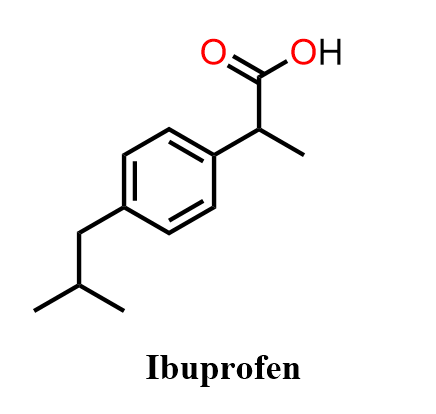
 Ibuprofen, the active ingredient in brands such as Advil, is a widely used non-steroidal anti-inflammatory drug (NSAID) that reduces pain, fever, and inflammation by inhibiting the cyclooxygenase (COX) enzymes responsible for prostaglandin synthesis. It is one of the most common over-the-counter medications worldwide and an excellent example of how fundamental organic reactions can assemble a biologically active molecule.
Ibuprofen, the active ingredient in brands such as Advil, is a widely used non-steroidal anti-inflammatory drug (NSAID) that reduces pain, fever, and inflammation by inhibiting the cyclooxygenase (COX) enzymes responsible for prostaglandin synthesis. It is one of the most common over-the-counter medications worldwide and an excellent example of how fundamental organic reactions can assemble a biologically active molecule.
This synthesis incorporates several major reaction types discussed throughout the course:
-
Friedel–Crafts Acylation (Electrophilic Aromatic Substitution) – used twice to introduce the aromatic ketones.
-
Wolff–Kishner and Clemmensen Reductions – used to convert an aryl ketone into the corresponding alkylbenzene.
-
Reduction of Ketones to Alcohols – forming the secondary alcohol intermediate.
-
Conversion of Alcohols to Alkyl Halides – using reagents such as HBr, PBr₃, HCl, or SOCl₂.
-
Grignard Reactions with CO₂ or SN2 formation of nitriles, followed by nitrile hydrolysis – used to install the carboxylic acid group.
Together, these steps demonstrate how electrophilic aromatic substitution, carbonyl reductions, alcohol–halide interconversion, organometallic carboxylation, and nitrile chemistry come together in the multistep preparation of Ibuprofen.
Practice
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to the answers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps, and the powerful set of Organic Chemistry 1 and 2 Summary Study Guides.
