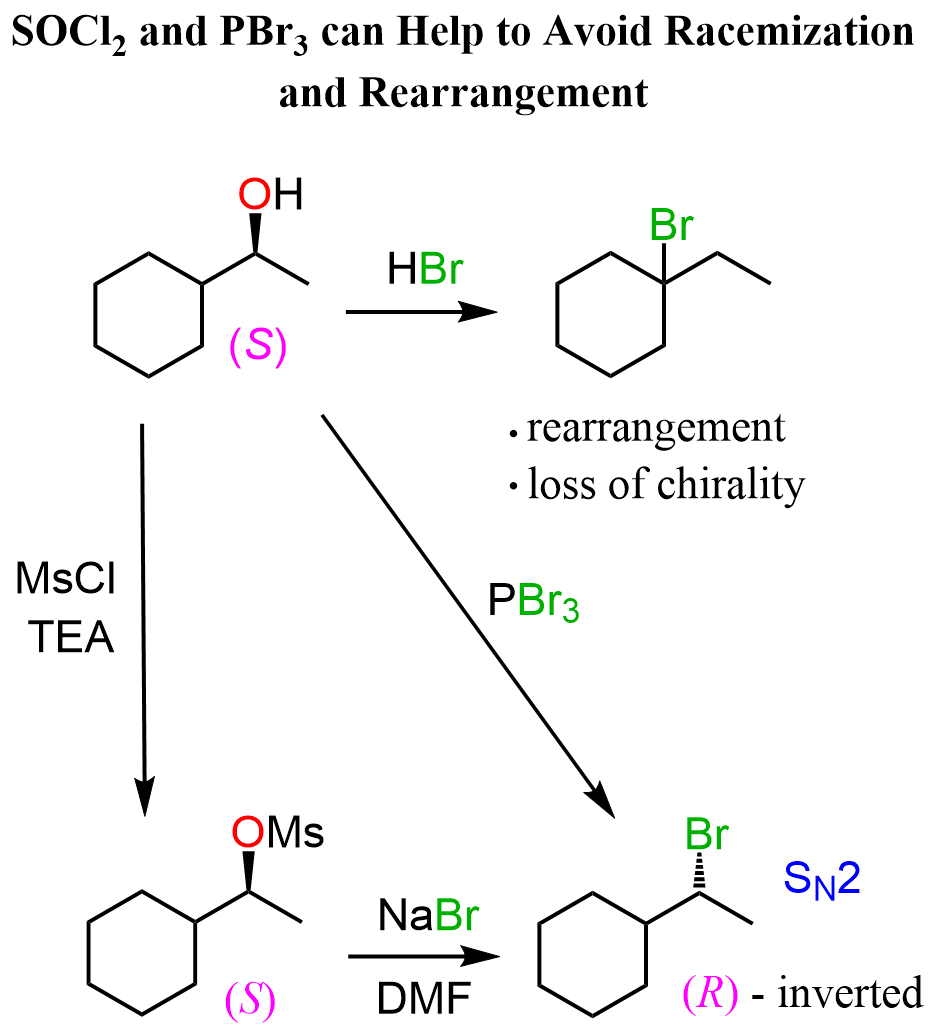
In the previous post, we discussed different methods for converting alcohols to alkyl halides, including their reaction with hydrohalic acids, the use of mesylates and tosylates, as well as reagents such as thionyl chloride (SOCl2) and phosphorus tribromide (PBr3).
