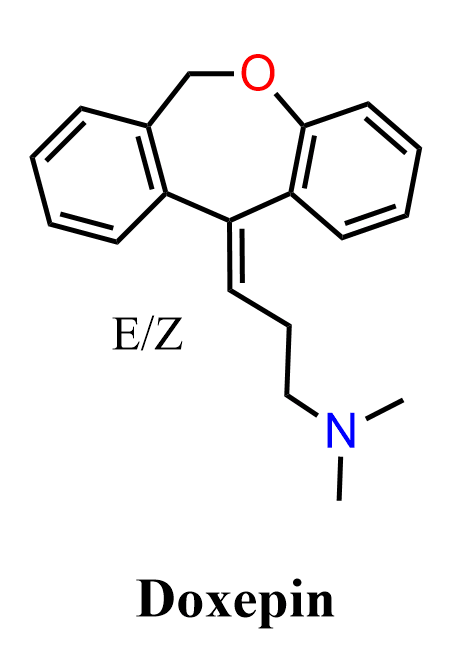
Doxepin is a tricyclic antidepressant widely used in the treatment of depression, anxiety disorders, and insomnia. It belongs to a class of drugs characterized by a three-ring system, consisting of two benzene rings fused to a central seven-membered ring, with a dialkylaminoalkyl side chain attached. Based on the substitution at the terminal nitrogen, tricyclic antidepressants are classified as tertiary amines and secondary amines. Doxepin is a tertiary amine, similar to imipramine and amitriptyline, and typically shows stronger effects on serotonin reuptake.
The exact mechanism of action of tricyclic antidepressants is not fully understood, but they are believed to inhibit the neuronal reuptake of serotonin and norepinephrine, increasing their concentration in the synaptic cleft and enhancing their effects. Additional interactions with histamine and adrenergic receptors also contribute to their pharmacological profile. Structurally, doxepin contains a dibenzoxepin ring system and an unsaturated side chain bearing a tertiary amine, making it a useful example for applying common organic reactions in multistep synthesis.
In this practice problem, we will discuss how to synthesize doxepin starting from 2-methylbenzoic acid (o-Toluic acid).
As with all examples in this section, this discussion is presented solely for educational purposes and is not intended to represent a practical or real-world method of preparation.
Practice
Identify the reagents and intermediates in the following synthesis of Doxepin. Draw the mechanism of the formation of intermediate 6 from intermediate 5.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to the answers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps, and the powerful set of Organic Chemistry 1 and 2 Summary Study Guides.
