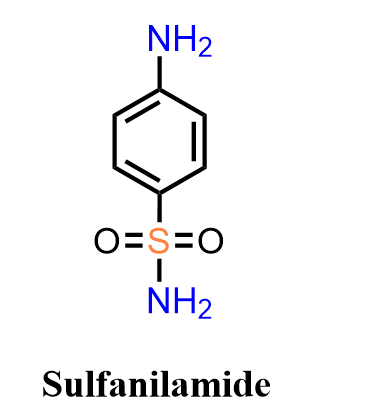
 Sulfanilamide, one of the earliest synthetic antimicrobial agents, holds a foundational place in medicinal chemistry because it introduced the concept of selectively inhibiting bacterial metabolism. As a member of the sulfonamide class, its structure mimics p-aminobenzoic acid (PABA), allowing it to competitively block dihydropteroate synthase and disrupt folate biosynthesis in bacteria. Although largely superseded by more advanced sulfa drugs, sulfanilamide remains an iconic molecule whose simple aromatic-sulfonamide framework is still referenced in discussions of structure-activity relationships, early antibiotic development, and the origins of chemotherapy.
Sulfanilamide, one of the earliest synthetic antimicrobial agents, holds a foundational place in medicinal chemistry because it introduced the concept of selectively inhibiting bacterial metabolism. As a member of the sulfonamide class, its structure mimics p-aminobenzoic acid (PABA), allowing it to competitively block dihydropteroate synthase and disrupt folate biosynthesis in bacteria. Although largely superseded by more advanced sulfa drugs, sulfanilamide remains an iconic molecule whose simple aromatic-sulfonamide framework is still referenced in discussions of structure-activity relationships, early antibiotic development, and the origins of chemotherapy.
In this exercise, we will discuss the synthesis of sulfanilamide, which brings together several important reaction types in aromatic and amine chemistry. These include:
-
Electrophilic Aromatic Substitution (EAS) – used to introduce substituents onto the benzene ring. The synthesis and subsequent reactions of aniline are especially relevant here, since the –NH₂ group strongly activates the ring and directs new substituents to the ortho and para positions.
-
Acylation and amide-forming reactions – essential steps for constructing key carbon–nitrogen bonds and for temporarily modifying the reactivity of the amino group through protection and deprotection sequences.
-
Sulfonation of the aromatic ring – an EAS reaction that installs the sulfonyl group, which is crucial for forming sulfonamides such as sulfanilamide.
Practice
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to the answers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps, and the powerful set of Organic Chemistry 1 and 2 Summary Study Guides.
