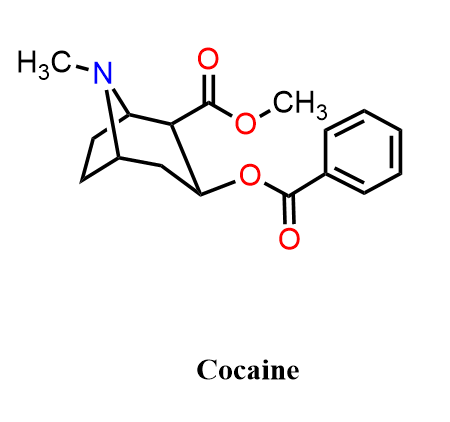 Cocaine is structurally related to the tropane alkaloids but has very different biological activity. It is a potent local anesthetic and powerful stimulant that works by inhibiting the reuptake of dopamine, norepinephrine, and serotonin. Although rarely, cocaine is still used medically in certain ENT (ear, nose, throat) procedures for its combined vasoconstrictive and anesthetic properties. However, its strong effects on the central nervous system and high potential for addiction have made it a controlled substance.
Cocaine is structurally related to the tropane alkaloids but has very different biological activity. It is a potent local anesthetic and powerful stimulant that works by inhibiting the reuptake of dopamine, norepinephrine, and serotonin. Although rarely, cocaine is still used medically in certain ENT (ear, nose, throat) procedures for its combined vasoconstrictive and anesthetic properties. However, its strong effects on the central nervous system and high potential for addiction have made it a controlled substance.
 Did you know that Coca-Cola originally contained small amounts of cocaine? When John Pemberton created the drink in 1886, it was made from coca leaf extract and cola nuts, and at the time, cocaine was legal and widely used in tonics for its stimulating, mood-lifting, and pain-relieving effects. Early consumers believed Coca-Cola boosted energy, reduced fatigue, improved mood, and helped with headaches, benefits largely attributed to the cocaine content combined with caffeine. Public concern and new regulations led Coca-Cola to remove cocaine by 1903, switching to coca leaves with the alkaloid extracted. The company still uses cocaine free coca leaf extract today, so the modern beverage contains no cocaine at all.
Did you know that Coca-Cola originally contained small amounts of cocaine? When John Pemberton created the drink in 1886, it was made from coca leaf extract and cola nuts, and at the time, cocaine was legal and widely used in tonics for its stimulating, mood-lifting, and pain-relieving effects. Early consumers believed Coca-Cola boosted energy, reduced fatigue, improved mood, and helped with headaches, benefits largely attributed to the cocaine content combined with caffeine. Public concern and new regulations led Coca-Cola to remove cocaine by 1903, switching to coca leaves with the alkaloid extracted. The company still uses cocaine free coca leaf extract today, so the modern beverage contains no cocaine at all.
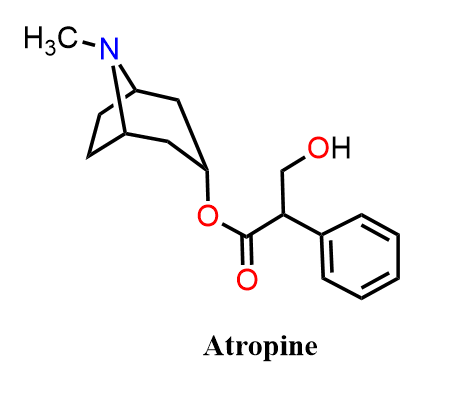 Atropine is a potent anticholinergic agent that works by competitively blocking muscarinic acetylcholine receptors. Clinically, it is used to increase heart rate in cases of bradycardia, dilate pupils (mydriasis) for ophthalmic procedures, dry secretions, and, most importantly, to counteract organophosphate and nerve-agent poisoning, where it prevents life-threatening overstimulation of the parasympathetic nervous system. Atropine is actually a racemic mixture, while its naturally occurring enantiomer is (–)-hyoscyamine, the biologically active form.
Atropine is a potent anticholinergic agent that works by competitively blocking muscarinic acetylcholine receptors. Clinically, it is used to increase heart rate in cases of bradycardia, dilate pupils (mydriasis) for ophthalmic procedures, dry secretions, and, most importantly, to counteract organophosphate and nerve-agent poisoning, where it prevents life-threatening overstimulation of the parasympathetic nervous system. Atropine is actually a racemic mixture, while its naturally occurring enantiomer is (–)-hyoscyamine, the biologically active form.
