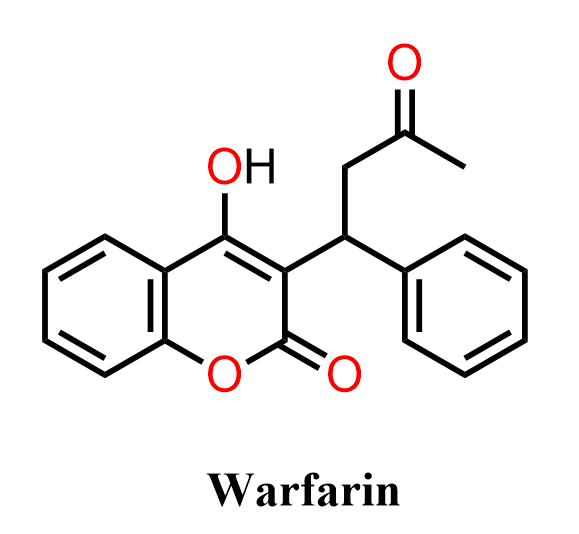
 Warfarin is an anticoagulant medication widely used to prevent and treat blood clots, including conditions such as deep vein thrombosis and pulmonary embolism. Chemically, it is a coumarin derivative, featuring a 4-hydroxycoumarin core linked to a substituted side chain. Its biological activity arises from inhibition of vitamin K–dependent clotting factor synthesis, making it a classic example of how relatively simple aromatic frameworks can be elaborated into highly active pharmaceutical agents. Because of its structure, warfarin is often discussed in organic chemistry in the context of coumarin chemistry, carbonyl reactivity, and conjugated aromatic systems.
Warfarin is an anticoagulant medication widely used to prevent and treat blood clots, including conditions such as deep vein thrombosis and pulmonary embolism. Chemically, it is a coumarin derivative, featuring a 4-hydroxycoumarin core linked to a substituted side chain. Its biological activity arises from inhibition of vitamin K–dependent clotting factor synthesis, making it a classic example of how relatively simple aromatic frameworks can be elaborated into highly active pharmaceutical agents. Because of its structure, warfarin is often discussed in organic chemistry in the context of coumarin chemistry, carbonyl reactivity, and conjugated aromatic systems.
In the following practice problem, we will focus on the organic synthesis of warfarin rather than its pharmacological properties. Specifically, we will examine how the key intermediate, 4-hydroxycoumarin, can be prepared and then transformed into warfarin through a C–C bond-forming reaction (typically a Michael-type addition) with an α,β-unsaturated ketone. The goal is to practice multistep synthesis design, including electrophilic aromatic substitution, condensation reactions, cyclization, and enolate chemistry, while understanding how these transformations build up the coumarin framework and ultimately lead to the final drug molecule.
As with all examples in this section, this discussion is presented solely for educational purposes and is not intended to represent a practical or real-world method of preparation.
Practice
Below are two pathways (A and B) for synthesizing warfarin, both proceeding via the formation of the intermediate 4-hydroxycoumarin. Determine whether the coumarin is aromatic or not, and draw the curved-arrow mechanisms for syntheses A and B.

This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to the answers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps, and the powerful set of Organic Chemistry 1 and 2 Summary Study Guides.