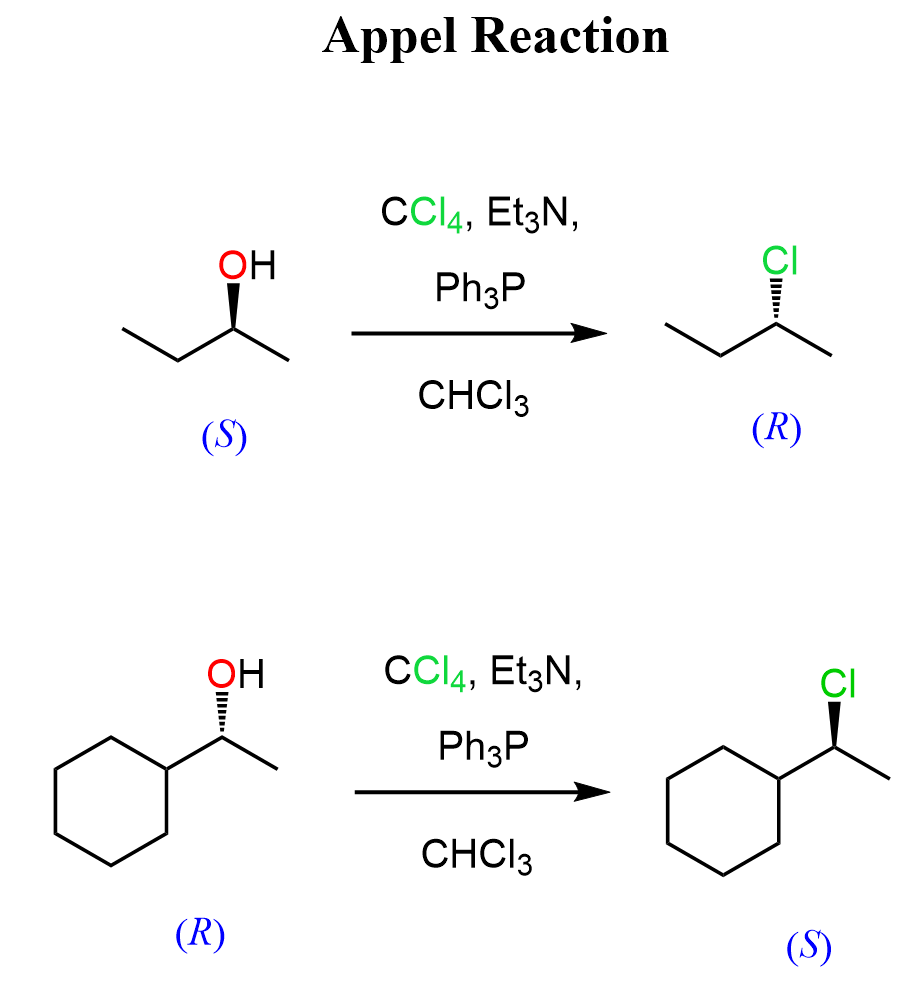
In today’s post, we will look at another strategy for converting alcohols to alkyl halides with control over the stereochemistry of the reaction. This transformation is known as the Appel reaction, developed by Rolf Appel in the 1970s.
In its classical form, it employs triphenylphosphine (PPh₃) and a tetrahalomethane such as carbon tetrachloride (CCl₄) or carbon tetrabromide (CBr₄) to transform primary and secondary alcohols into the corresponding alkyl chlorides or bromides.
The Mechanism of the Appel Reaction
The reaction begins with the reaction between triphenylphosphine (1) and the tetrahalomethane (CCl4 in this case) to generate a reactive phosphonium species along with a halide ion. Right upon the nucleophilic attack by PPh3, the chlorophosphonium salt intermediate 2 is formed, which is then attacked by the alcoxide ion, forming an alkoxyphosphonium intermediate 3, which effectively converts the poor leaving group (OH) into a much better one. At this stage, the halide ion performs a backside attack on the carbon bearing the oxygen, displacing the phosphonium group in an SN2 fashion and forming the alkyl halide with an inverted configuration.
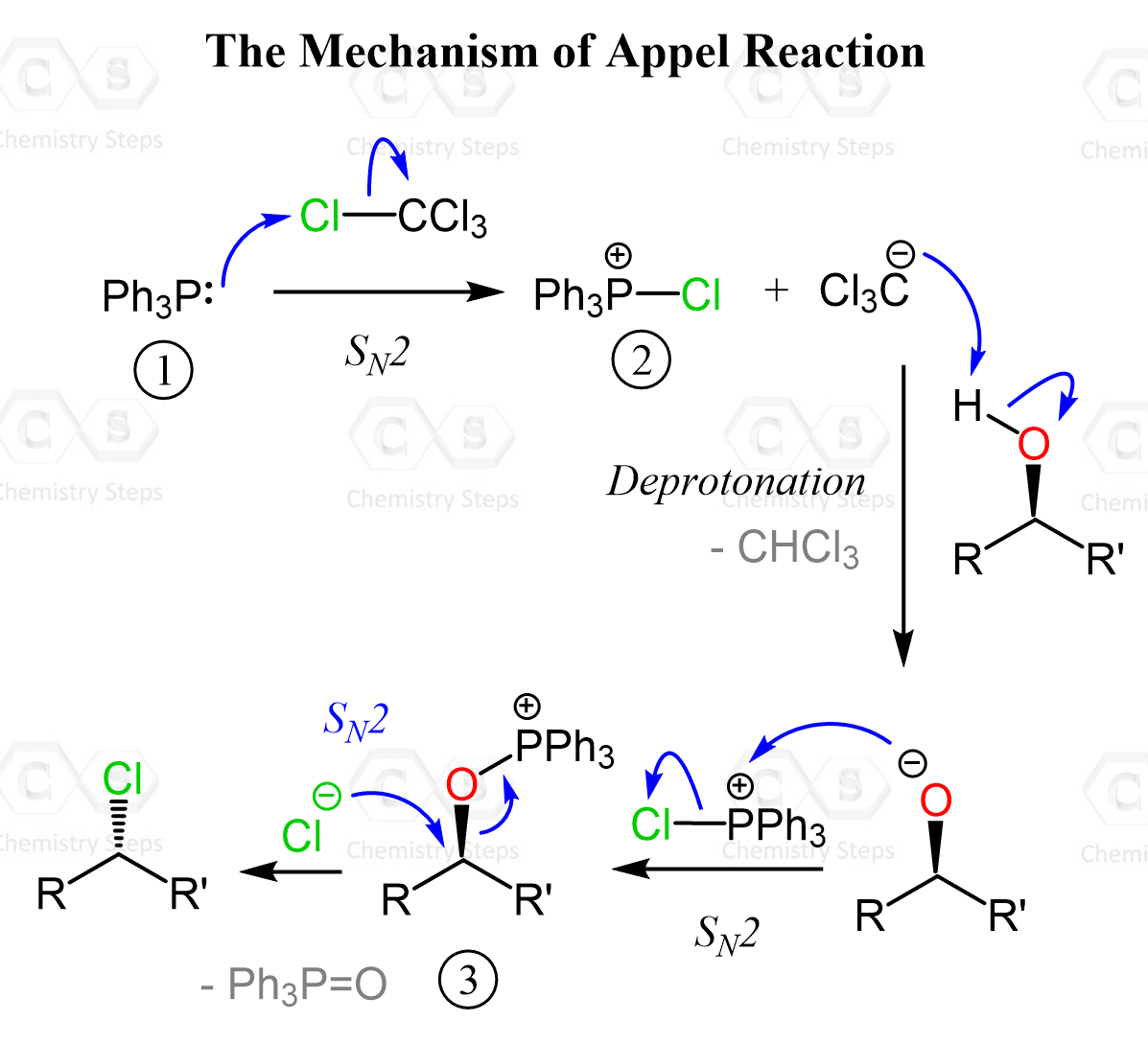
The byproducts are typically triphenylphosphine oxide and a haloform (such as CHCl₃), both of which can complicate purification.
