Phenol is the simplest member of this family of compounds. It consists of a benzene ring bearing a single hydroxyl group, and many important aromatic compounds can be viewed as derivatives of phenol.
As more hydroxyl groups are introduced onto the aromatic ring, a variety of related structures appear. For example, compounds with two hydroxyl groups include the three dihydroxybenzene isomers: catechol (1,2-dihydroxybenzene), resorcinol (1,3-dihydroxybenzene), and hydroquinone (1,4-dihydroxybenzene).
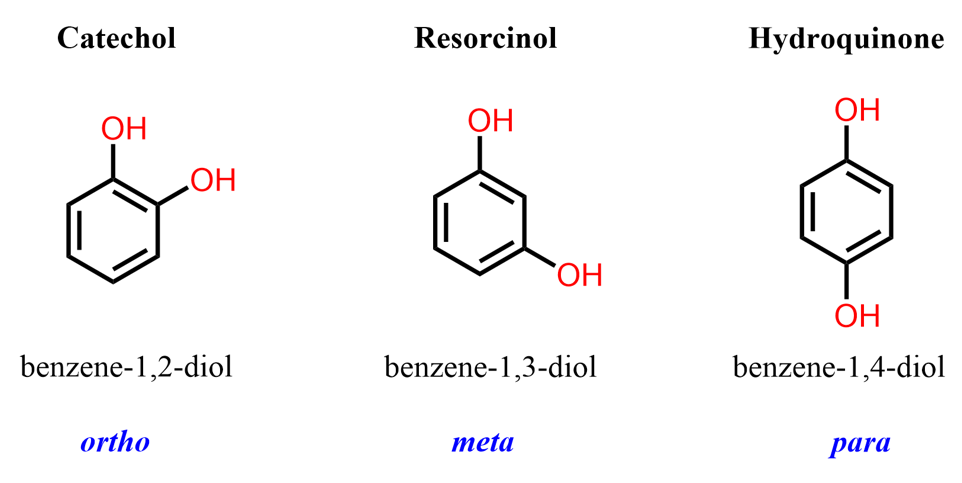
Among these, catechol is of particular interest because the two adjacent hydroxyl groups give it unique chemical and biological properties.
Many biologically active molecules, such as adrenaline, dopamine, and norepinephrine, share this catechol motif. The catechol ring consists of a benzene ring bearing two adjacent hydroxyl groups, and it plays an important role in the chemistry and biological activity of numerous natural compounds and pharmaceuticals.
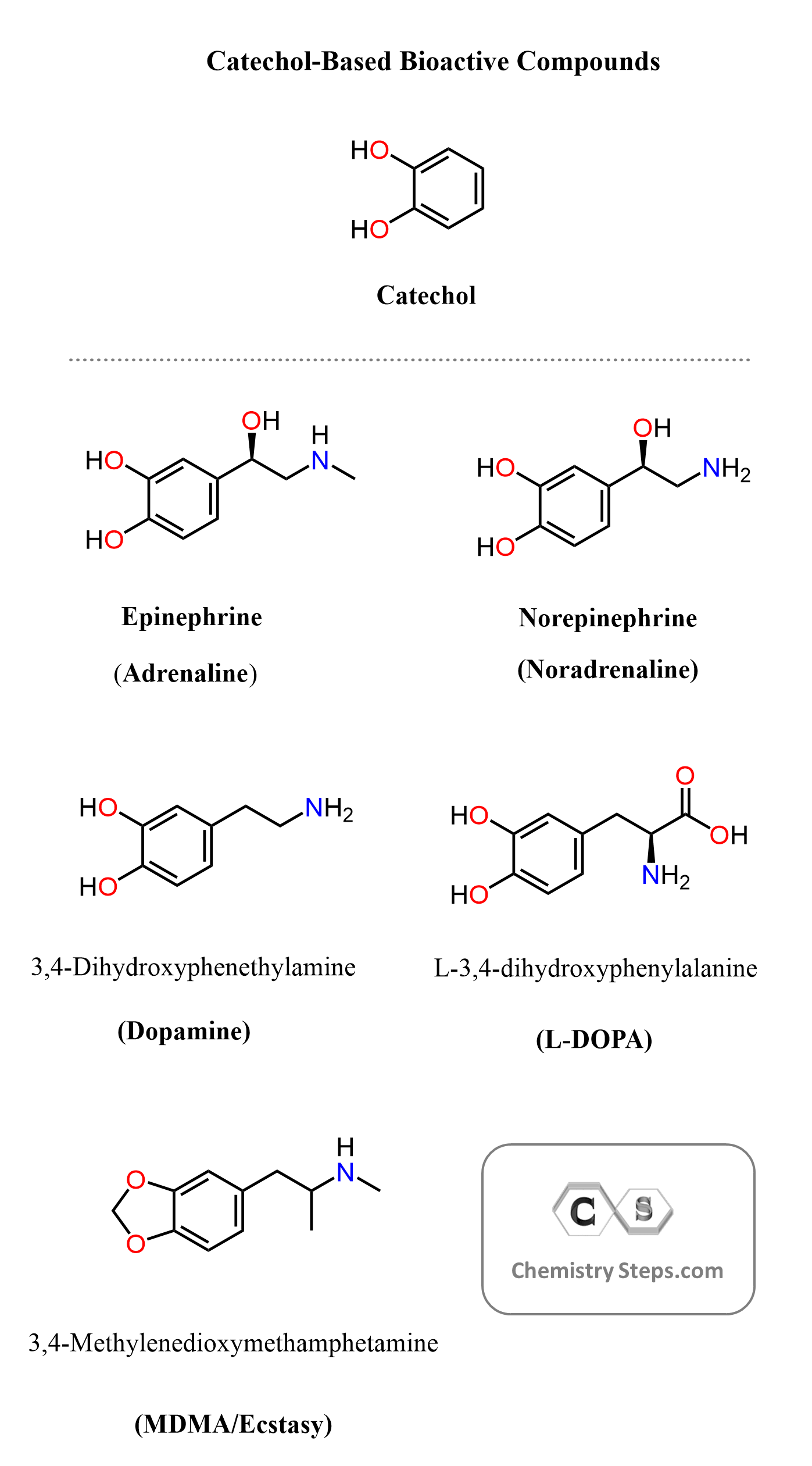
The catechol unit appears in many neurotransmitters, hormones, and drugs that affect the nervous and cardiovascular systems. Some of the most well-known examples include the natural catecholamines dopamine, norepinephrine, and epinephrine. These molecules regulate important physiological processes such as mood, alertness, blood pressure, and the body’s fight-or-flight response.
In addition to naturally occurring compounds, many therapeutic drugs are designed around the catechol motif because it interacts effectively with adrenergic and dopaminergic receptors. For example, drugs such as dobutamine and isoproterenol mimic the activity of natural catecholamines and are used in the treatment of heart conditions and circulatory problems.
Catechol structures are also common in plant-derived natural products, particularly polyphenols such as catechin and hydroxytyrosol, which are known for their antioxidant properties.
Because the catechol group is so versatile chemically – capable of hydrogen bonding, metal binding, and oxidation-reduction reactions – it appears in a wide range of biologically active molecules.
In this post, we will focus on the organic synthesis of several well-known compounds that are related to this structural motif, including adrenaline, dopamine, and ecstasy.

These examples illustrate different synthetic strategies used to construct molecules that either contain a catechol ring or are closely related to it, and they highlight how small structural changes can lead to dramatically different biological effects.
As with all examples in this section, this discussion is presented solely for educational purposes and is not intended to represent a practical or real-world method of preparation.
Practice
Synthesis of Adrenaline
Identify the missing reagents and intermediates in the synthesis of Adrenaline from Catechol.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to the answers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps, and the powerful set of Organic Chemistry 1 and 2 Summary Study Guides.