Speaking of atropine, let’s now take a closer look at how this important tropane alkaloid is synthesized.
The Synthesis of Atropine
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to the answers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps, and the powerful set of Organic Chemistry 1 and 2 Summary Study Guides.
Two main building blocks make up Atropine, and they react in the final step of its synthesis. These are Tropine and Tropic Acid (I will show the reaction here).
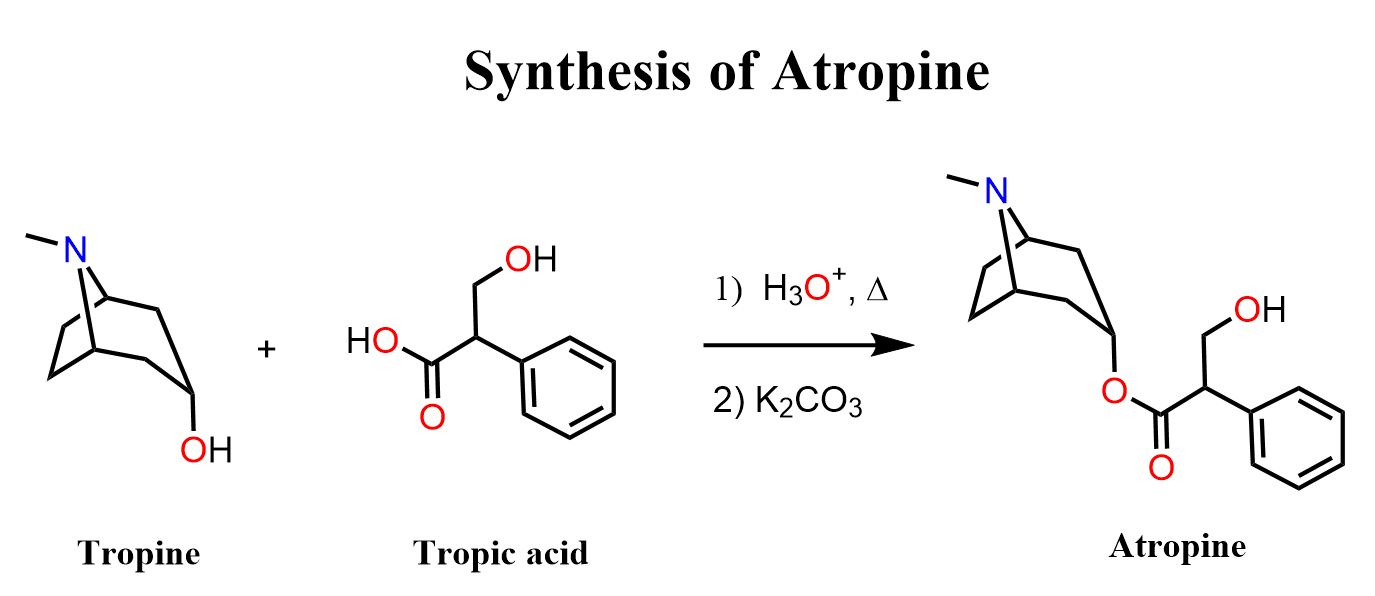
So, let’s see how Tropine and Tropic Acid are prepared.
The Synthesis of Tropine
The ketone (cycloheptanone, aka suberone) was first transformed into cycloheptene through aminocycloheptane, then into cycloheptadiene and cycloheptatriene.
When the cycloheptadiene was treated with bromine, the thermodynamic 1,4-addition occurred, allowing the formation of cycloheptatriene upon reaction with the bulky base quinoline.
This transformation was essentially repeating steps of adding bromine followed by Hofmann elimination, which is performed on the amine using CH₃I and Ag₂O.