It is useful to compare the Appel reaction with the Mitsunobu reaction, as both rely on triphenylphosphine to activate an alcohol. However, the Mitsunobu reaction uses PPh₃ in combination with an azo reagent (such as DEAD or DIAD) and an acidic nucleophile, allowing substitution of the OH group with a wide range of nucleophiles, again with inversion of configuration.

In contrast, the Appel reaction specifically introduces a halide nucleophile and does not require an external acidic component. While both reactions proceed through related phosphonium intermediates, they serve different synthetic purposes.
Other Uses of the Appel Reaction
In a broader sense, the Appel reaction can be viewed as a method for converting the OH group into a good leaving group, enabling subsequent substitution or elimination reactions. While substitution to form alkyl halides is the most common application, elimination pathways can also be accessed under certain conditions. A relevant example is the conversion of aldehydes to nitriles via oximes, where an Appel-type system is used to dehydrate the oxime, effectively eliminating water to form the carbon–nitrogen triple bond.
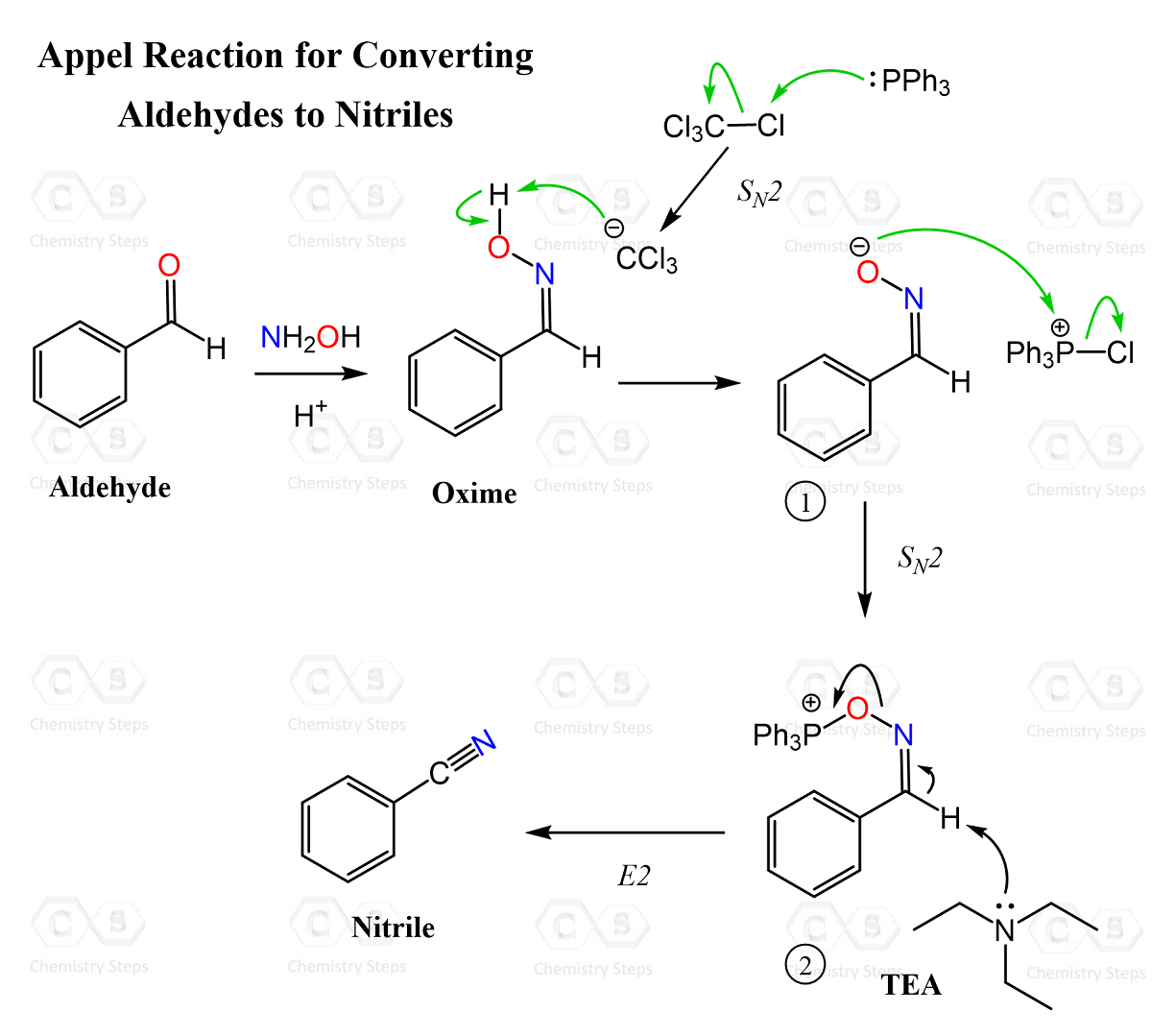
Despite its utility, the Appel reaction has several important drawbacks. The use of carbon tetrachloride (CCl₄) is a major concern due to its toxicity, environmental hazards, and role in ozone depletion. Additionally, the reaction requires stoichiometric triphenylphosphine, leading to the formation of large amounts of triphenylphosphine oxide, which is often difficult to remove and reduces the overall efficiency of the process.
