You’ve already learned what alkanes are, their structure, how they’re named, and you probably even remember the first ten representatives (methane through decane).
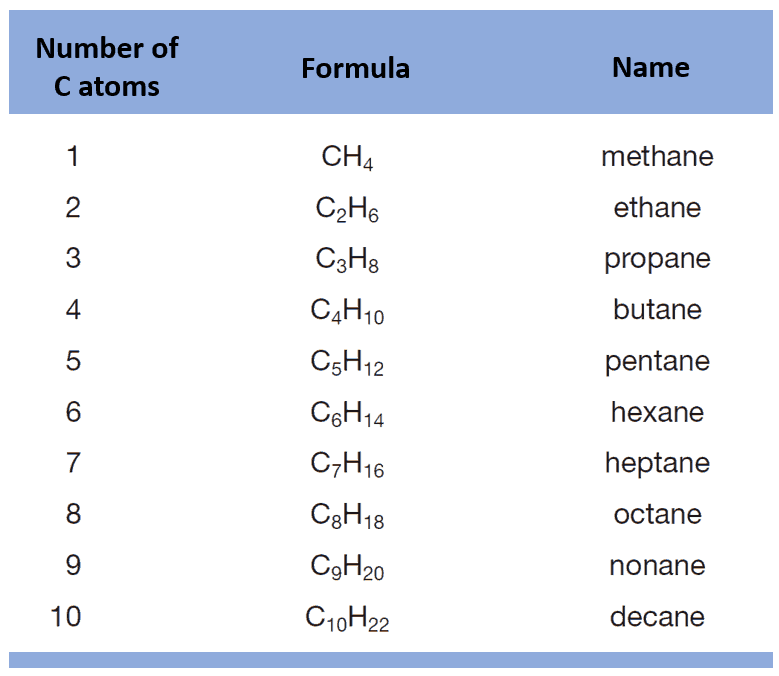
Now, if we take any alkane and replace one of its hydrogen atoms with a halogen (F, Cl, Br, or I), we get a new class of compounds called alkyl halides.
For example, let’s draw the bond-line structure of pentane and replace one of the terminal hydrogens with a chlorine atom:
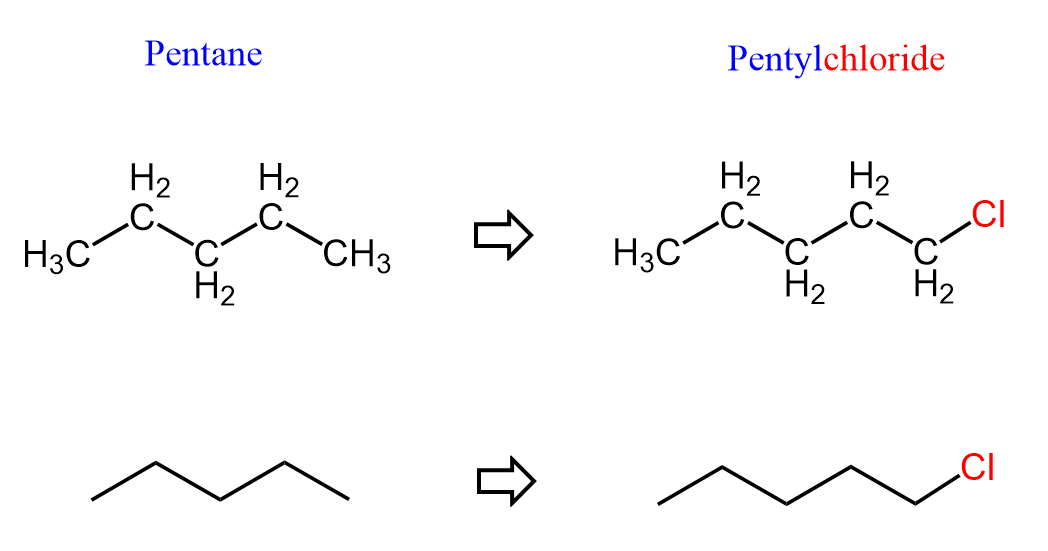
This is pentyl chloride, which is an alkyl halide. As a reminder, alkyl groups are fragments of alkanes formed by removing one hydrogen atom, allowing them to attach to a parent chain or functional group. For example, methyl (–CH₃), ethyl (–CH₂CH₃), and propyl (–CH₂CH₂CH₃).