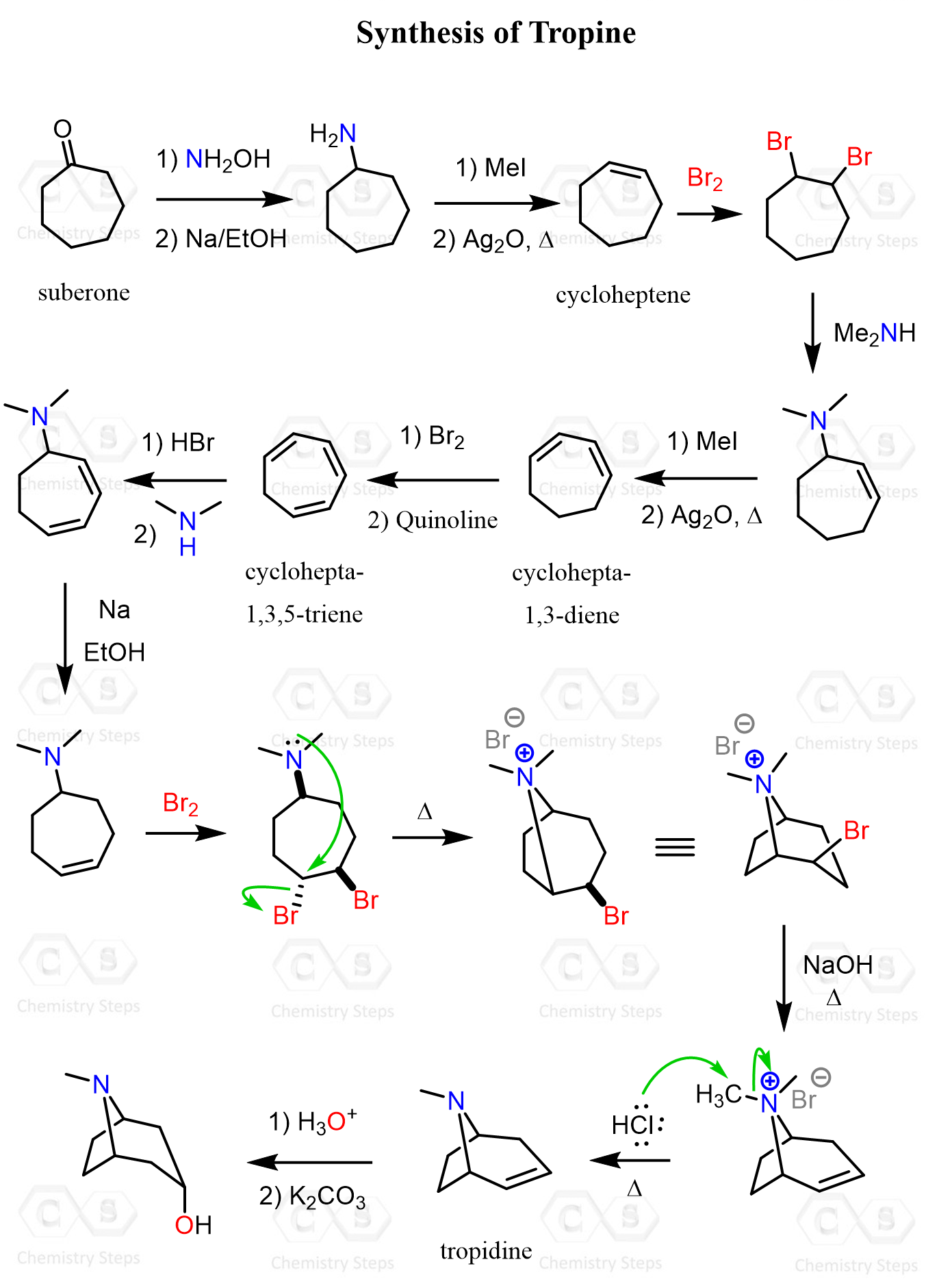
The cycloheptatriene underwent addition of hydrogen bromide, followed by nucleophilic substitution with dimethylamine and reduction using Na in ethanol. This is similar to the Birch reduction, and the isomer with the double bond on the opposite side of the ring was obtained.
The bromination of this intermediate gives a racemic mixture of two trans dibromides. At this point, we have a nucleophilic group and two Br atoms, which are excellent leaving groups. What is going to happen next?
The intramolecular SN2 substitution of the Br trans to the amino group produced the desired cyclic framework of tropane in the form of an ammonium salt, which was demethylated using HCl as a source of chloride ion and subjected to elimination by NaOH to yield tropidine.
Tropidine was then converted into tropine via acid-catalyzed hydration of the double bond. Finally, Fischer esterification with methanol and benzoylation produced racemic cocaine.
The Synthesis of Tropic Acid
The classical synthesis of tropic acid begins with acetophenone, which is converted to a cyanohydrin. Hydrolysis and dehydration of this cyanohydrin form an intermediate α,β-unsaturated acid containing a C=C double bond. This intermediate is then treated with HCl, and, unlike what one may expect, the chloride adds to the anti-Markovnikov position.
One possible explanation for this unusual regioselectivity is the instability of a carbocation located next to the electron-withdrawing carboxyl group. I have not done deep research on this, but perhaps some intramolecular stabilization of the primary cation plays a part as well. Regardless, upon hydrolysis, the outcome is the formation of tropic acid, which now needs to be reacted with the tropnine.
