We have seen some important transformations of carboxylic acid derivatives, including the hydrolysis of nitriles to carboxylic acids under acidic or basic conditions. So, one question you may encounter on homework assignments is the reverse conversion, that is, going from a carboxylic acid to a nitrile.
Unlike nitrile hydrolysis, this transformation cannot be done in a single step. Instead, the strategy involves converting the carboxylic acid into a more reactive derivative and then introducing nitrogen in a controlled way.
The most common approach is to first convert the carboxylic acid into a primary amide and then dehydrate it to the corresponding nitrile. So, we first install nitrogen at the carbonyl carbon, then remove water to form the carbon-nitrogen triple bond.
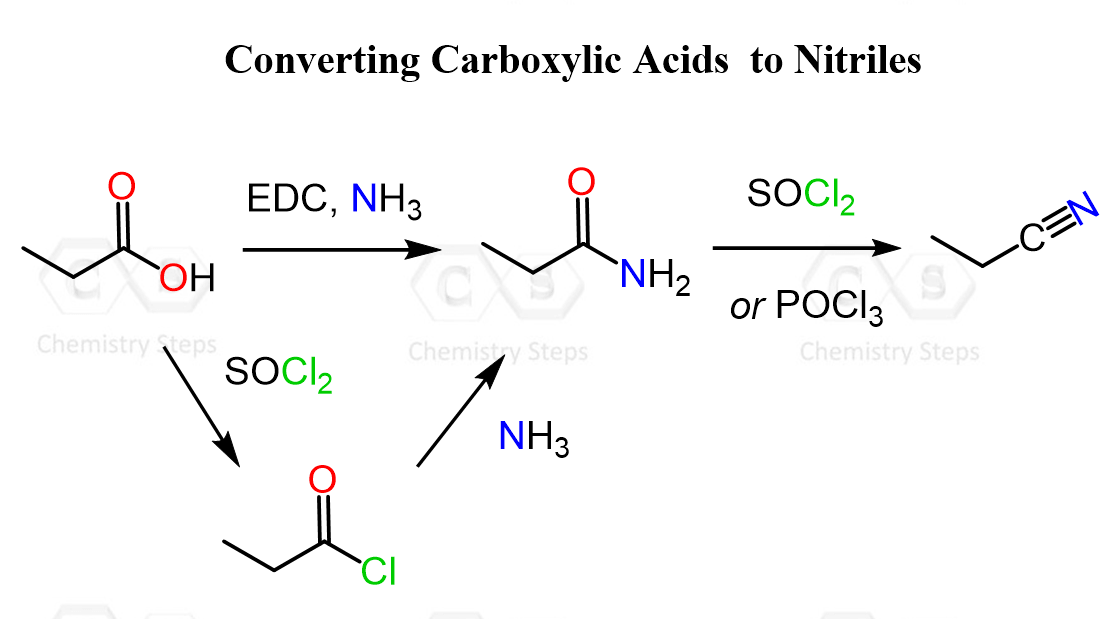
Typical reagents for the dehydration step include thionyl chloride (SOCl₂), phosphorus pentoxide (P₂O₅), or phosphoryl chloride (POCl₃), which promote elimination of water from the amide to form the nitrile.
Nitrile via Oximes
Another, less common route involves reducing the carboxylic acid to an aldehyde, converting the aldehyde into an oxime, and then dehydrating the oxime to a nitrile. The key idea here is the conversion of the oxime to a nitrile via an Appel-type transformation, where triphenylphosphine (PPh₃) and carbon tetrachloride (CCl₄) are traditionally used to promote dehydration.
However, this classical protocol has important drawbacks. CCl₄ is toxic, environmentally hazardous, and associated with ozone depletion, while the stoichiometric use of PPh₃ generates large amounts of triphenylphosphine oxide (Ph₃PO), complicating purification.
An important improvement was reported in AIP Conference Proceedings 2063, 030019 (2019), where oxalyl chloride ((COCl)₂) is used instead of CCl₄.

While this route is longer, it is useful for connecting different topics, especially if you are working through carbonyl and oxime chemistry.
Overall, the key takeaway is that converting a carboxylic acid to a nitrile is not a direct substitution, but rather a sequence of transformations: activation, nitrogen incorporation, and dehydration.
Organic Chemistry Reaction Maps
Never struggle again to figure out how to convert an alkyl halide to an alcohol, an alkene to an alkyne, a nitrile to a ketone, a ketone to an aldehyde, and more! The comprehensive powerfull Reaction Maps of organic functional group transformations are here!
Check Also
- Preparation of Carboxylic Acids
- Naming Carboxylic Acids
- Naming Nitriles
- Naming Esters
- Naming Carboxylic Acid Derivatives – Practice Problems
- Fischer Esterification
- Ester Hydrolysis by Acid and Base-Catalyzed Hydrolysis
- What is Transesterification?
- Esters Reaction with Amines – The Aminolysis Mechanism
- Ester Reactions Summary and Practice Problems
- Preparation of Acyl (Acid) Chlorides (ROCl)
- Reactions of Acid Chlorides (ROCl) with Nucleophiles
- Reaction of Acyl Chlorides with Grignard and Gilman (Organocuprate) Reagents
- Reduction of Acyl Chlorides by LiAlH4, NaBH4, and LiAl(OtBu)3H
- Preparation and Reaction Mechanism of Carboxylic Anhydrides
- Amides – Structure and Reactivity
- Naming Amides
- Amides Hydrolysis: Acid and Base-Catalyzed Mechanism
- Amide Dehydration Mechanism by SOCl2, POCl3, and P2O5
- Amide Reduction Mechanism by LiAlH4
- Amides Preparation and Reactions Summary
- Amides from Carboxylic Acids-DCC and EDC Coupling
- The Mechanism of Nitrile Hydrolysis To Carboxylic Acid
- Nitrile Reduction Mechanism with LiAlH4 and DIBAL to Amine or Aldehyde
- The Mechanism of Grignard and Organolithium Reactions with Nitriles
- Carboxylic Acids to Ketones
- Esters to Ketones
- Carboxylic Acids and Their Derivatives Practice Problems
- Carboxylic Acids and Their Derivatives Quiz