It is worth mentioning here that pentyl chloride is not the IUPAC name – it is a common name, whereas IUPAC names are the formal and official names of compounds. Think about this like names and nicknames.
According to IUPAC rules, we’d call this molecule 1-chloropentane. So, the main part of the molecule, known as the parent chain, is pentane, and the chlorine is a substituent. We have a dedicated article on the IUPAC rules with lots of examples, so feel free to check that out as well.

Primary, Secondary, and Tertiary Alkyl Halides
Depending on what carbon atom the halogen is connected to, we can have a primary, secondary, or a tertiary alkyl halide. For example, if, instead of the terminal carbon atom, we connected the chlorine to the middle one, we’d get 3-chloropentane, which is a secondary alkyl halide:

Now, why is it a secondary alkyl halide? To identify this, we need to determine how many carbons are connected to the carbon atom connected to the halogen. In this case, the middle carbon is connected to two other carbons, therefore, it is a secondary carbon, and thus we have a secondary alkyl halide.
In short, these definitions are assigned to carbon atoms based on the number of other carbon atoms they are connected to:
- Primary carbons are connected to one carbon only.
- Secondary carbons are connected to two carbon atoms.
- Tertiary carbons are connected to three carbon atoms.
- And if four carbons are connected to a carbon, then it is a quaternary carbon.
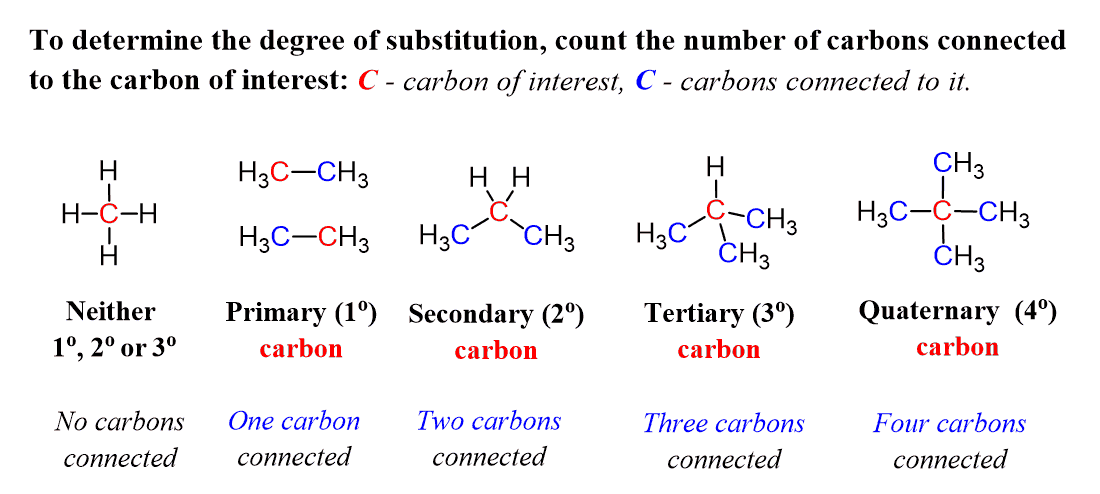
Building on this, we can draw a few examples of primary, secondary, and tertiary alkyl halides. Notice that we cannot have quaternary alkyl halides because if the carbon is connected to four carbon atoms, it has no room to bond with another atom.
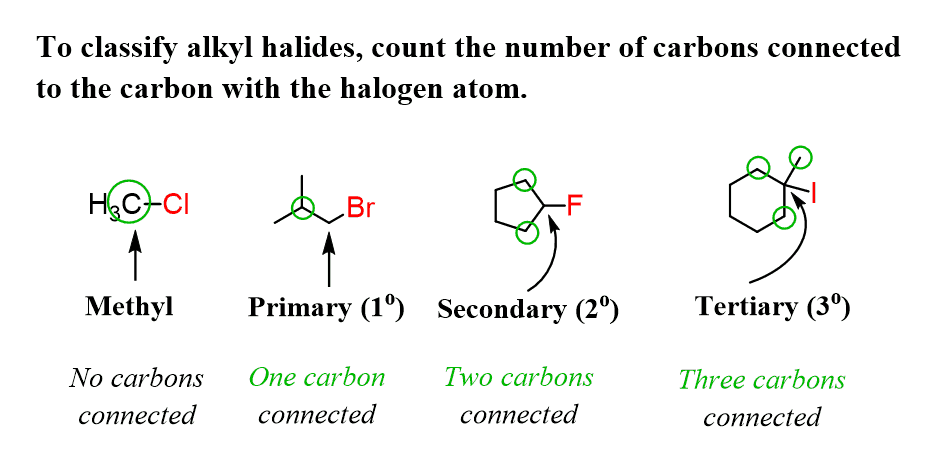
Why Are Alkyl Halides Important?