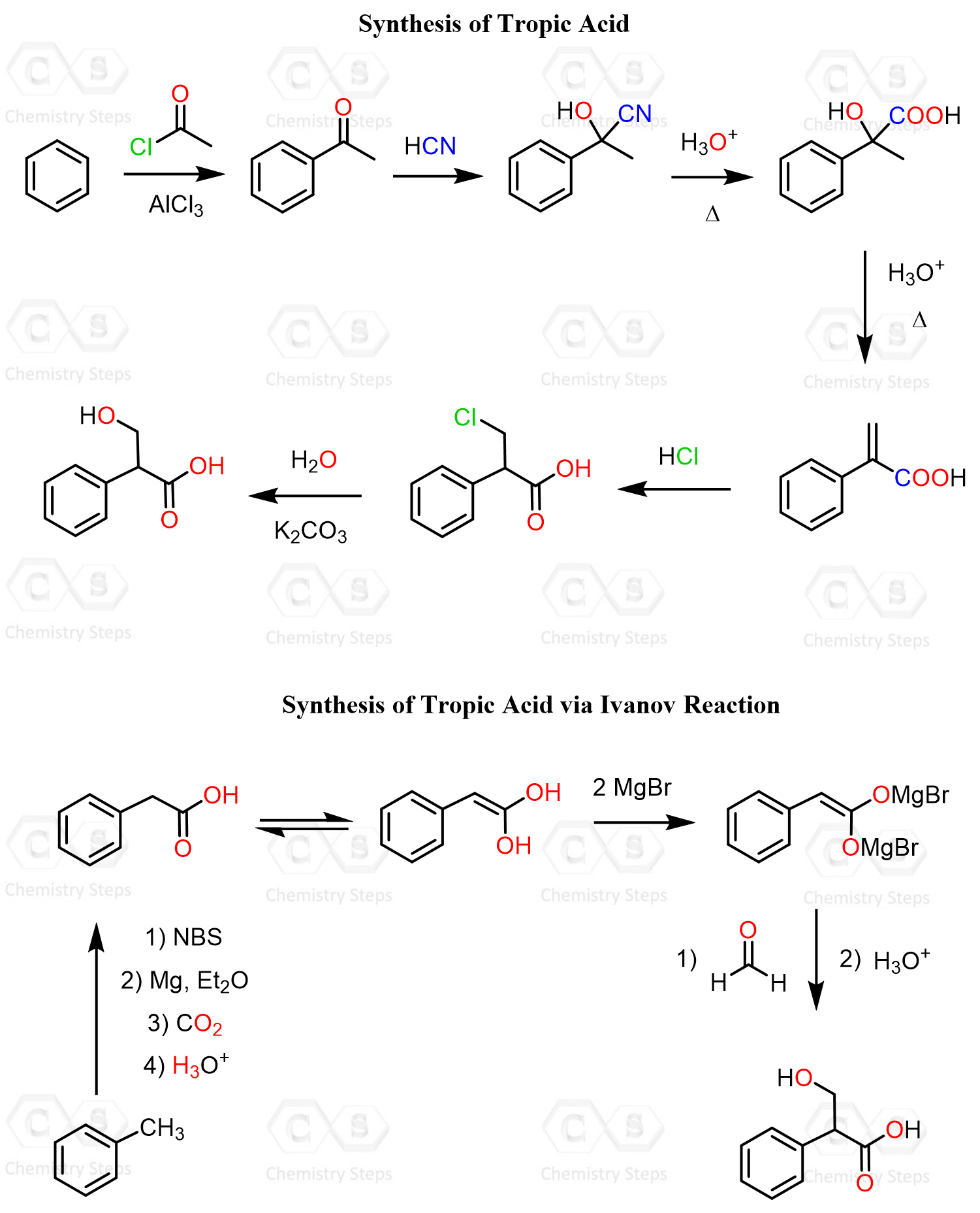
A more modern alternative synthesis of tropic acid is achieved using the Ivanov reaction. The Ivanov reaction is a carbon–carbon bond-forming process involving the addition of dianions (endiolates) of arylacetic acids, aka Ivanov reagents, to electrophilic substrates such as aldehydes, ketones, isocyanates, or alkyl halides, giving β-hydroxy acids.
In the case of tropic acid, it can be prepared by the Ivanov reaction between phenylacetic acid and formaldehyde, which produces the corresponding β
