We have seen many times in SN1, SN2, E1, and E2 reactions that halogens, more specifically, their ions called halides, are the leaving group.
A common question many students wonder about, and make the mistake of, is whether an OH group can be a leaving group. So, essentially, have an OH instead of the halogen and perform a substitution or elimination reaction.
Let’s put it straight up and say that, NO, –OH is not a leaving group, and you should never kick it out like a halogen in substitution and elimination reactions.

The reason for this is that the hydroxide ion, –OH, is a strong base, which means the negative charge is poorly stabilized.
Remember, in order to be a good leaving group, the species must be a weak base, meaning the negative charge must be well-stabilized.
How to Convert the OH into a Good Leaving Group?
There are different ways of converting the hydroxyl into a good leaving group.
The most common is the use of mesylates and tosylates. These are the analogs of halides, and they undergo all types of substitution and elimination reactions.
Here is a list of reactions showing how alcohols are converted to mesylates and tosylates, and the latter undergo SN1, SN2, E1, and E2 reactions:
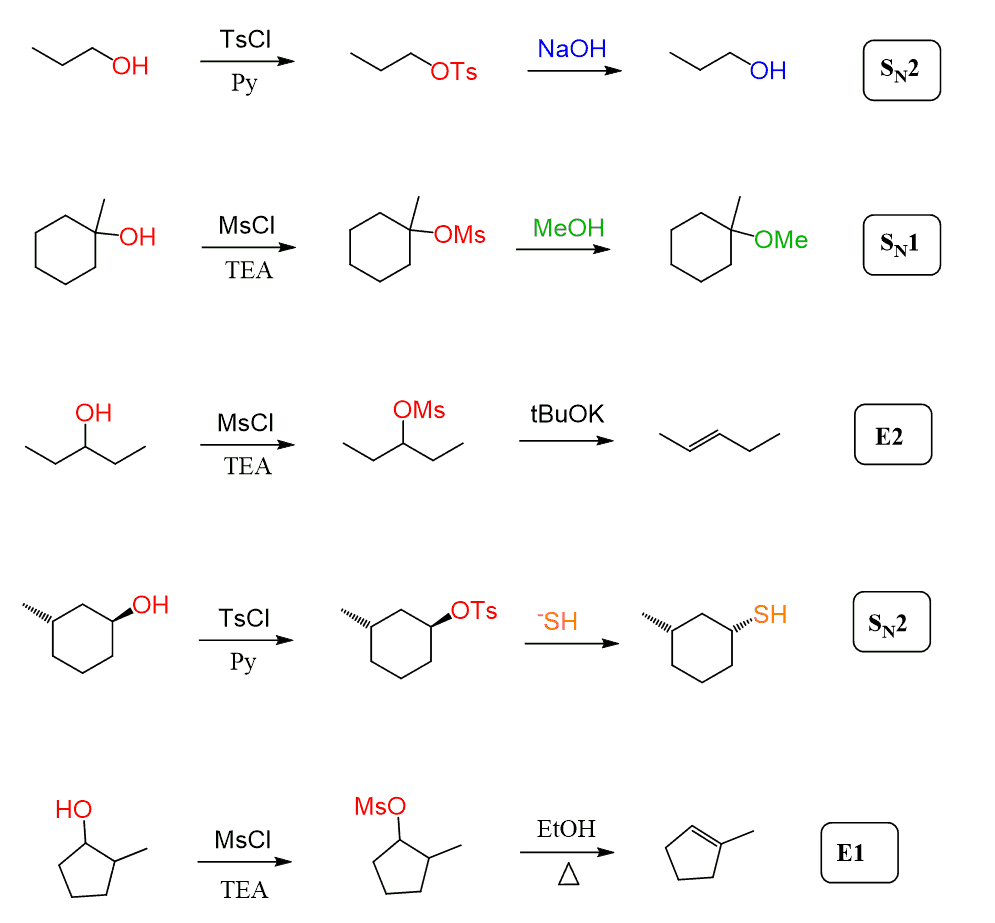
Check the corresponding article for more details, including the mechanism of mesylate and tosylate formation.
Another common strategy, especially used for secondary chiral alcohols, is the use of SOCl₂ and PBr₃ reagents, as these convert the OH group into the corresponding halide via inversion of configuration. So, this allows control over the stereochemistry of the reaction.