Alkyl halides are derivatives of alkanes, but they greatly differ in their reactivity compared to alkanes. Alkanes are made of all nonpolar bonds and, except for radical halogenations, they do not undergo any significant transformations such as substitution or elimination reactions.
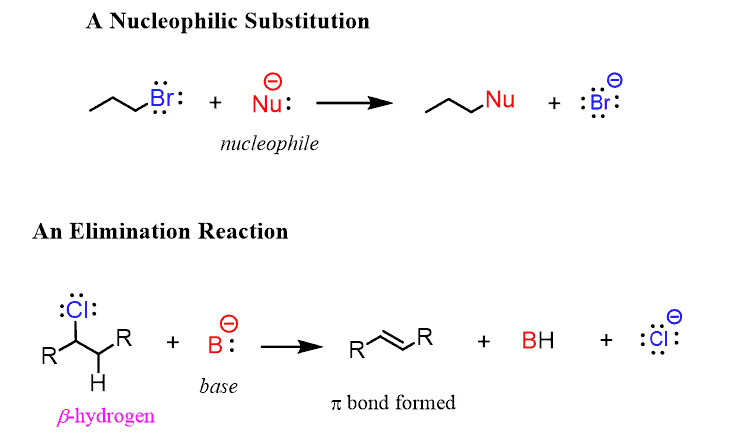
These names may sound intimidating, but do not worry—you do not need to understand what they are right now. To be fair, they are going to be a large portion of what you cover in Organic Chemistry 1, so when you finish the topics of curved arrows and acid-base chemistry, try to go over them for a little heads-up.
Vinyl and Aryl Halides
Alkanes are the simplest class of hydrocarbons, but you might also have learned about alkenes, alkynes, and arenes (aromatic compounds). When one or more hydrogen atoms in these hydrocarbons are replaced with a halogen, an alkyl, vinyl, or aryl halide is formed:
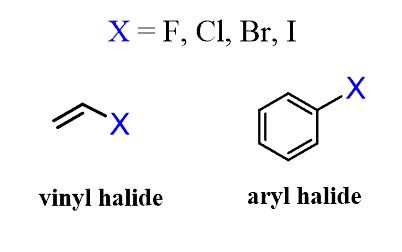
Summary
Let’s keep this short and to the point.
- You have learned what alkyl halides are – these are organic compounds where a halogen atom (F, Cl, Br, or I) is bonded to an sp³-hybridized carbon.
- Depending on the number of carbon atoms connected to the carbon bearing the halogen, there can be primary, secondary, or tertiary alkyl halides.
- Alkyl halides are an important class of organic compounds because they are used as a starting point for synthesizing molecules with other functional groups.
You are going to see alkyl halides throughout the semester when discussing substitution and elimination reactions.
Check Also
- Naming Alkanes by IUPAC Nomenclature Rules Practice Problems
- Naming Bicyclic Compounds
- Naming Bicyclic Compounds-Practice Problems
- How to Name a Compound with Multiple Functional Groups
- Primary, Secondary, and Tertiary Carbon Atoms in Organic Chemistry
- Constitutional or Structural Isomers with Practice Problems
- Degrees of Unsaturation or Index of Hydrogen Deficiency
- The Wedge and Dash Representation
- Sawhorse Projections
- Newman Projections with Practice Problems
- Staggered and Eclipsed Conformations
- Conformational Isomers of Propane
- Newman Projection and Conformational Analysis of Butane
- Newman Projection of Chair Conformation
- Gauche Conformation
- Gauche Conformation, Steric, Torsional Strain Energy Practice Problems
- Ring Strain
- Steric vs Torsional Strain
- Conformational Analysis
- Drawing the Chair Conformation of Cyclohexane
- Ring Flip: Drawing Both Chair Conformations with Practice Problems
- 1,3-Diaxial Interactions and A value for Cyclohexanes
- Ring-Flip: Comparing the Stability of Chair Conformations with Practice Problems
- Cis and Trans Decalin
- IUPAC Nomenclature Practice Problems
- IUPAC Nomenclature Summary Quiz
- Alkanes and Cycloalkanes Practice Quiz
