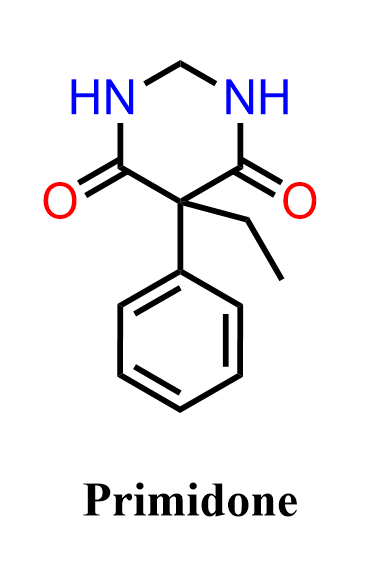
Primidone is an anticonvulsant (antiepileptic) medication used primarily to treat seizure disorders, including generalized tonic-clonic and partial seizures. Chemically, it is a barbiturate-related compound (a deoxybarbiturate), meaning it is structurally similar to barbiturates but has a slightly different pharmacological profile. In the body, primidone is partially metabolized into phenobarbital and phenylethylmalonamide (PEMA), which also contribute to its anticonvulsant effects by reducing neuronal excitability in the central nervous system. Because of this relationship, it is often discussed alongside barbiturates in medicinal and organic chemistry contexts, especially when analyzing heterocyclic urea derivatives and carbonyl chemistry.
In the following practice problem, we will focus not on the pharmacology but on the organic synthesis aspect of primidone. Specifically, we will work through a retrosynthetic and forward synthesis pathway starting from simple aromatic feedstocks such as benzene and toluene, applying key reactions like electrophilic aromatic substitution, side-chain functionalization, and carbonyl/urea formation. The goal is to practice multistep synthesis planning – identifying functional group transformations, choosing appropriate reagents, and understanding how simple aromatic hydrocarbons can be elaborated into a complex pharmaceutical molecule.
As with all examples in this section, this discussion is presented solely for educational purposes and is not intended to represent a practical or real-world method of preparation.
Practice
Below are shown two routes for preparing Primidone from Toluene. Identify the missing reagents and intermediates in the synthesis of Primidone.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to the answers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps, and the powerful set of Organic Chemistry 1 and 2 Summary Study Guides.