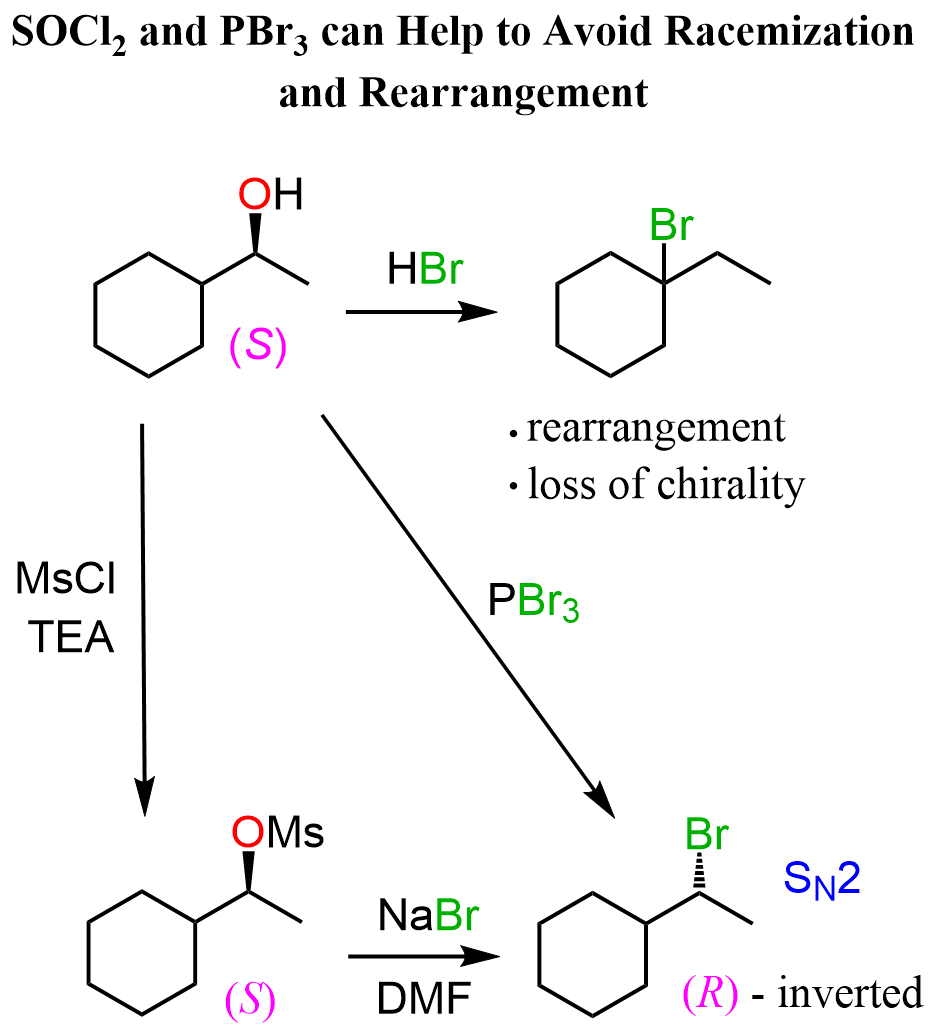
Notice that in the top-right corner, we have a conversion of the alcohol to an alkyl halide by reacting it with hydrohalic acids such as HBr, but the product is formed via a carbocation rearrangement.
This is one disadvantage of using HX acids with alcohols, as there is little to no control over the stereochemistry, and rearrangements become possible. The particular rearrangement in this scheme is a ring-expansion rearrangement.
Check the linked articles for more details about the reaction of alcohols with hydrohalic acids, carbocation rearrangements, and strategies to avoid them.
Can OH Ever Be a Leaving Group?
In Organic Chemistry 2, you will learn about what is called an E1cB elimination, where the OH does serve as a leaving group. Yes, no mesylate, tosylate, or other tools are used.
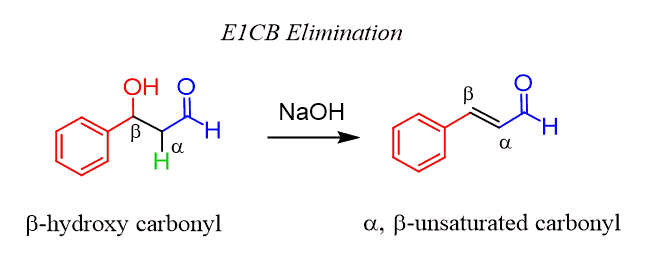
The key role here is left to the C=O carbonyl group, which helps to remove the hydrogen, and the OH is lost by a keto-enol tautomerization.
You don’t need to worry about this – it is just an illustration of the rare exception when –OH acts as a leaving group. Do NOT do this in SN1, SN2, E1, and E2 reactions.
To Summarize
This was a short post answering a common question of whether the OH can be a leaving group. The conversion of the OH into a good leaving group, and the reactions of corresponding derivatives such as mesylates, tosylates, and alkyl halides, is what makes the entire chapter of nucleophilic substitution and elimination reactions.
Check out the related articles below, and consider becoming a CS member to gain access to all our study guides, Reaction Maps, Practice Problems, and Multiple-Choice Quizzes.
Let me know in the comments if you have any questions, and good luck, which comes with studying hard, in your organic chemistry class.
Check Also
- Introduction to Alkyl Halides
- Nomenclature of Alkyl Halides
- Nucleophilic Substitution Reactions – An Introduction
- All You Need to Know About the SN2 Reaction Mechanism
The SN2 Mechanism: Kinetics, Thermodynamics, Curved Arrows, and Stereochemistry with Practice Problems - The Stereochemistry of SN2 Reactions
- The SN1 Nucleophilic Substitution Reaction
- Reactions of Alkyl Halides with Water
- The Stereochemistry of SN1 Reaction Mechanism
- The SN1 Mechanism: Kinetics, Thermodynamics, Curved Arrows, and Stereochemistry with Practice Problems
- The Substrate and Nucleophile in SN2 and SN1 Reactions
- Carbocation Rearrangements in SN1 Reactions with Practice Problems
- Ring Expansion Rearrangements
- Ring Contraction Rearrangements
- When Is the Mechanism SN1 or SN2?
- Reactions of Alcohols with HCl, HBr, and HI Acids
- SOCl2 and PBr3 for Conversion of Alcohols to Alkyl Halides
- Alcohols in Substitution Reactions with Tons of Practice Problems
- How to Choose Molecules for Doing SN2 and SN1 Synthesis-Practice Problems
- Exceptions in SN2 and SN1 Reactions
- Nucleophilic Substitution and Elimination Practice Quiz
- Reactions Map of Alkyl Halides