For these reasons, alternative methods have been developed. As discussed earlier, Appel-type dehydrations using oxalyl chloride ((COCl)₂) in combination with catalytic Ph₃PO and a base such as triethylamine provide a more practical and environmentally acceptable approach in certain transformations.
Check Also
- Introduction to Alkyl Halides
- Nomenclature of Alkyl Halides
- Nucleophilic Substitution Reactions – An Introduction
- All You Need to Know About the SN2 Reaction Mechanism
The SN2 Mechanism: Kinetics, Thermodynamics, Curved Arrows, and Stereochemistry with Practice Problems - The Stereochemistry of SN2 Reactions
- Stability of Carbocations
- The SN1 Nucleophilic Substitution Reaction
- Reactions of Alkyl Halides with Water
- The Stereochemistry of the SN1 Reaction Mechanism
- The SN1 Mechanism: Kinetics, Thermodynamics, Curved Arrows, and Stereochemistry with Practice Problems
- The Substrate and Nucleophile in SN2 and SN1 Reactions
- Carbocation Rearrangements in SN1 Reactions with Practice Problems
- Ring Expansion Rearrangements
- Ring Contraction Rearrangements
- When Is the Mechanism SN1 or SN2?
- Reactions of Alcohols with HCl, HBr, and HI Acids
- SOCl2 and PBr3 for Conversion of Alcohols to Alkyl Halides
- Alcohols in SN1 and SN2 Reactions
- How to Choose Molecules for Doing SN2 and SN1 Synthesis-Practice Problems
- SN1 vs E1 Reactons
- SN2 vs E2 Reactins
- Exceptions in SN2 and SN1 Reactions
- Nucleophilic Substitution and Elimination Practice Quiz
- Reactions Map of Alkyl Halides
- Reaction Map of Alcohols
